Last updated: February 19, 2026
Ciprodex (ciprofloxacin/dexamethasone otic suspension) is a combination antibiotic and corticosteroid product used for treating bacterial infections of the ear. Its market trajectory is largely defined by patent expiry dates, generic competition, and the subsequent pricing pressures and market share shifts. Analysis of its patent portfolio and regulatory filings provides a roadmap for understanding its commercial lifecycle.
What are the Key Patents Covering Ciprodex?
The primary patent landscape for Ciprodex is anchored by patents held by Bayer Pharmaceuticals and its affiliates. The original U.S. patent for the combination product and its specific formulation is crucial.
- U.S. Patent No. 5,688,805: This patent, titled "Otic suspension composition," covers the specific formulation of ciprofloxacin and dexamethasone in an otic suspension. It was filed on March 11, 1996, and issued on November 17, 1998. The listed expiration date, considering statutory extensions, places its final expiration significantly into the future, but this is for the original composition.
- Orange Book Listing: The U.S. Food and Drug Administration (FDA) Orange Book lists patents and exclusivity periods for approved drug products. Ciprodex has had multiple patents listed, some of which relate to its formulation, method of use, and manufacturing. For instance, patents related to polymorphs or specific manufacturing processes can extend market exclusivity beyond the core composition patent.
- Pediatric Exclusivity: In some cases, pediatric exclusivity could have been granted, extending the market exclusivity for a period of six months. This is typically awarded if new clinical investigations are conducted in accordance with an approved pediatric study plan.
The effective period of market exclusivity is a composite of the base patent term, any extensions (such as Patent Term Adjustment or Pediatric Exclusivity), and other regulatory exclusivities like New Chemical Entity (NCE) or New Drug Application (NDA) exclusivity.
When Did Generic Competition Emerge for Ciprodex?
The emergence of generic competition for Ciprodex was directly tied to the expiration of its key patents and the successful approval of Abbreviated New Drug Applications (ANDAs) by generic manufacturers.
- Initial Generic Approvals: Generic versions of Ciprodex began to appear in the market following the expiration of the primary patent protection that prevented ANDA approvals. This typically occurs when the patent is no longer considered valid by the FDA for blocking generic entry, or when a Paragraph IV certification on the patent has been litigated and failed by the brand.
- Key Generic Entrants: Multiple generic pharmaceutical companies have entered the market with their own versions of ciprofloxacin/dexamethasone otic suspension. These include manufacturers such as Teva Pharmaceuticals, Sandoz (a Novartis company), and Aurobindo Pharma. The FDA's approval of ANDAs from these companies signifies that their products are bioequivalent to the branded Ciprodex and can be substituted.
- Impact on Market Share: The introduction of multiple generic competitors invariably leads to a significant erosion of market share for the branded product, Ciprodex. This is a standard dynamic in the pharmaceutical industry, where generic products typically offer a lower price point.
The timeline for generic entry is a critical factor in forecasting revenue streams for both the innovator and the generic manufacturers.
What is the Current Market Status of Ciprodex and its Generics?
The current market for Ciprodex is characterized by a mature, highly competitive landscape dominated by generic offerings.
- Branded vs. Generic Pricing: Branded Ciprodex, manufactured by Bayer, commands a premium price. However, the availability of multiple generic equivalents has driven down prices significantly. Generic manufacturers compete on cost, making their products more accessible to healthcare providers and payers.
- Market Share Distribution: While exact real-time market share data fluctuates, the overwhelming majority of prescriptions for ciprofloxacin/dexamethasone otic suspension are filled by generic products. Branded Ciprodex retains a smaller, niche share, often influenced by physician preference or specific payer contracts.
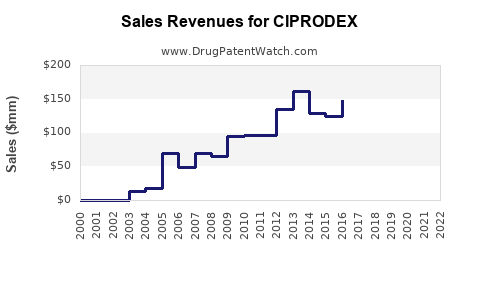
- Sales Data Trends: Historical sales data for branded Ciprodex would show a sharp decline following the widespread availability of generics. Conversely, the aggregate sales of all generic ciprofloxacin/dexamethasone otic suspensions would have increased substantially. Specific revenue figures for branded Ciprodex have been integrated into broader Bayer product segment reporting, making precise standalone numbers less frequently disclosed post-generic entry. For example, in Bayer’s fiscal year 2022 reporting, specialty products saw a decline primarily due to "loss of patent protection for certain products." While Ciprodex is not explicitly named in every report, it exemplifies this trend.
- Geographic Variations: Market penetration and the speed of generic adoption can vary by country due to differences in patent law, regulatory approval processes, and healthcare reimbursement systems. However, in major markets like the United States, generic entry is typically swift once patents expire.
What are the Financial Implications of Patent Expiry for Ciprodex?
The financial implications of patent expiry for Ciprodex are substantial and follow a predictable pattern in the pharmaceutical industry.
- Loss of Exclusivity Revenue: The primary financial impact is the dramatic decline in revenue for the innovator company (Bayer) due to the loss of market exclusivity. During the period of exclusivity, the innovator company benefits from a de facto monopoly, allowing for higher pricing and greater profit margins.
- Generic Manufacturer Profitability: For generic manufacturers, patent expiry represents a significant market opportunity. Companies that successfully develop and gain approval for their ANDAs can capture a substantial portion of the market at a lower price point, generating considerable revenue through high-volume sales.
- Pricing Erosion: The introduction of generic competition leads to aggressive price reductions. The price of the drug can drop by 70-90% or more from the branded price once multiple generics are available. This dramatically alters the revenue generated per prescription.
- R&D Investment Recovery: The period of market exclusivity is critical for pharmaceutical companies to recoup their substantial investments in research and development, clinical trials, and regulatory approval processes. Patent expiry marks the end of this recovery phase for the innovator.
- Market Size and Growth (Post-Generic): The overall market size for ciprofloxacin/dexamethasone otic suspension, in terms of total dollar value, may initially contract after generic entry due to price drops. However, increased accessibility and lower costs can lead to an increase in the number of prescriptions filled, potentially stabilizing or even growing the total market volume in units.
The financial trajectory shifts from high-margin, lower-volume sales for the innovator to lower-margin, higher-volume sales for generic producers.
What are the Regulatory Pathways for Generic Entry of Ciprodex?
The regulatory pathway for generic entry into the U.S. market for Ciprodex is governed by the Hatch-Waxman Act and managed by the FDA.
- Abbreviated New Drug Application (ANDA): Generic manufacturers must file an ANDA with the FDA. This application demonstrates that the generic product is bioequivalent to the branded reference listed drug (RLD), which is Ciprodex. Bioequivalence is typically established through pharmacokinetic studies.
- Patent Certification (Paragraph IV): A critical step in the ANDA process is patent certification. Generic applicants must certify that the patents covering the RLD are either invalid, unenforceable, or will expire by a certain date. Filing a Paragraph IV certification, which asserts that a patent is invalid or will not be infringed, can trigger a 30-month stay of FDA approval if the patent holder sues for infringement. This stay can be overcome if the patent is found invalid or not infringed in court.
- Approval Criteria: For an ANDA to be approved, the FDA must find that the generic product meets all the same quality, safety, and efficacy standards as the RLD. The active ingredients must be the same, and the inactive ingredients must be identified and deemed safe.
- Exclusivity for First Generic Entrant: Under the Hatch-Waxman Act, the first generic manufacturer to successfully challenge a listed patent (through a Paragraph IV certification and subsequent litigation) or the first to receive ANDA approval after patent expiration can be granted 180 days of marketing exclusivity. This exclusivity prevents other generics from entering the market for that specific drug until the 180-day period expires, allowing the first entrant a period of limited competition.
Understanding these regulatory steps is crucial for assessing the timing and competitive landscape of generic market entry.
What is the Competitive Landscape Beyond Branded and Generic Pharmaceuticals?
While the primary competition for Ciprodex revolves around its branded version and generic equivalents, other factors and potential competitive pressures exist.
- Alternative Treatments: For otitis media with effusion, which Ciprodex is often prescribed for, watchful waiting or the use of other antibiotics or anti-inflammatory agents could be considered. For other ear infections, alternative classes of antibiotics or different corticosteroid formulations might be used.
- Combination Product Development: The success of the ciprofloxacin/dexamethasone combination could spur the development of new combination otic products with improved efficacy, broader spectrum of activity, or better patient compliance profiles. However, developing novel combinations faces significant R&D hurdles and regulatory scrutiny.
- New Delivery Systems: Innovations in drug delivery systems for otic suspensions could offer a competitive edge. This might include formulations with longer-acting effects, reduced administration frequency, or improved penetration into the middle ear.
- Biosimil Landscape (Not Applicable to Small Molecules): It is important to note that biosimil competition is relevant for biologic drugs, not for small-molecule pharmaceuticals like Ciprodex.
- Market Consolidation and Payer Influence: The pharmaceutical market is subject to consolidation among manufacturers and increasing influence from payers (insurance companies, pharmacy benefit managers). These entities can influence prescribing patterns by favoring certain generics or negotiating exclusive supply agreements.
The long-term competitive environment will depend on the emergence of superior therapeutic alternatives and ongoing innovation in otic drug delivery and formulation.
Key Takeaways
- Ciprodex patent protection, particularly U.S. Patent No. 5,688,805, has largely expired, paving the way for generic competition.
- Generic versions of ciprofloxacin/dexamethasone otic suspension are available from multiple manufacturers, including Teva, Sandoz, and Aurobindo.
- The introduction of generics has led to significant price erosion and a substantial decline in market share for branded Ciprodex.
- Generic manufacturers gain market entry through the FDA's Abbreviated New Drug Application (ANDA) process, requiring demonstration of bioequivalence to the reference listed drug.
- The financial trajectory for Ciprodex shifted from innovator revenue generation during exclusivity to high-volume, lower-margin sales by generic companies post-exclusivity.
Frequently Asked Questions
-
When was the primary U.S. patent for Ciprodex composition granted?
U.S. Patent No. 5,688,805, covering the otic suspension composition, was granted on November 17, 1998.
-
Which companies are major generic manufacturers of ciprofloxacin/dexamethasone otic suspension?
Key generic manufacturers include Teva Pharmaceuticals, Sandoz, and Aurobindo Pharma.
-
What is the typical price reduction observed for generic drugs compared to their branded counterparts?
Generic drugs can experience price reductions of 70-90% or more compared to the branded drug price after market entry.
-
What is the regulatory pathway for a generic drug manufacturer to enter the U.S. market?
Manufacturers must file an Abbreviated New Drug Application (ANDA) with the FDA, demonstrating bioequivalence to the reference listed drug and addressing relevant patent certifications.
-
Can pediatric exclusivity extend market protection for Ciprodex?
Yes, pediatric exclusivity can grant an additional six months of market exclusivity if new clinical investigations are conducted in response to an approved pediatric study plan.
Citations
[1] United States Patent and Trademark Office. (1998). U.S. Patent No. 5,688,805. Retrieved from USPTO website.
[2] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (The Orange Book). Retrieved from FDA website.
[3] Bayer AG. (2023). Annual Report 2022. Retrieved from Bayer AG investor relations.
[4] U.S. Food and Drug Administration. (n.d.). Generic Drugs Program. Retrieved from FDA website.
[5] Food and Drug Administration. (1984). Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman Act). U.S. Government Publishing Office.