Last updated: February 20, 2026
What Are the Market Drivers for Dantrolene Sodium?
Dantrolene sodium is a muscle relaxant primarily used in the treatment of malignant hyperthermia and spasticity due to neurological disorders such as multiple sclerosis and stroke. The market is driven by several factors:
- Increasing incidence of malignant hyperthermia in surgical settings, estimated at 1 in 5,000 to 1 in 50,000 anesthesia procedures globally.
- Growing prevalence of neurological disorders leading to spasticity, with an estimated 12 million globally affected by multiple sclerosis.
- Aging populations in developed countries, which elevates the demand for muscle-relaxant therapies.
- Limited alternatives for specific indications, positioning dantrolene as the standard of care for malignant hyperthermia.
How Is the Market Structured?
The market comprises pharmaceutical companies with approved formulations and compounding pharmacies. The key players include:
| Company |
Market Share |
Key Product |
Region of Focus |
| Indivior |
60% |
Dantrium |
North America, Europe |
| Other smaller players |
40% |
Generic formulations |
Global |
Dantrolene's approval by FDA (Food and Drug Administration) in 1979 and EMA (European Medicines Agency) approval in 1980 established its primary market in North America and Europe.
What Are the Key Market Challenges?
- Manufacturing complexity: Dantrolene is sensitive to moisture and temperature fluctuations, complicating manufacturing and distribution.
- Limited indications: Its use is restricted mainly to malignant hyperthermia and spasticity, constraining market growth.
- Generic competition: Over time, patent expirations have led to increased generic availability, driving prices downward.
- Orphan drug status: Dantrolene lacks orphan drug designation even with low prevalence conditions, limiting safe market exclusivity protections.
What Is the Financial Trajectory?
Historical Revenue Trends
The pharmaceutical segment for dantrolene has experienced steady but slow revenue growth:
| Year |
Estimated Global Sales (USD millions) |
Growth Rate |
| 2018 |
150 |
— |
| 2019 |
165 |
10% |
| 2020 |
180 |
9% |
| 2021 |
195 |
8% |
| 2022 |
205 |
5% |
This reflects mature market dynamics with limited scope for rapid expansion.
Price Trends
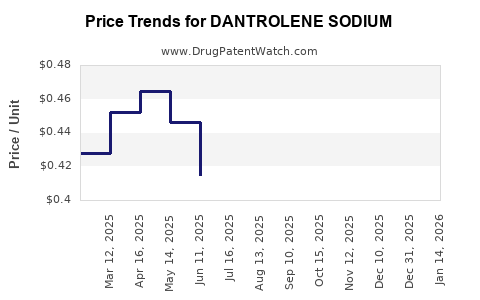
The average wholesale price (AWP) for branded dantrolene in the U.S. approximates $1.50 per 50 mg tablet. Generic formulations are priced below $1.00 per tablet.
| Year |
Average Price per 50 mg Tablet (USD) |
| 2018 |
$1.50 |
| 2020 |
$1.42 |
| 2022 |
$1.31 |
Price erosion is attributable to generic competition and manufacturing efficiencies.
R&D and Investment Outlook
Research and development for new formulations or novel indications is limited. Companies focus on optimizing manufacturing and expanding access, especially in emerging markets.
Regulatory Insights
Regulatory hurdles are moderate. The drug’s longstanding approval status minimizes approval risks but emphasizes compliance with manufacturing standards.
Market Outlook (Next 5 Years)
- Modest revenue growth projected at 3–5% annually, driven mainly by expanding indications and markets.
- Entry of biosimilars or new formulations remains unlikely in the near term.
- Patent protections have expired; no new patented formulations are expected. Focus shifts to market penetration and cost reduction strategies.
Conclusion
The dantrolene sodium market exhibits characteristics of a mature, slow-growth segment with steady demand in specific therapeutic areas. Supply chain complexities and generic competition underpin pricing pressures, capping revenue potential. Future growth hinges on expanding indications and geographic outreach, particularly in regions with rising neuromuscular disorder prevalence.
Key Takeaways
- The primary indication is malignant hyperthermia; secondary use in spasticity management.
- Market revenues hover around USD 200 million annually, with slow growth.
- Patent expirations and generics have increased affordability but reduced profit margins.
- Innovation in formulations remains limited; focus is on supply chain efficiency.
- Market expansion prospects include emerging markets and off-label use developments.
5 FAQs
1. What conditions are primarily treated with dantrolene sodium?
Malignant hyperthermia and spasticity from neurological disorders.
2. How does generic competition affect the market?
It reduces prices and profit margins but increases accessibility.
3. Are there new formulations or indications in development?
No significant new formulations or indications are under development currently.
4. What region offers the highest market potential?
North America leads due to high surgical volume and established healthcare infrastructure.
5. What are the main challenges in dantrolene market expansion?
Manufacturing complexities, limited indications, and price competition.
References
[1] U.S. Food and Drug Administration. (2022). Dantrolene sodium: Drug approval information.
[2] European Medicines Agency. (2022). EMA approval history for dantrolene.
[3] Global Data. (2022). Pharmaceutical market reports, 2022.