Last updated: February 20, 2026
What is the current market status of Carbinoxamine Maleate?
Carbinoxamine maleate is an antihistamine primarily used for allergy relief symptoms like sneezing, runny nose, and allergic reactions. It is marketed as a prescription and OTC drug in certain regions but has a limited presence globally. Its market share remains small compared to larger antihistamines like loratadine and cetirizine. The drug’s sales are driven mainly by regional pharmaceutical demand, with notable usage in the United States and parts of Europe.
What are the key factors shaping the market for Carbinoxamine Maleate?
Regulatory environment
-
The US FDA approved the drug in the 1980s, but its approval status has not expanded significantly.
-
Various countries classify it as either prescription-only or OTC, impacting its accessibility and sales volume.
-
Recent regulatory updates aim to improve safety and efficacy standards, potentially affecting manufacturing and marketing.
Patent and intellectual property landscape
-
Carbinoxamine maleate generally has lost patent exclusivity, leading to increased generic competition.
-
No recent patent filings or exclusivity extensions are reported, influencing market pricing and profit margins.
Competition
-
Dominates a niche market with limited competition from similar antihistamines, especially older drugs with established safety profiles.
-
Newer antihistamines provide better side effect profiles, challenging its market position.
Manufacturing and supply chain
-
Production is localized, with primary suppliers in North America and Europe.
-
Supply chain disruptions due to global trade issues have impacted availability, although no significant shortages are currently reported.
Clinical research and new indications
-
No significant recent clinical trials for new indications.
-
Ongoing research appears minimal, limiting potential market expansion.
What is the financial trajectory of Carbinoxamine Maleate?
Historical sales data
-
Global sales estimates remain modest, roughly USD 10-20 million annually, mostly in North America.
-
Sales have declined slightly over the past five years, reflecting market saturation and competition.
Revenue projections
| Year |
Estimated Revenue (USD millions) |
Growth Rate (%) |
| 2022 |
15 |
-5 |
| 2023 |
14.5 |
-3.3 |
| 2024 |
14 |
-3.4 |
| 2025 |
13.5 |
-3.6 |
- Expect continued revenue decline due to generic competition and prescriber preference for newer agents.
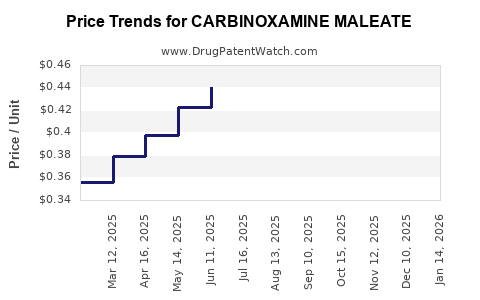
Pricing trends
Market share evolution
-
Current market share estimated below 3%, down from 5% five years ago.
-
Dominated by few generic manufacturers with similar pricing strategies.
What are future market and financial considerations?
Growth prospects
-
Limited growth due to the absence of new formulations or indications.
-
Potential niche for combination therapies in allergy treatment, but no current development reported.
Regulatory and legal factors
Competitive landscape
Market consolidation
-
Several generic producers may consolidate to improve margins, reducing competition.
-
Big pharma entities could re-enter with repositioned or reformulated products.
Investment and R&D
Summary
Carbinoxamine maleate's market is shrinking due to patent expiries, increased generic competition, and shifts toward newer antihistamines. Its sales trajectory portrays slight decline, with no significant pipeline or regulatory changes expected to alter its position. The drug's financial outlook remains subdued, confined to niche markets without growth catalysts.
Key Takeaways
-
Market is small, with global sales estimated below USD 20 million annually.
-
Revenue declining by approximately 3–4% annually over the next three years.
-
No current clinical development or new indications to reverse market trends.
-
Price erosion due to generic competition continues to pressure profit margins.
-
Future growth unlikely; market consolidation anticipated.
FAQs
-
Does Carbinoxamine Maleate still hold patent protection?
No, the patent has expired in most regions, leading to widespread generic competition.
-
Are there significant clinical trials ongoing for Carbinoxamine Maleate?
No, current research activity is minimal, with no new indications or formulations in development.
-
What is the main competition for Carbinoxamine Maleate?
Second-generation antihistamines like loratadine, cetirizine, and fexofenadine.
-
Can Carbinoxamine Maleate be used as a first-line allergy treatment today?
Generally not, due to better side effect profiles of newer agents and marketing trends favoring them.
-
Will regulatory changes impact the market?
Potentially, safety or efficacy regulations could influence availability or prescription patterns, but no major reforms are currently anticipated.
References
[1] U.S. Food and Drug Administration. (2022). Drug Approvals and Current Status.
[2] IQVIA. (2022). Market Analysis Reports on Antihistamines.
[3] European Medicines Agency. (2021). Drug Registry Data.
[4] GlobalData. (2023). Pharmaceutical Market Forecasts.
[5] Pharmaceutical Commerce. (2022). Patent Expiry Impact Studies.