CIBINQO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Cibinqo, and when can generic versions of Cibinqo launch?
Cibinqo is a drug marketed by Pfizer and is included in one NDA. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has sixty-five patent family members in forty-four countries.
The generic ingredient in CIBINQO is abrocitinib. Two suppliers are listed for this compound. Additional details are available on the abrocitinib profile page.
DrugPatentWatch® Generic Entry Outlook for Cibinqo
Cibinqo was eligible for patent challenges on January 14, 2026.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be January 14, 2036. This may change due to patent challenges or generic licensing.
There is one Paragraph IV patent challenge for this drug. This may lead to patent invalidation or a license for generic production.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for CIBINQO?
- What are the global sales for CIBINQO?
- What is Average Wholesale Price for CIBINQO?
Summary for CIBINQO
| International Patents: | 65 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 39 |
| Clinical Trials: | 2 |
| Patent Applications: | 387 |
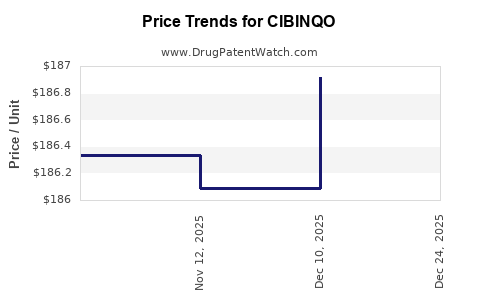
| Drug Prices: | Drug price information for CIBINQO |
| What excipients (inactive ingredients) are in CIBINQO? | CIBINQO excipients list |
| DailyMed Link: | CIBINQO at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for CIBINQO
Generic Entry Date for CIBINQO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for CIBINQO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) | Phase 2 |
| Pfizer | Phase 2 |
| Innovaderm Research Inc. | Phase 4 |
Pharmacology for CIBINQO
| Drug Class | Janus Kinase Inhibitor |
| Mechanism of Action | Janus Kinase Inhibitors P-Glycoprotein Inhibitors |
Paragraph IV (Patent) Challenges for CIBINQO
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| CIBINQO | Tablets | abrocitinib | 50 mg, 100 mg and 200 mg | 213871 | 3 | 2026-01-14 |
US Patents and Regulatory Information for CIBINQO
CIBINQO is protected by three US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of CIBINQO is ⤷ Start Trial.
This potential generic entry date is based on patent 9,035,074.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pfizer | CIBINQO | abrocitinib | TABLET;ORAL | 213871-001 | Jan 14, 2022 | RX | Yes | No | 9,549,929 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Pfizer | CIBINQO | abrocitinib | TABLET;ORAL | 213871-002 | Jan 14, 2022 | RX | Yes | No | 9,035,074 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Pfizer | CIBINQO | abrocitinib | TABLET;ORAL | 213871-001 | Jan 14, 2022 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Pfizer | CIBINQO | abrocitinib | TABLET;ORAL | 213871-003 | Jan 14, 2022 | RX | Yes | Yes | 9,545,405 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Pfizer | CIBINQO | abrocitinib | TABLET;ORAL | 213871-002 | Jan 14, 2022 | RX | Yes | No | 9,545,405 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Pfizer | CIBINQO | abrocitinib | TABLET;ORAL | 213871-003 | Jan 14, 2022 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Pfizer | CIBINQO | abrocitinib | TABLET;ORAL | 213871-001 | Jan 14, 2022 | RX | Yes | No | 9,545,405 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for CIBINQO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Pfizer Europe MA EEIG | Cibinqo | abrocitinib | EMEA/H/C/005452Cibinqo is indicated for the treatment of moderate-to-severe atopic dermatitis in adults who are candidates for systemic therapy. | Authorised | no | no | no | 2021-12-09 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for CIBINQO
When does loss-of-exclusivity occur for CIBINQO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 4857
Patent: DERIVADOS DE PIRROLO[2,3-D]PIRIMIDINA
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 14220357
Patent: Pyrrolo [2, 3 -d]pyrimidine derivatives as inhibitors of Janus- related Kinases (JAK)
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2015019634
Patent: derivados de pirrolo[2,3-d]pirimidina como inibidores de janus kinase (jak)
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 00703
Patent: DERIVES DE PYRROLO[2,3-D]PYRIMIDINE EN TANT QU'INHIBITEURS DE JANUS KINASES (JAK) (PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 15002303
Patent: Derivados de pirrolo[2,3-d]pirimidina
Estimated Expiration: ⤷ Start Trial
China
Patent: 5008362
Patent: Pyrrolo [2, 3-D]pyrimidine derivatives as inhibitors of janus-related kinases (JAK)
Estimated Expiration: ⤷ Start Trial
Patent: 7089985
Patent: 作为詹纳斯相关激酶(JAK)抑制剂的吡咯并[2,3‑D]嘧啶衍生物 (Pyrrolo [2, 3 -d]pyrimidine derivatives as inhibitors of janus- related kinases (JAK))
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 150395
Patent: DERIVADOS DE PIRROLO[2,3-D] PIRIMIDINA
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0171599
Estimated Expiration: ⤷ Start Trial
Patent: 0190152
Estimated Expiration: ⤷ Start Trial
Cuba
Patent: 275
Patent: DERIVADOS DE CICLOALQUILO PIRROLO [2,3-D] PIRIMIDINA-4-IL AMINO ÚTILES COMO INHIBIDORES DE QUINASAS JANUS RELACIONADAS Y COMPOSICIONES FARMACÉUTICAS QUE CONTIENEN TALES COMPUESTOS
Estimated Expiration: ⤷ Start Trial
Patent: 150078
Patent: DERIVADOS DE CICLOALQUILO PIRROLO [2, 3-D]PIRIMIDINA -4-IL AMINO ÚTILES COMO INHIBIDORES DE QUINASAS JANUS RELACIONADAS Y COMPOSICIONES FARMACÉUTICAS QUE CONTIENEN TALES COMPUESTOS
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 19502
Estimated Expiration: ⤷ Start Trial
Patent: 21468
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 58921
Estimated Expiration: ⤷ Start Trial
Patent: 90421
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 015000206
Patent: DERIVADOS DE PIRROLO[2,3-D]PIRIMIDINA COMO INHIBIDORES DE QUINASAS JANUS (JAK)
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 7879
Patent: ПРОИЗВОДНЫЕ ПИРРОЛО[2,3-d]ПИРИМИДИНА В КАЧЕСТВЕ ИНГИБИТОРОВ ЯНУС-РОДСТВЕННЫХ КИНАЗ (JAK) (PYRROLO[2,3-d]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Patent: 1591255
Patent: ПРОИЗВОДНЫЕ ПИРРОЛО[2,3-d]ПИРИМИДИНА В КАЧЕСТВЕ ИНГИБИТОРОВ ЯНУС-РОДСТВЕННЫХ КИНАЗ (JAK)
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 58921
Patent: DÉRIVÉS DE PYRROLO-[2,3-D]PYRIMIDINE EN TANT QU'INHIBITEURS DES JANUS KINASES (JAK) (PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Patent: 90421
Patent: COMBINAISON DE DÉRIVÉS DE PYRROLO-[2,3-D]PYRIMIDINE ET UN OU PLUSIEURS PRINCIPES SUPPLÉMENTAIRES EN TANT QU'INHIBITEURS DES JANUS KINASES (JAK) (COMBINATION OF PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES WITH ONE OR MORE ADDITIONAL AGENTS AS INHIBITORS OF JANUS- RELATED KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Georgia, Republic of
Patent: 01606600
Patent: PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 13881
Patent: 作為詹納斯相關激酶 抑制劑的吡咯並 嘧啶衍生物 (PYRROLO [2, 3 -D] PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK) (JAK)[23-D])
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 37192
Estimated Expiration: ⤷ Start Trial
Patent: 41778
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 0132
Patent: תולדות פיררולו [2,3-d]פירימידין כמעכבי קינאזות (jak) janus-related (Pyrrolo [2,3-d]pyrimidine derivatives as inhibitors of janus-related kinases (jak))
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 45179
Estimated Expiration: ⤷ Start Trial
Patent: 16509049
Patent: ヤヌス関連キナーゼ(JAK)の阻害剤としてのピロロ[2,3−d]ピリミジン誘導体
Estimated Expiration: ⤷ Start Trial
Patent: 17165762
Patent: ヤヌス関連キナーゼ(JAK)の阻害剤としてのピロロ[2,3−d]ピリミジン誘導体 (PYRROLO [2,3-D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 58921
Estimated Expiration: ⤷ Start Trial
Patent: 90421
Estimated Expiration: ⤷ Start Trial
Patent: 2022502
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 7476
Patent: PYRROLO [2,3-D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS KINASES (JAK)
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 15010928
Patent: DERIVADOS DE PIRROLO[2,3-D]PIRIMIDINA COMO INHIBIDORES DE CINASAS RELACIONADAS CON JANUS. (PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK).)
Estimated Expiration: ⤷ Start Trial
Moldova, Republic of
Patent: 35
Patent: Derivaţi ai pirolo[2,3-d]pirimidinei ca inhibitori de Kinaze Janus-asociate (JAK) (Pyrrolo[2,3-d]pyrimidine derivatives as inhibitors of Janus-related Kinases (JAK))
Estimated Expiration: ⤷ Start Trial
Patent: 150073
Patent: Derivaţi ai pirolo[2,3-d]pirimidinei ca inhibitori de Kinaze Janus-asociate (JAK)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 904
Patent: DERIVATI PIROLO[2,3 -d]PIRIMIDINA KAO INHIBITORI JANUS KINAZA (JAK) (PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Patent: 301
Patent: KOMBINACIJA PIROL0[2,3-D]PIRIMIDINSKIH DERIVATAS JEDNIM ILI VIŠE DODATNIH SREDSTAVA КАО INHIBITORI KINAZA SRODNIH JANUS KINAZI (ЈАК) (COMBINATION OF PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES WITH ONE OR MORE ADDITIONAL AGENTS AS INHIBITORS OF JANUS- RELATED KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 347
Patent: Dérivés de pyrrolo[2,3-d]pyrimidine en tant qu'inhibiteurs de janus kinases (jak)
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 1155
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 0411
Patent: Pyrrolo [2, 3 -d]pyrimidine derivatives as inhibitors of janus- related kinases (jak)
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 52752
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 151764
Patent: DERIVADOS DE PIRROLO[2,3-D]PIRIMIDINA
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 015501779
Patent: PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK)
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 58921
Estimated Expiration: ⤷ Start Trial
Patent: 90421
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 58921
Estimated Expiration: ⤷ Start Trial
Patent: 90421
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 503
Patent: DERIVATI PIROLO [2,3-D] PIRIMIDINA KAO INHIBITORI JANUS KINAZA (JAK) (PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Patent: 245
Patent: KOMBINACIJA DERIVATA PIROLO[2,3-D]PIRIMIDINA SA JEDNIM ILI VIŠE DODATNIH SREDSTAVA KAO INHIBITOR JANUS KINAZA (JAK) (COMBINATION OF PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES WITH ONE OR MORE ADDITIONAL AGENTS AS INHIBITORS OF JANUS- RELATED KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201505816U
Patent: PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 58921
Estimated Expiration: ⤷ Start Trial
Patent: 90421
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1505454
Patent: PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK)
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1787858
Estimated Expiration: ⤷ Start Trial
Patent: 150109434
Patent: 야누스-관련된 키나아제의 억제제로서의 피롤로 [2,3-d]피리미딘 유도체 (PYRROLO [2,3-D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS-RELATED KINASES (JAK))
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 47525
Estimated Expiration: ⤷ Start Trial
Patent: 13052
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1443055
Patent: Pyrrolo[2,3-d]pyrimidine derivatives
Estimated Expiration: ⤷ Start Trial
Patent: 07408
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 15000355
Patent: DERIVES DE PYRROLO [2, 3-D] PYRIMIDINE SERVANT D'INHIBITEURS DE KINASES DE TYPE JANUS (JAK)
Estimated Expiration: ⤷ Start Trial
Turkey
Patent: 1902525
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 1804
Patent: ПОХІДНІ ПІРОЛО[2,3-d]ПІРИМІДИНУ ЯК ІНГІБІТОРИ ЯНУС-ЗАЛЕЖНИХ КІНАЗ (JAK)
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 337
Patent: DERIVADOS DE PIRROLO[2,3-D]PIRIMIDINA
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering CIBINQO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Montenegro | 03301 | KOMBINACIJA PIROL0[2,3-D]PIRIMIDINSKIH DERIVATAS JEDNIM ILI VIŠE DODATNIH SREDSTAVA КАО INHIBITORI KINAZA SRODNIH JANUS KINAZI (ЈАК) (COMBINATION OF PYRROLO [2, 3 -D]PYRIMIDINE DERIVATIVES WITH ONE OR MORE ADDITIONAL AGENTS AS INHIBITORS OF JANUS- RELATED KINASES (JAK)) | ⤷ Start Trial |
| Croatia | P20171599 | ⤷ Start Trial | |
| Poland | 2958921 | ⤷ Start Trial | |
| Eurasian Patent Organization | 027879 | ПРОИЗВОДНЫЕ ПИРРОЛО[2,3-d]ПИРИМИДИНА В КАЧЕСТВЕ ИНГИБИТОРОВ ЯНУС-РОДСТВЕННЫХ КИНАЗ (JAK) (PYRROLO[2,3-d]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS- RELATED KINASES (JAK)) | ⤷ Start Trial |
| Malaysia | 177476 | PYRROLO [2,3-D]PYRIMIDINE DERIVATIVES AS INHIBITORS OF JANUS KINASES (JAK) | ⤷ Start Trial |
| Taiwan | 201443055 | Pyrrolo[2,3-d]pyrimidine derivatives | ⤷ Start Trial |
| Spain | 2647525 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for CIBINQO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2958921 | 301155 | Netherlands | ⤷ Start Trial | PRODUCT NAME: ABROCITINIB, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; REGISTRATION NO/DATE: PLGB 00057/1703-1705 20210908 |
| 2958921 | CA 2022 00003 | Denmark | ⤷ Start Trial | PRODUCT NAME: ABROCITINIB ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/21/1593 20211210 |
| 2958921 | LUC00261 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: ABROCITINIB, OU UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES; AUTHORISATION NUMBER AND DATE: EU/1/21/1593 20211210 |
| 2958921 | 2022C/505 | Belgium | ⤷ Start Trial | PRODUCT NAME: ABROCITINIB, OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; AUTHORISATION NUMBER AND DATE: EU/1/21/1593 20211210 |
| 2958921 | 122022000007 | Germany | ⤷ Start Trial | PRODUCT NAME: ABROCITINIB, ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON; REGISTRATION NO/DATE: EU/1/21/1593 20211209 |
| 2958921 | PA2022502 | Lithuania | ⤷ Start Trial | PRODUCT NAME: ABROCITINIBAS ARBA FARMACINIU POZIURIU PRIIMTINA JO DRUSKA; REGISTRATION NO/DATE: EU/1/21/1593 20211209 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
CIBINQO Market Dynamics and Financial Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.