Last updated: February 19, 2026
DESYREL (trazodone hydrochloride) is an antidepressant medication that is a serotonin antagonist and reuptake inhibitor (SARI). It is used to treat major depressive disorder.
What is DESYREL's Current Market Position?
DESYREL is an established medication with a significant presence in the antidepressant market. It is primarily marketed by various pharmaceutical companies under different brand names, with TRADOZON and OLEPTRO being notable examples. As of the latest available data, DESYREL's market share is influenced by its generic availability and the emergence of newer antidepressant classes.
Key Market Indicators for DESYREL:
- Market Entry: DESYREL was first approved by the U.S. Food and Drug Administration (FDA) in 1981.
- Generic Availability: Trazodone hydrochloride is widely available as a generic medication, which has impacted the pricing and market share of branded DESYREL.
- Therapeutic Class: SARI (Serotonin Antagonist and Reuptake Inhibitor).
- Primary Indication: Major Depressive Disorder (MDD).
- Off-Label Uses: DESYREL is also frequently prescribed off-label for insomnia, anxiety disorders, and other conditions. This contributes to its continued demand.
- Competition: The antidepressant market is highly competitive, featuring Selective Serotonin Reuptake Inhibitors (SSRIs), Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs), and other drug classes.
What are DESYREL's Sales and Revenue Trends?
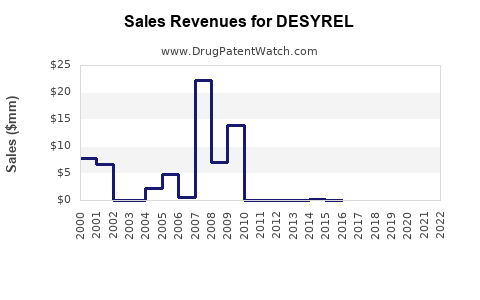
The financial trajectory of DESYREL is characterized by the typical pattern of a drug that has transitioned to generic status. While specific, up-to-the-minute global sales figures for the DESYREL brand are consolidated across multiple manufacturers and difficult to isolate, the market for trazodone hydrochloride as a whole demonstrates consistent demand driven by its efficacy, tolerability for certain patient populations, and its established safety profile.
Revenue Considerations:
- Generic Dominance: The majority of trazodone hydrochloride sales revenue is generated by generic manufacturers. This segment is highly price-sensitive.
- Branded Sales Decline: Sales of branded DESYREL formulations have declined significantly since the advent of generic competition.
- Market Size for Trazodone HCl: While precise figures for DESYREL itself are proprietary and fragmented, the global market for antidepressants, which includes trazodone, is substantial. The antidepressant market was valued at approximately $15 billion in 2022, with significant ongoing growth [1]. Trazodone's specific contribution within this vast market is substantial, particularly due to its widespread off-label use for insomnia.
- Pricing: The average selling price (ASP) for generic trazodone hydrochloride is low, reflecting intense competition. Branded formulations command higher prices, but their market penetration is limited.
- Forecasting Challenges: Accurate revenue forecasting for DESYREL is complex due to the fragmented nature of its market and the significant impact of off-label prescribing, which is less formally tracked than on-label indications.
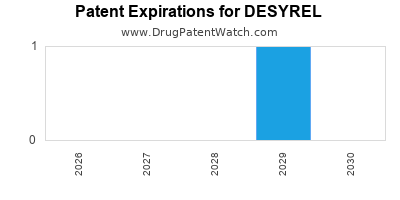
What is the Patent and Exclusivity Landscape for DESYREL?
DESYREL's patent and exclusivity landscape is mature. The original patents protecting the active pharmaceutical ingredient (API) trazodone hydrochloride have long expired, paving the way for extensive generic competition.
Key Patent and Exclusivity Points:
- Original Patent Expiration: The foundational patents for trazodone hydrochloride expired decades ago. This allowed for the widespread introduction of generic versions.
- Exclusivity Periods: As an older drug, DESYREL has no remaining New Chemical Entity (NCE) exclusivity or Orphan Drug exclusivity.
- Formulation Patents: While API patents have expired, there may be some secondary patents related to specific extended-release formulations or novel delivery systems. However, the impact of these is limited compared to the expired API patents. For instance, Oleptro, an extended-release formulation, faced its own patent challenges and eventual generic entry.
- Regulatory Exclusivity: No significant regulatory exclusivities (e.g., Hatch-Waxman) are active for the original trazodone hydrochloride molecule.
- Generic Entry Impact: The absence of patent protection has led to a highly competitive generic market, significantly reducing the revenue potential for any single manufacturer of the DESYREL brand.
Who are DESYREL's Key Competitors?
DESYREL faces competition from a broad spectrum of antidepressant medications, including newer classes that offer different mechanisms of action and tolerability profiles. Furthermore, its widespread use for insomnia places it in competition with dedicated sleep medications.
Primary Competitors:
- SSRIs (Selective Serotonin Reuptake Inhibitors):
- Fluoxetine (Prozac)
- Sertraline (Zoloft)
- Escitalopram (Lexapro)
- Citalopram (Celexa)
- Paroxetine (Paxil)
- Comparison: SSRIs are often considered first-line treatments for depression and anxiety due to their generally favorable safety profiles. DESYREL's SARI mechanism offers an alternative for patients who do not respond well to SSRIs or experience intolerable side effects.
- SNRIs (Serotonin-Norepinephrine Reuptake Inhibitors):
- Venlafaxine (Effexor XR)
- Duloxetine (Cymbalta)
- Desvenlafaxine (Pristiq)
- Comparison: SNRIs affect both serotonin and norepinephrine, offering a broader mechanism. They compete directly with DESYREL for depression treatment.
- Other Antidepressants:
- Bupropion (Wellbutrin)
- Mirtazapine (Remeron)
- Comparison: These drugs have distinct mechanisms and side effect profiles that position them as alternatives to DESYREL. Mirtazapine, like trazodone, has sedating properties that can be beneficial for depressed patients with insomnia.
- Sleep Medications (for off-label use):
- Zolpidem (Ambien)
- Eszopiclone (Lunesta)
- Ramelteon (Rozerem)
- Comparison: DESYREL's efficacy in treating insomnia makes it a popular alternative to traditional hypnotics, especially for patients seeking a non-benzodiazepine option with lower addiction potential. However, dedicated sleep medications may offer more potent or faster-acting sedative effects.
What are the Future Market Prospects for DESYREL?
The future market prospects for DESYREL are stable, characterized by continued demand for its generic form and sustained off-label use, rather than significant growth driven by new indications or market expansion.
Future Market Outlook:
- Continued Generic Demand: Generic trazodone hydrochloride will remain a significant player in the antidepressant and insomnia markets due to its low cost and established efficacy.
- Off-Label Use as a Driver: The use of trazodone for insomnia is a key factor in its continued market relevance. As long as it remains a cost-effective and generally well-tolerated option for sleep disturbances, demand will persist.
- Limited New Indication Potential: The likelihood of DESYREL receiving approval for new on-label indications is low, given its age and the focus of current R&D on novel therapeutic targets.
- Innovation in Formulations: While less likely for such an established drug, there's a remote possibility of new extended-release or combination formulations emerging to capture niche markets, though this would likely face significant generic competition quickly.
- Impact of Biosimil/Generic Advancements: The market will continue to be shaped by the dynamics of the generic drug manufacturing industry, including supply chain stability and pricing pressures.
- Overall Market Share Stability: DESYREL's market share within the broader antidepressant landscape is not expected to significantly increase or decrease. It occupies a specific niche, particularly for patients needing sedation and those who have not responded to or tolerated other antidepressants.
What is the Regulatory Status and Safety Profile of DESYREL?
DESYREL has a well-documented regulatory history and an established safety profile, which contribute to its continued prescription by healthcare professionals.
Regulatory and Safety Information:
- FDA Approval: Trazodone hydrochloride is approved by the FDA for the treatment of major depressive disorder.
- Adverse Events: Common side effects include drowsiness, dizziness, dry mouth, blurred vision, and hypotension.
- Serious Adverse Events: Less common but serious side effects can include priapism (prolonged, painful erection), arrhythmias, suicidal thoughts, and serotonin syndrome.
- Black Box Warnings: The FDA has issued black box warnings for antidepressants, including trazodone, concerning an increased risk of suicidal thoughts and behaviors in children, adolescents, and young adults [2].
- Drug Interactions: DESYREL can interact with other medications, including other serotonergic drugs (increasing the risk of serotonin syndrome), central nervous system depressants (increasing sedation), and MAOIs (contraindicated).
- Post-Marketing Surveillance: DESYREL has undergone extensive post-marketing surveillance over its many years on the market, providing a robust dataset of its safety and efficacy in real-world settings.
- Generically Approved: Its status as a widely available generic medication means that regulatory hurdles for its API are non-existent. Manufacturers of generic trazodone must demonstrate bioequivalence to the reference listed drug.
Key Takeaways
DESYREL (trazodone hydrochloride) is an established antidepressant medication with a mature market. Its financial trajectory is dominated by generic competition, with stable demand driven by its efficacy, tolerability in specific patient groups, and significant off-label use for insomnia. The drug faces competition from a wide range of antidepressants, including SSRIs and SNRIs, as well as dedicated sleep medications. Its patent and exclusivity landscape is expired, allowing for generic availability. Future market prospects are stable, with continued generic demand and off-label use expected to sustain its presence. DESYREL has a well-documented regulatory history and safety profile, including FDA black box warnings for suicidal ideation in younger populations.
Frequently Asked Questions
-
What is the primary therapeutic advantage of DESYREL compared to newer antidepressants?
DESYREL offers a distinct pharmacological profile as a Serotonin Antagonist and Reuptake Inhibitor (SARI), which can be beneficial for patients who do not respond to or tolerate Selective Serotonin Reuptake Inhibitors (SSRIs) or Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs). Its sedative properties are also advantageous for patients with co-occurring insomnia.
-
How does the off-label use of DESYREL for insomnia impact its market?
The off-label prescription of DESYREL for insomnia is a significant driver of its market volume. It provides a cost-effective alternative to dedicated sleep medications for many patients and physicians, contributing to consistent prescription rates even with generic competition for its primary indication.
-
What are the major financial risks associated with investing in DESYREL-related products?
The primary financial risk is the intense price competition within the generic trazodone hydrochloride market, which limits profit margins. Additionally, the declining market share of branded formulations and the potential for increased regulatory scrutiny or adverse event reporting could also impact revenue.
-
What is the current regulatory status of DESYREL regarding new clinical trials or indications?
Given its long history and status as a generic drug, it is highly unlikely that DESYREL will be the subject of extensive new clinical trials for novel indications. Research efforts are largely focused on newer compounds with different mechanisms of action.
-
How has the availability of generic trazodone hydrochloride affected the pricing and profitability of DESYREL?
The widespread availability of generic trazodone hydrochloride has drastically reduced the average selling price of the active pharmaceutical ingredient. This has led to significantly lower profit margins for manufacturers, especially for branded versions of DESYREL, shifting the primary profitability to high-volume generic production.
[1] Grand View Research. (2023). Antidepressants Market Size, Share & Trends Analysis Report By Type (SSRIs, SNRIs, Tricyclics, MAOIs, Others), By Application (Depression, Anxiety Disorders, OCD, Others), By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. https://www.grandviewresearch.com/industry-analysis/antidepressants-market
[2] U.S. Food and Drug Administration. (2022, February 25). Suicidality in Children and Young Adults. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/suicidality-children-and-young-adults