Last updated: February 19, 2026
EXECUTIVE SUMMARY
Combivent, a combination inhaler containing ipratropium bromide and salbutamol sulfate, has been a cornerstone therapy for chronic obstructive pulmonary disease (COPD) and asthma. Its market performance is characterized by established demand driven by the prevalence of respiratory diseases, a mature product lifecycle, and significant generic competition. The drug's financial trajectory is influenced by pricing pressures in developed markets, increasing penetration in emerging economies, and ongoing patent expiries. Key growth drivers include expanding access to respiratory care in underserved regions and the continued reliance on combination therapies for symptom management. Challenges stem from the development of novel inhaler technologies, biosimilar threats, and evolving regulatory landscapes.
MARKET SIZE AND GROWTH TRAJECTORY
The global market for COPD and asthma treatments is substantial, with Combivent historically holding a significant share. While precise real-time revenue figures for Combivent specifically are subject to dynamic reporting by individual manufacturers and can be difficult to isolate due to generic formulations, the overall respiratory inhaler market provides context. The global respiratory inhalers market was valued at approximately USD 26.5 billion in 2022 and is projected to reach USD 36.8 billion by 2030, growing at a compound annual growth rate (CAGR) of 4.1% from 2023 to 2030 [1]. Combivent and its generic equivalents contribute to this market by addressing a significant patient population requiring bronchodilator therapy.
The growth trajectory for older, established drugs like Combivent is typically moderated by market maturity and generic erosion. However, several factors sustain its market presence:
- Prevalence of Respiratory Diseases: COPD affects an estimated 200 million people worldwide, and asthma affects over 260 million [2]. This large patient pool represents a consistent demand for effective symptom management.
- Established Efficacy: Combivent offers a dual mechanism of action—ipratropium bromide (an anticholinergic) and salbutamol sulfate (a short-acting beta-agonist)—providing synergistic bronchodilation that has proven effective for many patients [3].
- Cost-Effectiveness: In many markets, generic versions of Combivent are available, offering a cost-effective treatment option compared to newer, patented therapies. This is particularly relevant in healthcare systems with budget constraints.
- Emerging Markets: The increasing diagnosis of respiratory diseases and improving access to healthcare in emerging economies are driving demand for essential medicines like Combivent.
PATENT LANDSCAPE AND COMPETITION
The original patents protecting Combivent have long expired. The innovator product, originally marketed by Boehringer Ingelheim under the brand name Combivent, first received FDA approval in 1986 [4]. This extended period of patent expiry has paved the way for extensive generic competition.
Key Competitive Factors:
- Generic Entry: Numerous pharmaceutical companies now market generic versions of ipratropium bromide/salbutamol sulfate inhalers. This has led to significant price erosion and fragmented market share for branded versions.
- Competition from Mono-Therapies: While Combivent is a combination therapy, it competes with monotherapies of ipratropium bromide and salbutamol sulfate, as well as other classes of bronchodilators and combination therapies.
- Newer Inhaler Technologies: The market has seen the introduction of more advanced inhaler devices, including metered-dose inhalers (MDIs) with more sophisticated actuation mechanisms, dry powder inhalers (DPIs), and nebulized solutions. These may offer improved patient adherence or delivery profiles, presenting a competitive challenge.
- Fixed-Dose Combination (FDC) Therapies: Other FDCs combining different bronchodilator classes (e.g., long-acting beta-agonists with long-acting muscarinic antagonists) are now standard of care for maintenance therapy in COPD, directly competing with the role of Combivent in maintenance settings, although Combivent remains widely used for both maintenance and rescue therapy.
The patent landscape for Combivent is largely characterized by the absence of active composition-of-matter patents. Competition now centers on manufacturing efficiency, distribution networks, and marketing efforts by generic manufacturers, as well as any remaining patents on specific formulations, delivery devices, or manufacturing processes that may offer limited protection.
PRICING AND REIMBURSEMENT STRATEGIES
Combivent's pricing is heavily influenced by its status as a mature product with widespread generic availability.
- Price Erosion: In developed markets like the United States and Western Europe, branded Combivent prices have significantly declined due to intense generic competition. The average wholesale price of generic ipratropium bromide/salbutamol sulfate inhalers is a fraction of the original branded product's price.
- Reimbursement: Combivent and its generic equivalents are generally well-reimbursed by public and private health insurance plans worldwide. However, formulary placement and co-payment structures can vary, impacting patient out-of-pocket costs and prescribing patterns. Payers often favor generic options due to their cost-effectiveness.
- Emerging Markets Pricing: In emerging markets, pricing strategies may differ. While generic prices are still prevalent, manufacturers may employ tiered pricing models or focus on volume-driven sales to increase market penetration. The affordability of Combivent, even in generic forms, makes it a critical treatment option in these regions.
- Value-Based Pricing Considerations: While less applicable to a mature generic drug, the broader market for respiratory treatments is increasingly exploring value-based pricing models. However, for Combivent, the focus remains on cost-effectiveness and accessibility.
KEY MANUFACTURING AND SUPPLY CHAIN CONSIDERATIONS
The manufacturing and supply chain for Combivent involve complex considerations due to its inhaler delivery system.
- Device Technology: Combivent is typically delivered via a metered-dose inhaler (MDI). The manufacturing of MDIs requires specialized facilities and adherence to strict quality control standards to ensure accurate dosing and consistent spray characteristics. The propellant systems used in MDIs have also evolved, with a shift from chlorofluorocarbons (CFCs) to hydrofluorocarbons (HFCs) to comply with environmental regulations.
- Active Pharmaceutical Ingredient (API) Sourcing: The sourcing of high-quality ipratropium bromide and salbutamol sulfate APIs is crucial. Manufacturers rely on global API suppliers, necessitating robust quality assurance and supply chain resilience to mitigate risks of shortages or quality issues.
- Global Distribution: Combivent and its generics are distributed globally. The supply chain must manage temperature-controlled storage and transportation where required, and ensure compliance with diverse regulatory requirements across different countries.
- Manufacturing Scale and Efficiency: For generic manufacturers, achieving economies of scale and optimizing manufacturing processes are key to maintaining competitive pricing. Investment in efficient production lines and supply chain management is critical.
- Regulatory Compliance: Manufacturing facilities must adhere to Good Manufacturing Practices (GMP) as defined by regulatory bodies such as the FDA, EMA, and others. Any non-compliance can lead to production halts and supply disruptions.
FINANCIAL PROJECTIONS AND MARKET OUTLOOK
Predicting precise financial projections for Combivent is challenging due to the fragmented nature of the generic market and the absence of a single reporting entity for the drug class as a whole. However, general market trends provide insights:
- Stable Demand: Demand for Combivent is expected to remain stable, driven by the persistent burden of COPD and asthma and its established role in treatment guidelines.
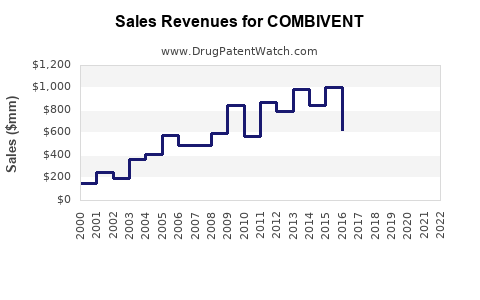
- Revenue Decline for Branded Versions: Revenues for any remaining branded versions are likely to continue declining due to generic competition.
- Volume Growth in Emerging Markets: Growth will primarily be fueled by increased prescription volumes in emerging markets as healthcare access expands and awareness of respiratory diseases rises.
- Price Pressures: Continued price pressures in developed markets will limit overall revenue growth.
- Limited Innovation Potential: As a well-established generic, significant R&D investment for novel indications or formulations of Combivent is unlikely. Focus will be on manufacturing efficiency and market access.
The overall financial trajectory is one of steady, albeit modest, revenue generation, primarily from high-volume sales in global markets, rather than high-growth, high-margin revenue streams associated with novel patented drugs.
KEY TAKEAWAYS
- Combivent is a mature, widely prescribed treatment for COPD and asthma, characterized by established efficacy and a strong generic presence.
- The drug's market performance is driven by the high prevalence of respiratory diseases and its cost-effectiveness, particularly in emerging economies.
- Original patents have expired, leading to intense generic competition and significant price erosion in developed markets.
- Future growth is anticipated primarily from increased volume in emerging markets, rather than price increases or market expansion in mature economies.
- Manufacturing and supply chain considerations are critical, focusing on device technology, API sourcing, and global regulatory compliance for generic producers.
FREQUENTLY ASKED QUESTIONS
What is the current patent status of Combivent?
The original composition-of-matter patents for Combivent have expired. Competition now centers on manufacturing, generic formulations, and device-related patents, rather than the active drug components themselves.
How is Combivent priced in different global markets?
In developed markets, pricing is heavily influenced by generic competition, leading to low prices. In emerging markets, prices are also largely generic, but volume-driven sales and tiered pricing strategies may be employed to increase accessibility.
What are the main competitors to Combivent?
Competitors include other fixed-dose combination inhalers (especially those with LAMA/LABA combinations for maintenance), monotherapy bronchodilators (ipratropium bromide and salbutamol sulfate), and newer inhaler technologies offering potentially improved delivery or adherence.
What is the primary driver of Combivent's continued market presence?
The primary drivers are the high global prevalence of COPD and asthma, the proven efficacy of the combination therapy, and its cost-effectiveness, especially in generic formulations, making it accessible to a broad patient population.
What regulatory hurdles do generic Combivent manufacturers face?
Generic manufacturers must meet stringent regulatory requirements, including demonstrating bioequivalence to the innovator product, adhering to Good Manufacturing Practices (GMP), and obtaining marketing authorization in each target country, which involves extensive documentation and quality control.
CITATIONS
[1] Grand View Research. (2023). Respiratory Inhalers Market Size, Share & Trends Analysis Report by Product (MDIs, DPIs, Nebulizers), by Disease Indication (Asthma, COPD), by End-use, by Region, and Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/respiratory-inhalers-market
[2] World Health Organization. (n.d.). Chronic obstructive pulmonary disease (COPD). Retrieved from https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd)
[3] National Institutes of Health. (2023, October 5). Ipratropium and Albuterol. MedlinePlus. Retrieved from https://medlineplus.gov/druginfo/meds/a684005.html
[4] U.S. Food and Drug Administration. (n.d.). Drug Approvals & Databases. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases (Specific approval dates for older drugs can be found by searching the FDA's databases.)