COTELLIC Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Cotellic, and when can generic versions of Cotellic launch?
Cotellic is a drug marketed by Genentech Inc and is included in one NDA. There are seven patents protecting this drug.
This drug has one hundred and eighty patent family members in forty-three countries.
The generic ingredient in COTELLIC is cobimetinib fumarate. One supplier is listed for this compound. Additional details are available on the cobimetinib fumarate profile page.
DrugPatentWatch® Generic Entry Outlook for Cotellic
Cotellic was eligible for patent challenges on November 10, 2019.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be December 30, 2036. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for COTELLIC
| International Patents: | 180 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 10 |
| Clinical Trials: | 35 |
| Patent Applications: | 1 |
| Drug Prices: | Drug price information for COTELLIC |
| What excipients (inactive ingredients) are in COTELLIC? | COTELLIC excipients list |
| DailyMed Link: | COTELLIC at DailyMed |
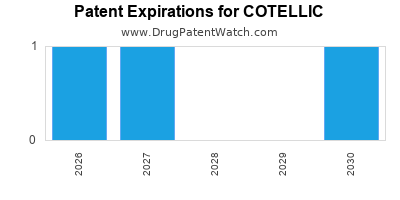

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for COTELLIC
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for COTELLIC
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Birmingham | Phase 2/Phase 3 |
| Cancer Research UK | Phase 2/Phase 3 |
| Royal Marsden NHS Foundation Trust | Phase 2/Phase 3 |
Pharmacology for COTELLIC
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Kinase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for COTELLIC
US Patents and Regulatory Information for COTELLIC
COTELLIC is protected by seven US patents and four FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of COTELLIC is ⤷ Sign Up.
This potential generic entry date is based on patent ⤷ Sign Up.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting COTELLIC
Immediate-release tablets containing combimetinib and methods of making and using the same
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Crystalline fumarate salt of (S)-[3,4-difluoro-2-(2-fluoro-4-iodophenylamino)phenyl] [3-hydroxy-3-(piperidin-2-yl) azetidin-1-yl]methanone
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Combination therapies
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Crystalline fumarate salt of (S)-[3,4-difluoro-2-(2-fluoro-4-iodophenylamino)phenyl] [3-hydroxy-3-(piperidin-2-yl) azetidin-1-yl]-methanone
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: AS A SINGLE AGENT FOR THE TREATMENT OF ADULT PATIENTS WITH HISTIOCYTIC NEOPLASMS
Azetidines as MEK inhibitors for the treatment of proliferative diseases
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Azetidines as MEK inhibitors for the treatment of proliferative diseases
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
FDA Regulatory Exclusivity protecting COTELLIC
TREATMENT OF ADULT PATIENTS WITH HISTIOCYTIC NEOPLASMS
Exclusivity Expiration: ⤷ Sign Up
INFORMATION ADDED TO SECTION 8.4 OF THE LABELING TO INCLUDE THE RESULT OF STUDY GO29665
Exclusivity Expiration: ⤷ Sign Up
TREATMENT OF ADULT PATIENTS WITH HISTIOCYTIC NEOPLASMS
Exclusivity Expiration: ⤷ Sign Up
PEDIATRIC EXCLUSIVITY
Exclusivity Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Genentech Inc | COTELLIC | cobimetinib fumarate | TABLET;ORAL | 206192-001 | Nov 10, 2015 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Genentech Inc | COTELLIC | cobimetinib fumarate | TABLET;ORAL | 206192-001 | Nov 10, 2015 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Genentech Inc | COTELLIC | cobimetinib fumarate | TABLET;ORAL | 206192-001 | Nov 10, 2015 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Genentech Inc | COTELLIC | cobimetinib fumarate | TABLET;ORAL | 206192-001 | Nov 10, 2015 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Genentech Inc | COTELLIC | cobimetinib fumarate | TABLET;ORAL | 206192-001 | Nov 10, 2015 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for COTELLIC
When does loss-of-exclusivity occur for COTELLIC?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 5483
Estimated Expiration: ⤷ Sign Up
Australia
Patent: 16288209
Estimated Expiration: ⤷ Sign Up
Patent: 21200202
Estimated Expiration: ⤷ Sign Up
Brazil
Patent: 2017028516
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 90222
Estimated Expiration: ⤷ Sign Up
Chile
Patent: 17003475
Estimated Expiration: ⤷ Sign Up
China
Patent: 7810183
Estimated Expiration: ⤷ Sign Up
Colombia
Patent: 18000086
Estimated Expiration: ⤷ Sign Up
Costa Rica
Patent: 180056
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 17264
Estimated Expiration: ⤷ Sign Up
Hong Kong
Patent: 52433
Estimated Expiration: ⤷ Sign Up
Israel
Patent: 6423
Estimated Expiration: ⤷ Sign Up
Patent: 5052
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 38950
Estimated Expiration: ⤷ Sign Up
Patent: 18519318
Estimated Expiration: ⤷ Sign Up
Patent: 21035967
Estimated Expiration: ⤷ Sign Up
Patent: 23025000
Estimated Expiration: ⤷ Sign Up
Malaysia
Patent: 2545
Estimated Expiration: ⤷ Sign Up
Mexico
Patent: 17017037
Estimated Expiration: ⤷ Sign Up
Morocco
Patent: 301
Estimated Expiration: ⤷ Sign Up
Peru
Patent: 180692
Estimated Expiration: ⤷ Sign Up
Philippines
Patent: 017502414
Estimated Expiration: ⤷ Sign Up
Russian Federation
Patent: 62181
Estimated Expiration: ⤷ Sign Up
Patent: 18103172
Estimated Expiration: ⤷ Sign Up
Patent: 21132394
Estimated Expiration: ⤷ Sign Up
Singapore
Patent: 202105196Y
Estimated Expiration: ⤷ Sign Up
South Africa
Patent: 1708760
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 180021775
Estimated Expiration: ⤷ Sign Up
Taiwan
Patent: 10556
Estimated Expiration: ⤷ Sign Up
Patent: 75187
Estimated Expiration: ⤷ Sign Up
Patent: 1718535
Estimated Expiration: ⤷ Sign Up
Patent: 2108568
Estimated Expiration: ⤷ Sign Up
Ukraine
Patent: 4728
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering COTELLIC around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Eurasian Patent Organization | 025871 | ИНГИБИТОРЫ MEK И СПОСОБЫ ИХ ПРИМЕНЕНИЯ (MEK INHIBITORS AND METHODS OF USING THE SAME) | ⤷ Sign Up |
| Australia | 2022201067 | Immediate-release tablets containing a drug and processes for forming the tablets | ⤷ Sign Up |
| South Africa | 201708760 | CRYSTALLINE FUMARATE SALT OF (S)-[3,4-DIFLUORO-2-(2-FLUORO-4-IODOPHENYLAMINO)PHENYL] [3-HYDROXY-3-(PIPERIDIN-2-YL) AZETIDIN-1-YL]-METHANONE | ⤷ Sign Up |
| South Korea | 20130058072 | AZETIDINES AS MEK INHIBITORS FOR THE TREATMENT OF PROLIFERATIVE DISEASES | ⤷ Sign Up |
| Japan | 7232284 | ⤷ Sign Up | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for COTELLIC
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1934174 | CR 2016 00021 | Denmark | ⤷ Sign Up | PRODUCT NAME: COBIMETINIB OG FARMACEUTISK ACCEPTABLE SALTE OG SOLVATER DERAF, SAERLIGT COBIMETINIB HEMIFUMARAT; REG. NO/DATE: EU/1/15/1048 20151124 |
| 1934174 | CA 2016 00021 | Denmark | ⤷ Sign Up | PRODUCT NAME: COBIMETINIB, EVENTUELT I EN HVILKEN SOM HELST FORM BESKYTTET AF GRUNDPATENTET, DER INDBEFATTER FARMACEUTISK ACCEPTABLE SALTE OG SOLVATER, SAERLIGT COBIMETINIB HEMIFUMARAT; REG. NO/DATE: EU/1/15/1048 20151124 |
| 1934174 | 364 5005-2016 | Slovakia | ⤷ Sign Up | PRODUCT NAME: KOBIMETINIB VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM PATENTOM; REGISTRATION NO/DATE: EU/1/15/1048/001 20151124 |
| 1934174 | PA2016016 | Lithuania | ⤷ Sign Up | PRODUCT NAME: KOBIMETINIBAS; REGISTRATION NO/DATE: EU/1/15/1048 20151120 |
| 1934174 | 650 | Finland | ⤷ Sign Up | |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


