CRESTOR Drug Patent Profile
✉ Email this page to a colleague
When do Crestor patents expire, and when can generic versions of Crestor launch?
Crestor is a drug marketed by IPR and is included in one NDA.
The generic ingredient in CRESTOR is rosuvastatin calcium. There are forty-two drug master file entries for this compound. Forty-four suppliers are listed for this compound. Additional details are available on the rosuvastatin calcium profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Crestor
A generic version of CRESTOR was approved as rosuvastatin calcium by WATSON LABS INC on April 29th, 2016.
Summary for CRESTOR
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 64 |
| Clinical Trials: | 177 |
| Patent Applications: | 1,716 |
| Formulation / Manufacturing: | see details |
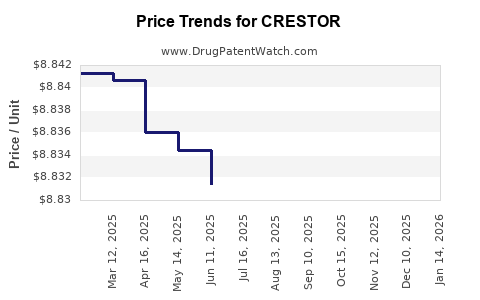
| Drug Prices: | Drug price information for CRESTOR |
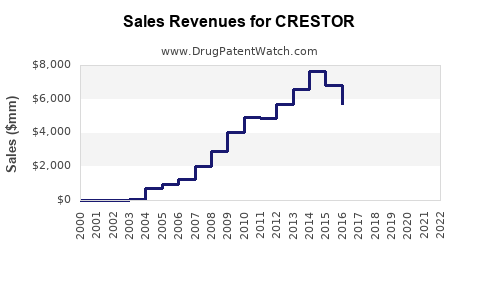
| Drug Sales Revenues: | Drug sales revenues for CRESTOR |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for CRESTOR |
| What excipients (inactive ingredients) are in CRESTOR? | CRESTOR excipients list |
| DailyMed Link: | CRESTOR at DailyMed |
Recent Clinical Trials for CRESTOR
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Wake Forest University Health Sciences | Phase 4 |
| University of Karachi | N/A |
| The Searle Company Limited | N/A |
Pharmacology for CRESTOR
| Drug Class | HMG-CoA Reductase Inhibitor |
| Mechanism of Action | Hydroxymethylglutaryl-CoA Reductase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for CRESTOR
Paragraph IV (Patent) Challenges for CRESTOR
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| CRESTOR | Tablets | rosuvastatin calcium | 5 mg, 10 mg, 20 mg and 40 mg | 021366 | 9 | 2007-08-13 |
US Patents and Regulatory Information for CRESTOR
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ipr | CRESTOR | rosuvastatin calcium | TABLET;ORAL | 021366-002 | Aug 12, 2003 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Ipr | CRESTOR | rosuvastatin calcium | TABLET;ORAL | 021366-005 | Aug 12, 2003 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Ipr | CRESTOR | rosuvastatin calcium | TABLET;ORAL | 021366-003 | Aug 12, 2003 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Ipr | CRESTOR | rosuvastatin calcium | TABLET;ORAL | 021366-004 | Aug 12, 2003 | AB | RX | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for CRESTOR
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Ipr | CRESTOR | rosuvastatin calcium | TABLET;ORAL | 021366-005 | Aug 12, 2003 | ⤷ Sign Up | ⤷ Sign Up |
| Ipr | CRESTOR | rosuvastatin calcium | TABLET;ORAL | 021366-003 | Aug 12, 2003 | ⤷ Sign Up | ⤷ Sign Up |
| Ipr | CRESTOR | rosuvastatin calcium | TABLET;ORAL | 021366-002 | Aug 12, 2003 | ⤷ Sign Up | ⤷ Sign Up |
| Ipr | CRESTOR | rosuvastatin calcium | TABLET;ORAL | 021366-002 | Aug 12, 2003 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for CRESTOR
See the table below for patents covering CRESTOR around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Portugal | 1144389 | ⤷ Sign Up | |
| Japan | 3267960 | ⤷ Sign Up | |
| Slovakia | 11782000 | FARMACEUTICKÁ KOMPOZÍCIA (PHARMACEUTICAL COMPOSITION) | ⤷ Sign Up |
| Australia | 6455900 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for CRESTOR
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0521471 | 03C0043 | France | ⤷ Sign Up | PRODUCT NAME: ROSUVASTATIN; NAT. REGISTRATION NO/DATE: NL 28 274 20030611; FIRST REGISTRATION: NL - 26872 20021106 |
| 0720599 | 92544 | Luxembourg | ⤷ Sign Up | PRODUCT NAME: EZETIMIBE EN COMBINAISON AVEC ROSUVASTATINE OU LEURS SELS PHARMACEUTIQUEMENT ACCEPTEES, Y COMPRIS ROSUVASTATINE SOUD FORME DE SEL DE ZINC; FIRST REGISTRATION DATE: 20140724 |
| 0720599 | C300688 | Netherlands | ⤷ Sign Up | PRODUCT NAME: EZETIMIBE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, EN ROSUVASTATINE; NAT. REGISTRATION NO/DATE: RVG114002-004RVG 114008-010 2014110811; FIRST REGISTRATION: 13-9663 - 9665 20140724 |
| 0521471 | C00521471/01 | Switzerland | ⤷ Sign Up | FORMER REPRESENTANTIVE: BOVARD AG PATENTANWAELTE, CH |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


