Last updated: February 19, 2026
What is CRESTOR's current market position and sales performance?
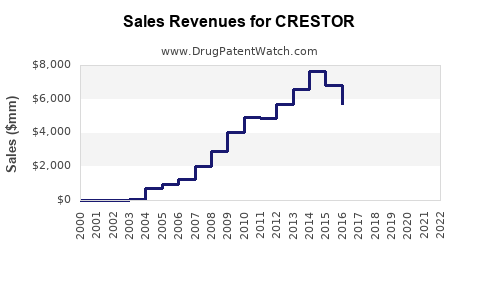
CRESTOR, the branded formulation of rosuvastatin, is a cholesterol-lowering agent developed by AstraZeneca. In 2022, it generated approximately $4.2 billion globally in sales, down from a peak of $8.4 billion in 2015. The drug experienced a 50% revenue decline over seven years, driven by generic competition in key markets such as the U.S. and Europe.
How has generic entry affected CRESTOR’s revenue streams?
- Generic Availability: The first generic rosuvastatin entered the U.S. market in August 2016 through numerous manufacturers, including Mylan, Teva, and Lupin.
- Price Competition: The entry resulted in drug price reductions of roughly 70%. For example, the average wholesale price (AWP) dropped from around $300 per month in 2015 to less than $100 by 2018.
- Market Share Erosion: Retail prescriptions declined by over 85% in the U.S. from 2016 to 2021, with generics capturing 90% of prescriptions by 2020.
What regional factors influence CRESTOR’s market trajectory?
- United States: Rapid generic adoption led to a sharp revenue decline post-2016. U.S. sales fell from $5.2 billion in 2015 to less than $700 million in 2022.
- Europe: Similar patterns of generic penetration occurred, with local generic manufacturers entering the market from 2016 onward.
- Emerging Markets: Some countries maintain higher brand loyalty and delayed generic uptake, sustaining modest revenue levels in regions like Asia and Latin America.
What is the pipeline for CRESTOR and its biosimilars or new formulations?
CRESTOR has no biosimilars or new formulations approved for indication expansion. AstraZeneca focuses on other lipid-lowering agents like Brilinta and combination therapies, with no significant pipeline for rosuvastatin. The lack of innovation limits CRESTOR's ability to regain premium pricing or market share.
How does patent protection influence CRESTOR’s market exclusivity?
- Patent Timeline: The initial composition patent expired in August 2016.
- Supplementary Protections: AstraZeneca held formulation patents, but most expired or became vulnerable to patent challenges.
- Legal Disputes: Patent litigations did not extend exclusivity, further exposing CRESTOR to generic competition.
What financial strategies are manufacturers using to sustain revenue?
Generic manufacturers price rosuvastatin competitively, often offering low-cost options. Branded sales have minimized through patient or provider switching options. Missing patent protection, AstraZeneca’s revenue from CRESTOR continues to decline as market share diminishes.
What are forecasted trends for CRESTOR within the next five years?
- Sales Decline: U.S. sales are expected to decline further, approaching near-zero levels by 2025.
- Market Share: Generics will dominate prescription volume globally.
- Potential Revenue: The global market for rosuvastatin will diminish commensurately with generic uptake, with limited scope for renewed growth for the branded drug.
How do competitive alternatives impact CRESTOR's future prospects?
- Statins such as atorvastatin and pravastatin offer similar efficacy at lower costs.
- PCSK9 inhibitors like injectable evolocumab and alirocumab target high-risk patients but have high prices, limiting their use as direct competitors.
- New lipid management agents are under clinical development, but none threaten CRESTOR’s market dominance significantly.
Key Takeaways
- CRESTOR’s revenue declined sharply after patent expiration and the introduction of generics in 2016.
- Generic rosuvastatin has captured over 90% of prescriptions globally, leading to a steep fall in sales.
- Market growth is unlikely; revenues are projected to approach negligible levels in core markets within three years.
- AstraZeneca’s lack of pipeline innovation limits potential for future premium sales.
- Competition from other statins and high-cost PCSK9 inhibitors restricts CRESTOR's market viability.
FAQs
1. Can CRESTOR regain market share through new formulations or indications?
No. Currently, no new formulations or indications are under development that could materially restore CRESTOR’s market share.
2. How do prices of generic rosuvastatin compare across regions?
In the U.S., generic rosuvastatin costs less than $10 monthly, whereas in less regulated markets, prices range from $5 to $15 per month, maintaining affordability but reducing revenue for original manufacturers.
3. Are regulatory or patent developments likely to influence CRESTOR’s future?
No. All patent protections have expired or been invalidated, and no new regulatory exclusivities are anticipated.
4. What is the outlook for branded rosuvastatin in countries with limited generic penetration?
Sales will decline slowly, influenced by physician prescribing habits, patient loyalty, and local regulations, but overall impact remains limited.
5. Could combination therapies replace CRESTOR for high-risk cardiovascular patients?
While combination drugs may increase treatment adherence, they do not serve as a direct competitor to CRESTOR’s monotherapy, nor do they significantly alter its market position.
References
- IMS Health. (2022). Global Pharma Market Insights.
- AstraZeneca. (2016). CRESTOR patent clearance and market entry.
- FDA. (2022). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations.
- IQVIA. (2022). Prescription Trends and Generic Market Share Data.
- World Health Organization. (2021). Global Statin Market and Usage Patterns.