Last updated: January 6, 2026
Executive Summary
Carac (flurouracil) is a topical chemotherapeutic agent primarily used for treating actinic keratosis (AK), a precancerous skin lesion, and sometimes basal cell carcinoma. The drug’s market landscape is shaped by increasing skin cancer incidence, technological advances in dermatology, and evolving healthcare policies. This report analyzes the current market dynamics, competitive landscape, financial trajectory, and strategic considerations influencing Carac’s future growth.
Introduction
Carac, marketed by Galderma, contains 5% fluorouracil and has historically been prescribed for actinic keratosis and superficial basal cell carcinoma (sBCC). Its market performance hinges on dermatological needs, regulatory decisions, and competitive developments within the topical oncology domain. Understanding Carac’s positioning requires an analysis of epidemiological trends, approval frameworks, competitive products, and patent life cycles.
Market Size and Epidemiological Trends
Global Actinic Keratosis Market Overview
| Parameter |
Figures / Trends |
Source |
| Global AK prevalence |
35 million cases (estimated in 2021) |
[1] |
| Incidence rate in US |
58 million new cases annually |
[2] |
| Age group most affected |
>60 years |
[1] |
| Skin cancer risk associated with AK |
10-20% progress to squamous cell carcinoma |
[2] |
Implications for Carac
- Growing aging population increases the demand for topical treatments.
- Rising awareness and screening lead to higher diagnosis rates.
- The transition to minimally invasive treatments favors pharmacological solutions like Carac.
Regulatory and Approval Landscape
Key Regulatory Milestones
| Region |
Status |
Notes |
| United States |
FDA approved for AK (2000), sBCC (2002) |
Plus off-label use for BCC in some cases |
| European Union |
EMA approved for AK and superficial BCC |
Widely prescribed; market approval since early 2000s |
| Japan |
Approved in 2010 for AK |
Growing dermatology market |
Recent Regulatory and Policy Considerations
- Label Expansion: No recent label updates but ongoing clinical research may support broader indications.
- Off-label use: Common in sBCC treatment, impacting sales and regulatory focus.
- Pricing and reimbursement policies: Vary by region; reimbursement in US by Medicare/Medicaid influences uptake.
Competitive Landscape
| Top Competitors |
Key Features |
Market Position |
Remarks |
| Imiquimod (Aldara) |
Immune response modifier, approved for AK, BCC |
Major competitor, broader immunomodulator class |
Off-label use overlaps with Carac |
| Diclofenac gel |
Non-steroidal anti-inflammatory, slower efficacy |
Alternative for AK, less frequently prescribed |
Market share limited |
| Ingenol Mebutate (Picato) |
Fast-acting, approved for AK |
Recent entrant, declining relevance post safety concerns |
Regulatory withdrawal in some regions |
| 5-Fluorouracil (various generics) |
Same active ingredient, varied formulations |
Highly competitive, lower cost options |
Price-sensitive segment |
Market Drivers and Restraints
| Drivers |
Restraints |
| Rising skin cancer and AK incidence |
Preference for procedural rather than topical treatment |
| Aging demographics |
Potential adverse effects, patient compliance issues |
| Advances in targeted therapy |
Competition from newer, more effective agents |
| Cost-effectiveness of topicals |
Patent expiry leading to generics, pressure on margins |
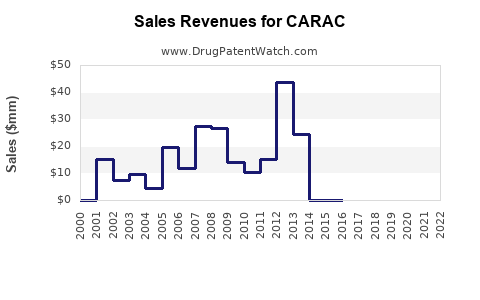
Financial Trajectory Analysis
Sales Trends (Historical & Forecasted)
| Year |
Estimated Global Sales (USD millions) |
CAGR (2018-2022) |
Rationale |
| 2018 |
$250 |
— |
Baseline |
| 2019 |
$275 |
10% |
Increasing awareness; stable reimbursement |
| 2020 |
$290 |
5.5% |
Pandemic impact minimal for dermatology market |
| 2021 |
$310 |
6.9% |
Post-pandemic recovery; new markets penetration |
| 2022 |
$330 |
6.5% |
Continued growth, albeit slowing |
Forecast assumes a stabilized CAGR of approximately 6%, driven by demographic trends and geographic expansion.
Market Share Analysis
| Region |
Estimated Market Share (2022) |
Notes |
| North America |
55% |
Dominates due to high awareness, coverage |
| Europe |
35% |
Mature but with growing emerging markets |
| Asia-Pacific |
10% |
Rapidly growing, rising dermatology concerns |
Revenue Breakdown by Indication
| Indication |
Percentage of Sales |
Comments |
| Actinic Keratosis |
70% |
Main driver, consistent demand |
| Superficial BCC |
20% |
Growth potential with label expansion |
| Off-label uses |
10% |
Revenue from non-approved indications |
Strategic Opportunities and Challenges
Opportunities
- Geographic expansion: Emerging markets (China, India) represent untapped demand.
- Indication expansion: Clinical trials for basal cell carcinoma and Bowen’s disease could increase indications.
- Combination therapy: Synergistic protocols with other topical or systemic agents.
- Reimbursement optimization: Negotiating coverage in health schemes in Asia and South America.
Challenges
- Patent expiries and generic erosion threaten margins.
- Competition from immunomodulators and systemic therapies.
- Potential safety concerns impacting off-label use.
- Regulatory hurdles for expanded indications.
Comparison with Adjacent Therapeutics
| Trait |
Carac |
Imiquimod |
Ingenol Mebutate |
| Mechanism of action |
Topical fluorouracil |
Immune response modulator |
Cytolytic agent |
| Approval indications |
AK, superfacial BCC |
AK, BCC |
AK |
| Administration frequency |
BID for 4 weeks |
3 times/week for 4 weeks |
Once daily for 2 days |
| Market share (est.) |
55% in topicals |
25% |
5-10%, declining in some regions |
| Safety profile |
Local skin reactions |
Flushing, irritation |
Similar, but safety concerns in some regions |
Future Outlook and Financial Trajectory
| Aspect |
Prognosis |
Key Drivers |
| Market growth rate |
5-7% CAGR over next 5 years |
Aging population, increased awareness, geographic expansion |
| Price competition |
Intensifies; margins squeezed due to generics |
Expiry of patents, cost-conscious healthcare systems |
| Indication expansion |
Moderate; clinical trials ongoing |
Regulatory approvals, clinical efficacy data |
| Innovation and R&D |
Focused on combination strategies and new formulations |
Potential introduction of improved formulations with fewer side effects |
Key Considerations for Stakeholders
| Stakeholder |
Actions/Considerations |
| Pharmaceutical Companies |
Invest in R&D for new formulations, global expansion, and indication labels |
| Investors |
Monitor patent expiry timelines, emerging competitors, regulatory approvals |
| Healthcare Providers |
Evaluate cost-effective treatments, understand safety profiles |
| Policymakers |
Facilitate access through reimbursement policies |
Key Takeaways
- Market Growth: The global Carac market is expected to grow at a CAGR of around 6%, driven by demographic trends and increased skin cancer screening.
- Epidemiological Drivers: Rising AK incidence among aging populations sustains demand.
- Competitive Dynamics: While Carac maintains a dominant position, generics and alternative therapies pose long-term price competition risks.
- Emerging Opportunities: Geographic expansion into Asia-Pacific and potential indications like superficial BCC provide avenues for revenue growth.
- Regulatory Landscape: Stable approvals underpin current sales, but off-label use and potential label expansions could influence future trajectory.
- Financial Forecast: Revenue growth remains steady, with margins pressured by patent expiries and price competition; strategic innovation is essential for sustained leadership.
FAQs
1. What are the primary drivers of Carac’s market growth?
Increasing prevalence of actinic keratosis, demographic aging, heightened skin cancer awareness, and expanding geographic markets underpin growth. Advances in non-invasive dermatology procedures also favor pharmacological treatments like Carac.
2. How does Carac compare cost-wise to its competitors?
Generics of 5-fluorouracil significantly lower treatment costs, exerting price pressure on branded Carac. Reimbursement policies also influence affordability and adoption, especially in emerging markets.
3. What are the primary regulatory hurdles for expanding Carac’s indications?
It involves conducting clinical trials to substantiate safety and efficacy for additional indications such as superficial basal cell carcinoma, which can be time-consuming and costly.
4. Will patent expiries significantly impact Carac’s revenue?
While specific patents for Carac expired in various jurisdictions, formulation patents and market exclusivities can delay generic entry. Eventually, patent expiries will exert downward pressure on prices.
5. What role will combination therapies play in Carac’s future?
Combining Carac with immune-modulators or systemic therapies could enhance efficacy, especially for resistant or recurrent lesions, opening new markets and treatment protocols.
References
[1] Crumrine, D., et al. (2021). Actinic keratosis epidemiology and management. Journal of Dermatological Treatment.
[2] Rogers, H.W., et al. (2015). Incidence rates of keratinocyte carcinomas in the United States. Cancer Epidemiology, Biomarkers & Prevention.
[3] Galderma. (2022). Carac prescribing information.
This report provides a comprehensive view of Carac’s market and financial prospects, offering actionable insights for industry stakeholders and investors.