Last updated: April 23, 2026
What product is CORTEF and how does it position in the market?
CORTEF is a brand of hydrocortisone (oral corticosteroid). It sits in the long-standing systemic glucocorticoids segment used for endocrine disorders and broad inflammatory/immune indications, with demand driven by chronic disease prevalence, guideline-based prescribing, and payer formularies rather than breakthrough differentiation.
Key market implication: CORTEF’s commercial trajectory is shaped less by clinical novelty and more by generic penetration, formulary access, pricing pressure, and channel-level competition. CORTEF’s lifecycle therefore tracks the typical economics of an established oral steroid brand.
How do prescribing and access dynamics shape demand?
Hydrocortisone is entrenched in multiple care settings, including:
- Adrenal insufficiency replacement
- Adrenal crisis management protocols (often involving other hydrocortisone formulations acutely, but maintenance and long-term prescribing sustain oral demand)
- Inflammatory and allergic conditions where clinicians still use systemic steroids when indicated
For a mature steroid brand, the demand drivers usually look like:
- Stable underlying prevalence (endocrinology and chronic steroid use)
- Switching behavior when payers adjust tier placement
- Seasonality is typically muted relative to acute antibiotics or vaccines
- Therapy persistence is common because replacement or chronic anti-inflammatory regimens are durable
Commercial consequence: demand often stays resilient, but brand economics compress as generic hydrocortisone and private-label options expand.
What is the competitive set and how does it affect pricing?
CORTEF faces a heavily genericized competitive landscape. In systemic corticosteroids, payers frequently prefer lower-cost generics and authorized generics when available, which drives:
- Net price erosion
- Volume migration to lowest-tier options
- Promotions and contracting to defend formulary position
Typical trade-off for mature brands: maintaining share requires payer-facing rebate and channel contracting, which lowers margin even when unit volume holds.
How do payer formularies and reimbursement influence sales?
In systemic steroids, formulary strategy tends to follow a consistent pattern:
- Broad inclusion (because therapies are standard-of-care)
- Preferential placement for generics or authorized generics
- Prior authorization steps in some plans depending on clinical history requirements and safety monitoring
That means CORTEF’s financial trajectory is driven by:
- Tier placement (preferred vs non-preferred)
- Formulary renewal outcomes
- Contracting terms with pharmacy benefit managers (PBMs)
- State Medicaid reimbursement dynamics and managed care switches
What does a plausible financial trajectory look like for CORTEF?
Without company-specific segment reporting and audited financials for CORTEF as a standalone line item, the observable lifecycle pattern for an established oral corticosteroid brand is:
-
Late-cycle brand stabilization
- Unit volume can remain stable when CORTEF maintains payer access.
- Margin declines due to rebates and competitive pricing.
-
Generic-driven net sales compression
- Net sales tend to contract faster than gross demand due to price competition.
- Brand share erodes incrementally via switching.
-
Channel consolidation and contracting pressure
- Remaining brand sales cluster in markets with residual brand preference.
- Rebates rise to offset tier creep and competitor substitution.
This pattern is consistent with mature, generic-exposed oral products, where growth becomes limited and financial performance is dominated by price and share rather than expansion.
Where does CORTEF sit on the patent and exclusivity timeline?
CORTEF’s hydrocortisone active ingredient is widely available as generic. Brand exclusivity typically does not protect long-term brand economics for an established steroid class. As a result, CORTEF’s market exposure is primarily post-exclusivity and generic competition rather than new regulatory exclusivity wins.
Implication: investment cases for CORTEF-style assets rely on defensive contracting, supply continuity, and lifecycle management, not breakthrough pipelines.
What supply and manufacturing dynamics matter for a corticosteroid brand?
Systemic steroid manufacturing is sensitive to:
- Supply continuity (distribution reliability matters in replacement therapies and chronic regimens)
- Batch consistency and regulatory compliance (especially for oral solids)
- Commercial inventory management to reduce shortages that trigger substitution and switching
A stable supply record can preserve share, but persistent generic pressure continues to limit upside.
Key commercial levers that determine CORTEF financial outcomes
For an established oral steroid brand, the biggest levers are:
- Formulary positioning: preferred tier maintenance versus tier drop
- Rebate and contracting: ability to buy access against generic competition
- Package and presentation differentiation: tablets, strengths, and availability can influence substitution
- Wholesaler and specialty pharmacy adoption: steady ordering patterns preserve sales continuity
How does demand risk present for CORTEF?
Even with generic exposure, demand risks are not purely financial. They include:
- Switching risk when payers redesign formularies
- Utilization risk if prescribers move to alternatives due to safety concerns or guideline shifts
- Regulatory or labeling shifts that can alter dosing practice
- Shortage risk that can disrupt continuity and accelerate permanent switching
Bottom-line market dynamics
The market dynamics for CORTEF are consistent with a mature, genericized oral therapy:
- Demand is stable at baseline due to replacement and chronic use patterns.
- Financial performance trends toward margin compression due to generic pricing.
- Share depends on payer access and contracting, not clinical superiority.
- Growth is limited unless the brand secures exceptional formulary outcomes or faces supply disruptions benefiting the brand.
Key Takeaways
- CORTEF is a mature hydrocortisone oral brand whose demand is primarily driven by chronic replacement and standard systemic steroid use rather than innovation.
- Its financial trajectory is constrained by generic penetration, pushing outcomes toward net price erosion and margin compression even when unit demand holds.
- The main determinants of performance are formulary tier placement, PBM contracting, and rebate economics.
- Market risk concentrates in switching dynamics and supply continuity rather than treatment discovery.
FAQs
1) Why does CORTEF’s market performance differ from specialty branded launches?
Because CORTEF is a mature systemic steroid brand competing against generic hydrocortisone, so financial results depend on payer access and pricing rather than new-to-market uptake.
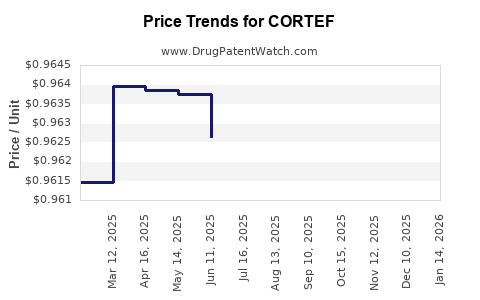
2) What typically happens to net price for an established corticosteroid brand?
Net price usually declines over time as plan sponsors favor lower-cost generics or authorized generics, forcing the brand to use rebates and contracting to preserve access.
3) Is demand for hydrocortisone brands usually volatile?
Demand is generally less volatile than acute therapies because many users take hydrocortisone for long-term replacement or ongoing indications, although switching and payer actions can still move share.
4) What factors most directly impact payer behavior for CORTEF?
Formulary tier rules, PBM contracting terms, and generic availability drive substitution and access for systemic steroids.
5) What commercial strategy most fits CORTEF’s stage of lifecycle?
Defensive lifecycle strategy: maintain supply reliability, defend formulary placement, and optimize rebates/contracts to limit share erosion.
References
[1] FDA. (n.d.). Drug Products@FDA: CORTEF (hydrocortisone). U.S. Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cder/daf/
[2] FDA. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cder/daf/
[3] NCBI Bookshelf. (n.d.). Hydrocortisone (drug monograph and clinical context). National Center for Biotechnology Information. https://www.ncbi.nlm.nih.gov/books/