Last updated: February 19, 2026
Clobex (clobetasol propionate) is a high-potency topical corticosteroid used for the short-term treatment of moderate to severe plaque psoriasis and other corticosteroid-responsive dermatoses. Its market performance is dictated by patent exclusivity, generic competition, regulatory approvals, and evolving treatment guidelines for dermatological conditions.
What is Clobex's Current Market Position?
Clobex, under its brand name, primarily targets severe dermatological inflammatory conditions where a potent topical corticosteroid is indicated. The global market for topical corticosteroids is mature, characterized by established generic presence and price sensitivity. However, high-potency agents like clobetasol propionate maintain a distinct market segment due to their efficacy in more severe cases.
The active pharmaceutical ingredient, clobetasol propionate, is a well-established compound with a long history of use. Its efficacy in reducing inflammation, itching, and redness associated with conditions like psoriasis is well-documented. The primary market drivers for Clobex include the prevalence of dermatological diseases, particularly psoriasis, and the willingness of healthcare providers to prescribe high-potency topical steroids for patients unresponsive to lower-potency options or other therapies.
Key Market Characteristics:
- Therapeutic Class: High-potency topical corticosteroid.
- Primary Indications: Plaque psoriasis, eczema, dermatitis.
- Market Maturity: Mature market with established generic alternatives.
- Competitive Landscape: Dominated by branded Clobex and a significant number of generic clobetasol propionate formulations.
What is the Patent Status and Exclusivity Landscape for Clobex?
The original patent protection for Clobex has long expired. This has led to widespread generic competition, significantly impacting the pricing and market share of the branded product. The primary exclusivity Clobex now relies on is through its formulation and specific indications, rather than core compound patents.
- Original Compound Patent Expiry: Early 2000s.
- Formulation Patents: Specific formulations of clobetasol propionate, such as foams, shampoos, or enhanced topical solutions, may have had their own patent protection periods. However, these have largely expired or are nearing expiration in major markets.
- Regulatory Exclusivity: For new indications or specific formulations, a period of regulatory exclusivity may have been granted by regulatory bodies like the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA). This exclusivity prevents generics from entering the market for a defined period. For instance, the FDA may grant 5-year New Chemical Entity (NCE) exclusivity or 3-year exclusivity for new indications or new formulations.
The lack of strong, extended patent protection for the core molecule means that market share is primarily defended through brand loyalty, physician preference, and the development of proprietary delivery systems or combination products.
How Have Sales and Revenue Performed for Clobex?
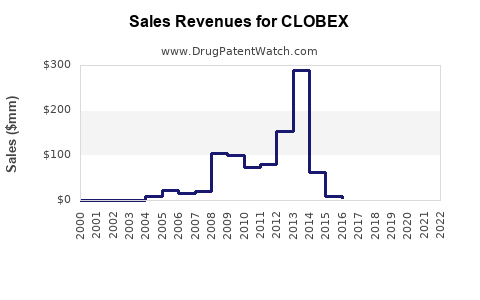
Detailed, real-time revenue figures for specific branded drugs like Clobex are often proprietary and not publicly disclosed by manufacturers, especially after patent expiry and the emergence of generic competition. However, the trajectory of branded Clobex sales is understood to follow a pattern typical for drugs in its lifecycle stage.
Following its launch and during its period of patent exclusivity, Clobex would have experienced peak sales. As generic versions of clobetasol propionate entered the market, branded Clobex sales would have significantly declined due to price erosion. Manufacturers often focus on maintaining market share through marketing efforts targeting healthcare providers and patients who prefer the branded product, or by developing new formulations.
General Sales Trajectory Post-Patent Expiry:
- Peak Sales Period: During patent exclusivity, characterized by high pricing and market dominance.
- Declining Sales: Post-patent expiry, due to generic entry, price pressure, and loss of market exclusivity.
- Stabilization/Niche Market: Sales may stabilize as the branded product caters to a specific segment of the market, often through value-added formulations or established physician trust.
Manufacturers may report Clobex as part of a larger therapeutic category portfolio, making precise Clobex-specific financial analysis challenging from public filings. For instance, if Galderma, a major marketer of Clobex, reports revenue for its dermatology portfolio, Clobex’s exact contribution is not isolated.
What is the Competitive Landscape for Clobex?
The competitive landscape for Clobex is bifurcated: the branded Clobex products and the multitude of generic clobetasol propionate formulations.
Branded Clobex Offerings:
- Clobex® Lotion (0.05%): A common formulation.
- Clobex® Spray (0.05%): Offers an alternative delivery system.
- Clobex® Shampoo (0.05%): For scalp psoriasis.
- Clobex® Cream (0.05%): Standard cream formulation.
- Clobex® Ointment (0.05%): For thicker, scaly lesions.
Generic Clobetasol Propionate:
Numerous pharmaceutical companies market generic versions of clobetasol propionate in various strengths and formulations (creams, ointments, lotions, shampoos). These generics compete primarily on price, offering a significantly lower cost to patients and healthcare systems.
Key Competitors (Beyond Generics):
While generics are the direct price competitors, other high-potency topical corticosteroids compete for patient treatment:
- Dovonex® (calcipotriene) / Dovobet® (calcipotriene and betamethasone dipropionate): Combination products offering a different mechanism of action.
- Temovate® (clobetasol propionate): Another branded clobetasol propionate product, directly competing with Clobex.
- Ultravate® (halobetasol propionate): Another high-potency topical corticosteroid.
The choice between Clobex and its generics or alternative treatments often depends on physician preference, patient insurance coverage, cost, and the specific characteristics of the dermatological condition.
What is the Regulatory Environment Impacting Clobex?
The regulatory environment is a critical factor for both branded Clobex and its generic counterparts. Regulatory bodies like the FDA and EMA oversee drug approvals, labeling, manufacturing quality, and post-market surveillance.
Key Regulatory Considerations:
- U.S. FDA: Clobex formulations have received FDA approval for specific indications. Generic clobetasol propionate products must demonstrate bioequivalence to the reference listed drug (RLD), which in this case would be a Clobex product. The FDA's Orange Book lists approved drug products with their approval dates, patent information, and exclusivity data.
- EMA (Europe): Similar approval pathways exist, with national authorities and the EMA evaluating safety, efficacy, and quality.
- Labeling and Indication Expansion: Manufacturers may seek FDA/EMA approval for new formulations or expanded indications for Clobex, potentially granting additional periods of market exclusivity. However, for a well-established molecule like clobetasol propionate, the potential for new, patentable indications is limited.
- Manufacturing Standards: Adherence to Good Manufacturing Practices (GMP) is essential for all manufacturers, branded and generic, to ensure product quality and safety. Regulatory inspections and potential recalls can significantly impact market access and reputation.
- Pharmacoeconomic Guidelines: Increasingly, healthcare systems and payers implement pharmacoeconomic guidelines that favor cost-effective treatments. This can lead to preferred formulary placement for generics over branded drugs, impacting Clobex's market access and sales volume.
The regulatory landscape also includes post-market pharmacovigilance, where any new safety concerns could lead to updated labeling, restrictions, or even market withdrawal, affecting all versions of the drug.
What is the Pricing and Reimbursement Scenario for Clobex?
Pricing and reimbursement for Clobex are heavily influenced by its patent status and the presence of generic alternatives.
- Branded Clobex Pricing: When under patent exclusivity, branded Clobex commanded premium pricing. Post-expiry, the price of branded Clobex has been subject to competition and may have been strategically managed to retain a portion of its market share, often at a price point lower than its peak but still higher than generics.
- Generic Clobetasol Propionate Pricing: Generic versions are priced significantly lower, typically 20-80% less than the branded product, depending on the market and the number of generic competitors. This price differential is the primary driver for generic adoption.
- Reimbursement Policies:
- Insurers and PBMs: Pharmacy benefit managers (PBMs) and insurance companies often place generics on lower tiers of their formularies, requiring higher co-pays for branded medications. This incentivizes patients and physicians to choose generics.
- Government Programs: Programs like Medicare and Medicaid in the U.S. also have formulary policies that typically favor lower-cost generic options for drugs like clobetasol propionate.
- Prior Authorization: In some cases, payers may require prior authorization for high-potency topical corticosteroids like Clobex, especially if lower-potency options have not been tried, adding a layer of administrative complexity for the prescriber.
The pricing and reimbursement environment for Clobex directly impacts its profitability. While the branded product may retain higher margins per unit, its overall revenue contribution is likely diminished compared to its peak. Generic manufacturers compete on volume and cost efficiency.
What are the Future Market Projections for Clobex?
The future market for Clobex is expected to remain stable within its niche, with continued pressure from generics and evolving dermatological treatment paradigms.
- Sustained Generic Dominance: Generic clobetasol propionate will continue to dominate the market in terms of volume and overall market spend due to cost-effectiveness.
- Niche for Branded Clobex: Branded Clobex will likely maintain a smaller, dedicated market share, catering to patients and physicians who prefer specific formulations or brand reliability. This might include specialized delivery systems like sprays or foams, if they offer a demonstrable advantage and are adequately differentiated.
- Impact of Newer Therapies: The emergence of novel treatments for psoriasis, such as biologic agents and targeted small molecules, could indirectly impact the market for topical corticosteroids. While topicals remain first-line for many patients, the availability of highly effective systemic or injectable therapies for moderate-to-severe disease might reduce the overall demand for potent topical steroids in certain patient populations.
- Formulation Innovation: Future growth for branded Clobex, if any, would likely stem from new, patent-protected formulations or combination products that offer enhanced efficacy, safety, or convenience compared to existing options. However, innovation in topical corticosteroids has slowed considerably.
- Geographic Variations: Market dynamics will vary by region, influenced by local healthcare policies, generic penetration rates, and physician prescribing habits. Developed markets with strong generic infrastructure will see higher generic uptake, while emerging markets may still offer some opportunities for branded products.
Key Future Trends:
- Continued Price Competition: Generics will maintain price pressure.
- Focus on Value-Added Formulations: Branded manufacturers may emphasize specific delivery systems.
- Competition from Systemic Therapies: Biologics and other advanced treatments will influence the use of topicals for severe disease.
Key Takeaways
- Clobex, a high-potency topical corticosteroid, operates in a mature market characterized by significant generic competition following the expiry of its compound patents.
- The branded product's market position relies on its established efficacy, specific formulations, and physician/patient preference rather than patent exclusivity.
- Generic clobetasol propionate formulations offer substantial cost advantages, leading to widespread adoption and price erosion for the branded product.
- Regulatory bodies like the FDA and EMA govern the market through approvals, labeling, and quality standards, impacting both branded and generic versions.
- Pricing and reimbursement policies strongly favor generic alternatives, making it challenging for branded Clobex to regain significant market share.
- The future market trajectory suggests continued generic dominance, with branded Clobex likely maintaining a stable but smaller niche, influenced by evolving dermatological treatments and potential formulation innovations.
Frequently Asked Questions
-
What is the primary differentiator between branded Clobex and its generic counterparts?
The primary differentiators are brand recognition, established trust, and potentially proprietary delivery systems or formulations. Generic clobetasol propionate competes primarily on price.
-
Has Clobex been approved for any new indications recently?
Information on recent, significant new indication approvals for branded Clobex is not readily available in the public domain, which is typical for older, off-patent molecules. Most current market activity involves generic competition.
-
What are the most significant challenges facing the Clobex brand in the current market?
The most significant challenge is intense price competition from generic clobetasol propionate. Additionally, the availability of newer, more targeted systemic therapies for severe dermatological conditions can reduce the overall demand for potent topical steroids.
-
How does the patent expiry of Clobex impact its overall market revenue?
Patent expiry leads to the entry of generic competitors, which significantly drives down pricing and erodes the market share and revenue of the branded product. The revenue trajectory typically shifts from peak growth to decline and then stabilization at a lower level.
-
Are there any specific Clobex formulations that have retained stronger market positioning due to their unique properties?
Certain formulations, such as Clobex Spray or Shampoo, might offer convenience or specific application advantages that maintain some preference among prescribers and patients, potentially allowing them to retain a slightly stronger market position compared to standard cream or ointment generics, though this is often subject to pricing dynamics.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/informationondrugs/ucm079347.htm
[2] Galderma. (n.d.). Product Information. Retrieved from https://www.galderma.com/ (Specific product pages are not cited as they change and are proprietary to the company).
[3] European Medicines Agency. (n.d.). Human medicines. Retrieved from https://www.ema.europa.eu/en/human-medicines
[4] National Psoriasis Foundation. (n.d.). Topical Treatments. Retrieved from https://www.psoriasis.org/ (General information regarding psoriasis treatment modalities).