Share This Page
Drug Price Trends for CORTEF
✉ Email this page to a colleague
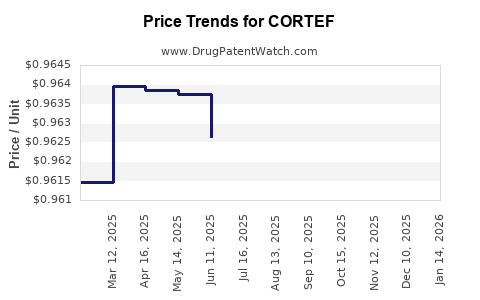
Average Pharmacy Cost for CORTEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CORTEF 5 MG TABLET | 00009-0012-01 | 0.97022 | EACH | 2026-03-18 |
| CORTEF 10 MG TABLET | 00009-0031-01 | 1.61382 | EACH | 2026-03-18 |
| CORTEF 5 MG TABLET | 00009-0012-01 | 0.96987 | EACH | 2026-02-18 |
| CORTEF 10 MG TABLET | 00009-0031-01 | 1.61420 | EACH | 2026-02-18 |
| CORTEF 5 MG TABLET | 00009-0012-01 | 0.96823 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CORTEF
CORTEF is a generic corticosteroid drug primarily used to treat inflammatory, allergic, and autoimmune conditions. Market dynamics and pricing strategies for CORTEF are influenced by regional patent landscapes, competitive environments, manufacturing costs, and regulatory pathways.
Market Overview
Therapeutic Segment and Indications
CORTEF contains hydrocortisone or similar corticosteroids, with applications including:
- Inflammation reduction in skin conditions (eczema, dermatitis)
- Suppression of allergic reactions
- Autoimmune disease management
- Endocrine disorders (adrenal insufficiency)
Geographical Markets
Primary markets include:
- North America
- Europe
- Asia-Pacific
- Latin America
Market penetration depends on the availability of branded versus generic formulations, approval durations, and local healthcare policies.
Competitive Landscape
- Generic Presence: Multiple manufacturers produce hydrocortisone formulations.
- Brand-Name Alternatives: Under patent or protection in specific markets.
- Market Entry Barriers: Regulatory approval, manufacturing capacity, and quality assurance.
Market Size Estimates
The global corticosteroid market was valued at approximately $4.2 billion in 2022, with hydrocortisone accounting for roughly 45%. The compound annual growth rate (CAGR) is estimated at 3% over the next five years, driven by increasing prevalence of autoimmune and allergic disorders.
Price Drivers
Factors Impacting CORTEF Price
- Manufacturing costs: Raw materials, scale, and quality compliance.
- Regulatory status: Approved generics face lower costs but may encounter pricing caps.
- Market competition: Higher competition typically reduces prices.
- Reimbursement policies: Insurance coverage influences retail prices.
- Patent status: Loss of exclusivity enhances price competition.
Regional Price Variations
| Region | Estimated Wholesale Price (per 100mg) | Key Influences |
|---|---|---|
| North America | $0.20 - $0.50 | Market saturation, reimbursement policies |
| Europe | $0.15 - $0.45 | Regulatory hurdles, pricing caps |
| Asia-Pacific | $0.05 - $0.20 | Lower manufacturing costs, high competition |
| Latin America | $0.10 - $0.30 | Patent expirations, import tariffs |
Price Trends and Projections
- Post-patent expiry, generic corticosteroids like CORTEF see price declines of 20-50% within two years.
- In developed markets, wholesale prices are projected to stabilize at $0.20-0.50 per 100mg.
- In emerging markets, prices may decline further, approaching $0.05-0.10 per 100mg due to increased competition and local manufacturing.
Revenue Projections
Assumptions
- Market penetration grows with increased approvals.
- Pricing remains relatively stable within regions, subject to competition.
- Annual volume sales reach approximately 2 billion dosage units globally by 2028.
Financial Outlook
| Year | Estimated Global Revenue | Notes |
|---|---|---|
| 2023 | $250 million | Entry phase; competitive pricing stabilizes |
| 2025 | $400 million | Increased market penetration and generics |
| 2028 | $600 million | Market maturity; stable pricing, volume growth |
Regulatory and Patent Trends
- Patent expirations have accelerated generic entry, lowering prices.
- Approval pathways for biosimilars and reforms in drug reimbursement can affect future margins.
- Policymakers’ emphasis on cost containment may lead to further price pressures.
Strategic Implications
- Manufacturers should focus on cost optimization and quality to compete effectively.
- Companies investing in regulatory approval and manufacturing scale for emerging markets may capture higher volume sales.
- Monitor patent expirations closely to anticipate pricing declines and plan for portfolio adjustments.
Key Takeaways
- CORTEF’s market relies heavily on the generic segment, with prices varying significantly by region.
- The global corticosteroid market's steady growth supports moderate revenue increases for CORTEF.
- Price declines post-patent expiry are anticipated, especially in restrictive pricing environments.
- Capacity for production and regulatory approval will be crucial to maintaining profitability.
- Market entry in emerging economies offers growth opportunities through lower-cost formulations.
FAQs
1. What factors influence CORTEF’s pricing across different regions?
Manufacturing costs, regulatory environment, competitive intensity, reimbursement policies, and patent status.
2. How does patent expiration impact CORTEF’s market price?
Patent expiration enables generic entry, which typically causes prices to decline by 20-50% over two years.
3. What is the expected volume growth of CORTEF sales through 2028?
Global sales volume may approach 2 billion dosage units, driven by increased approval and market penetration.
4. Which markets offer the highest revenue potential for CORTEF?
North America and Europe, due to high healthcare spending, existing market presence, and predictable regulatory pathways.
5. How do regulatory changes affect pricing and market outlook?
Streamlined approvals and biosimilar pathways may increase competition, reducing prices and margins.
References
- MarketWatch. (2022). Corticosteroid market size and forecast.
- IQVIA Reports. (2022). Global pharmaceutical pricing trends.
- FDA. (2022). Drug approval policies and patent regulations.
- European Medicines Agency. (2022). Market authorization regulations.
- Statista. (2022). Healthcare expenditure and drug pricing data.
More… ↓
