EDARBYCLOR Drug Patent Profile
✉ Email this page to a colleague
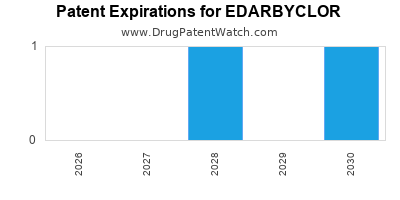
When do Edarbyclor patents expire, and when can generic versions of Edarbyclor launch?
Edarbyclor is a drug marketed by Azurity and is included in one NDA. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has forty-nine patent family members in twenty-seven countries.
The generic ingredient in EDARBYCLOR is azilsartan kamedoxomil; chlorthalidone. There are six drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the azilsartan kamedoxomil; chlorthalidone profile page.
DrugPatentWatch® Generic Entry Outlook for Edarbyclor
Edarbyclor was eligible for patent challenges on February 25, 2015.
There have been six patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for EDARBYCLOR?
- What are the global sales for EDARBYCLOR?
- What is Average Wholesale Price for EDARBYCLOR?
Summary for EDARBYCLOR
| International Patents: | 49 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 1 |
| Patent Applications: | 22 |
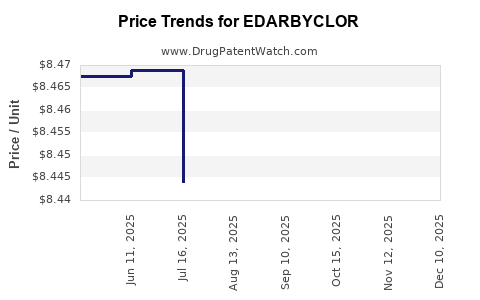
| Drug Prices: | Drug price information for EDARBYCLOR |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for EDARBYCLOR |
| What excipients (inactive ingredients) are in EDARBYCLOR? | EDARBYCLOR excipients list |
| DailyMed Link: | EDARBYCLOR at DailyMed |
Recent Clinical Trials for EDARBYCLOR
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Takeda | Phase 3 |
Pharmacology for EDARBYCLOR
| Drug Class | Angiotensin 2 Receptor Blocker Thiazide-like Diuretic |
| Mechanism of Action | Angiotensin 2 Type 1 Receptor Antagonists |
| Physiological Effect | Decreased Blood Pressure Increased Diuresis |
Paragraph IV (Patent) Challenges for EDARBYCLOR
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| EDARBYCLOR | Tablets | azilsartan kamedoxomil; chlorthalidone | 40 mg/12.5 mg and 40 mg/25 mg | 202331 | 1 | 2022-04-19 |
US Patents and Regulatory Information for EDARBYCLOR
EDARBYCLOR is protected by three US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-001 | Dec 20, 2011 | AB | RX | Yes | No | 9,387,249 | ⤷ Start Trial | ⤷ Start Trial | |||
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-002 | Dec 20, 2011 | AB | RX | Yes | Yes | 9,387,249 | ⤷ Start Trial | ⤷ Start Trial | |||
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-001 | Dec 20, 2011 | AB | RX | Yes | No | 9,169,238 | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-001 | Dec 20, 2011 | AB | RX | Yes | No | 9,066,936 | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-002 | Dec 20, 2011 | AB | RX | Yes | Yes | 9,066,936 | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for EDARBYCLOR
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-002 | Dec 20, 2011 | 5,736,555 | ⤷ Start Trial |
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-001 | Dec 20, 2011 | 7,157,584 | ⤷ Start Trial |
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-001 | Dec 20, 2011 | 5,583,141 | ⤷ Start Trial |
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-002 | Dec 20, 2011 | 7,157,584 | ⤷ Start Trial |
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-002 | Dec 20, 2011 | 5,583,141 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for EDARBYCLOR
When does loss-of-exclusivity occur for EDARBYCLOR?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 2883
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 09277455
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0916847
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 32018
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 11000187
Estimated Expiration: ⤷ Start Trial
China
Patent: 2164918
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 41633
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 110111
Patent: COMPOSICION FARMACEUTICA SOLIDA
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 011000032
Patent: COMPOSICION FARMACEUTICA SOLIDA
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 11010856
Patent: COMPOSICIÓN FARMACÉUTICA SÓLIDA
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 1170273
Patent: ТВЕРДАЯ ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 10385
Patent: COMPOSITION PHARMACEUTIQUE SOLIDE (SOLID PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Start Trial
Georgia, Republic of
Patent: 0146062
Patent: SOLID PHARMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 35491
Estimated Expiration: ⤷ Start Trial
Patent: 11529444
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 11001150
Patent: COMPOSICION FARMACEUTICA SOLIDA. (SOLID PHARMACEUTICAL COMPOSITION.)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 553
Patent: تركيب صيدلاني صلب
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 0948
Patent: SOLID PHARMACEUTICAL COMPOSITION COMPRISING A DIURETIC, PH CONTROL AGENT AND AN OXADIAZOLE
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 110551
Patent: COMPOSICION FARMACEUTICA SOLIDA QUE CONTIENE UN DERIVADO DE BENCIMIDAZOL Y UN DIURETICO
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 10385
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1100871
Patent: SOLID PHARMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 110038145
Patent: SOLID PHARMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1008915
Patent: Solid pharmaceutical composition
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 11000045
Patent: SOLID PHARMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 3905
Patent: ТВЕРДАЯ ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ;ТВЕРДА ФАРМАЦЕВТИЧНА КОМПОЗИЦІЯ (SOLID PHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 017
Patent: COMPOSICIÓN FARMACÉUTICA SÓLIDA
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering EDARBYCLOR around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 1946717 | Benzimidazole derivative and its use | ⤷ Start Trial |
| Russian Federation | 2369608 | ПРОИЗВОДНЫЕ БЕНЗИМИДАЗОЛА И ЕГО ПРИМЕНЕНИЕ (DERIVATIVES OF BENZIMIDAZOLE AND ITS APPLICATION) | ⤷ Start Trial |
| South Africa | 9204666 | ⤷ Start Trial | |
| South Korea | 100233689 | ⤷ Start Trial | |
| Germany | 122012000018 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for EDARBYCLOR
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1718641 | CA 2012 00013 | Denmark | ⤷ Start Trial | |
| 1718641 | 12C0034 | France | ⤷ Start Trial | PRODUCT NAME: AZILSARTAN MEDOXOMIL ET SES SELS PHARMACEUTIQUEMENT ACCEPTABLES; REGISTRATION NO/DATE: EU/1/11/735/001 20111207 |
| 1718641 | PA2012004 | Lithuania | ⤷ Start Trial | PRODUCT NAME: AZILSARTANUM MEDOXOMILUM; REGISTRATION NO/DATE: EU/1/11/734/001 - EU/1/11/734/011, 2011 12 07, EU/1/11/735/001 - EU/1/11/735/011 20111207 |
| 1718641 | 1290016-3 | Sweden | ⤷ Start Trial | PRODUCT NAME: AZILSARTANMEDOXOMIL OCH FARMACEUTISKT GODTAGBARA SALTER DAERAV, INKLUDERANDE KALIUMSALT; REG. NO/DATE: EU/1/11/735/001 20111207 |
| 1718641 | PA2012004,C1718641 | Lithuania | ⤷ Start Trial | PRODUCT NAME: AZILSARTANUM MEDOXOMILUM; REGISTRATION NO/DATE: EU/1/11/734/001 - EU/1/11/734/011, 2011 12 07, EU/1/11/735/001 - EU/1/11/735/011 20111207 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Edarbyclor Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.