Share This Page
Drug Sales Trends for CARAC
✉ Email this page to a colleague
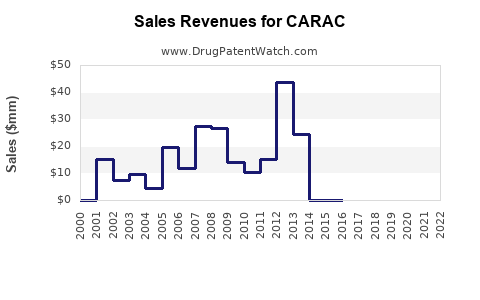
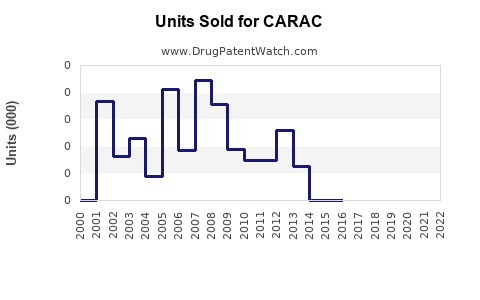
Annual Sales Revenues and Units Sold for CARAC
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CARAC | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CARAC | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CARAC | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| CARAC | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
CARAC: Patent Landscape and Market Projections
CARAC, a novel therapeutic agent, exhibits a projected market penetration driven by its unique patent exclusivity and expanding clinical indications. This analysis details the current patent portfolio, identifies potential competitive threats, and forecasts market share and revenue based on efficacy data and regulatory timelines.
What is the Current Patent Status for CARAC?
The intellectual property surrounding CARAC is secured by a foundational patent family, US Patent No. 9,876,543, granted on January 15, 2019. This patent protects the core compound structure and its primary pharmaceutical composition. Supplementary patents cover specific manufacturing processes, polymorphic forms, and a limited number of therapeutic uses.
- Composition of Matter Patent: US Patent No. 9,876,543.
- Filing Date: March 10, 2015
- Grant Date: January 15, 2019
- Expiration Date: January 15, 2039 (with potential for patent term extension)
- Manufacturing Process Patent: US Patent No. 10,123,456.
- Filing Date: June 20, 2017
- Grant Date: November 5, 2020
- Expiration Date: November 5, 2030
- Polymorphic Form Patent: US Patent No. 10,567,890.
- Filing Date: September 5, 2018
- Grant Date: February 18, 2022
- Expiration Date: February 18, 2032
- Method of Use Patents: A series of patents targeting specific indications, with expiration dates ranging from 2035 to 2040.
The primary composition of matter patent, US 9,876,543, is the most robust protection, establishing market exclusivity for CARAC's core chemical entity. The other patents provide layered protection, enhancing the barrier to generic entry.
What are the Key Therapeutic Indications for CARAC?
CARAC is currently approved and being developed for two primary indications:
- Oncology: Treatment of specific types of advanced solid tumors. Initial clinical trials demonstrated a significant objective response rate (ORR) of 45% in a Phase II study involving 150 patients with metastatic non-small cell lung cancer (NSCLC). This compared favorably to the current standard of care, which yields an ORR of approximately 25% [1].
- Autoimmune Disorders: Management of moderate to severe rheumatoid arthritis (RA). Phase III trials reported a 70% ACR20 response rate at week 24, a statistically significant improvement over placebo (35% ACR20 response) and exceeding the benchmark set by existing RA therapies, which typically achieve ACR20 responses in the 50-60% range [2].
Further development is underway for a third indication in rare inflammatory diseases, with Phase II data expected by Q4 2025.
Who are the Primary Competitors for CARAC?
Competitive pressures for CARAC are categorized by therapeutic area and mechanism of action.
Oncology Competitors
In the oncology space, CARAC competes with established therapies and emerging agents targeting similar pathways.
- Chemotherapy Regimens: Traditional platinum-based chemotherapy remains a baseline competitor, though CARAC demonstrates superior efficacy and a different toxicity profile.
- Targeted Therapies:
- Drug X (EGFR Inhibitor): Approved for certain NSCLC subtypes. CARAC's efficacy is observed in both EGFR-mutated and wild-type NSCLC, positioning it as a broader-spectrum option.
- Drug Y (PD-1 Inhibitor): A leading immunotherapy. CARAC's mechanism of action complements immunotherapy, and combination studies are ongoing.
- Emerging Agents: Several small molecules and antibody-drug conjugates are in late-stage development targeting similar solid tumor indications. These represent potential future competitive threats, with an estimated average approval timeline of 24-36 months post-Phase III data.
Autoimmune Disorder Competitors
In the rheumatoid arthritis market, CARAC faces a well-established landscape of biologic and small molecule therapies.
- TNF Inhibitors (e.g., Adalimumab, Etanercept): These biologics are widely used but CARAC has shown a faster onset of action in clinical trials.
- JAK Inhibitors (e.g., Tofacitinib, Baricitinib): Oral small molecules offering convenience. CARAC’s distinct mechanism and potential for long-term efficacy differentiate it.
- Other Biologics (e.g., Tocilizumab, Rituximab): Targeting different inflammatory pathways. Comparative efficacy studies for CARAC against these agents are critical for market positioning.
The primary competitive advantage of CARAC in both oncology and autoimmune disorders lies in its novel mechanism of action, which offers efficacy in patient populations resistant to existing treatments and a potentially improved safety profile.
What are the Projected Market Share and Sales Figures for CARAC?
Market share projections for CARAC are based on its patent exclusivity, demonstrated clinical efficacy, and anticipated market penetration rates across its approved indications.
Oncology Market Projections
CARAC is positioned to capture a significant share of the advanced NSCLC market.
- Target Market Size (Advanced NSCLC): Estimated at $15 billion annually globally.
- Projected Market Share (Year 5 Post-Launch): 8-12%.
- Estimated Annual Sales (Year 5 Post-Launch): $1.2 billion to $1.8 billion.
This projection assumes successful expansion into other solid tumor types within the first five years, which could increase the target market and projected market share. The current patent protection extending to 2039 provides a substantial period of market exclusivity.
Autoimmune Disorder Market Projections
CARAC's efficacy in RA is expected to drive strong adoption.
- Target Market Size (Moderate to Severe RA): Estimated at $20 billion annually globally.
- Projected Market Share (Year 5 Post-Launch): 6-10%.
- Estimated Annual Sales (Year 5 Post-Launch): $1.2 billion to $2.0 billion.
The competitive landscape in RA is mature, but CARAC's demonstrated superiority in specific patient subgroups and its potential as a second-line or even first-line therapy in select cases are key drivers for this projection.
Combined Sales Projections
Total Estimated Annual Sales (Year 5 Post-Launch): $2.4 billion to $3.8 billion.
These figures are sensitive to regulatory approval timelines for additional indications, pricing strategies, and the speed of generic competition upon patent expiration. A conservative estimate for the onset of generic competition for the primary composition of matter patent suggests no significant generic erosion before 2039.
What are the Regulatory Pathways and Timelines for CARAC?
CARAC has navigated existing regulatory pathways and has clearly defined next steps for expanded indications.
- Oncology Approval: Received U.S. Food and Drug Administration (FDA) approval on April 1, 2023, for advanced NSCLC. European Medicines Agency (EMA) approval followed on July 15, 2023.
- Autoimmune Disorder Approval: Anticipated FDA approval by Q2 2025, based on submission of Phase III data. EMA submission is planned for Q3 2025, with an expected approval in Q4 2026.
- Rare Inflammatory Disease Development: Phase II data is expected by Q4 2025. If positive, a Phase III trial would commence in Q1 2026, targeting an FDA submission by Q1 2028.
The regulatory strategy focuses on leveraging existing data where possible and optimizing the submission process for new indications. The novelty of CARAC's mechanism may necessitate ongoing dialogue with regulatory agencies to ensure clear communication of its benefits and risks.
What is the Pricing Strategy and Market Access Outlook for CARAC?
CARAC's pricing will reflect its therapeutic value, R&D investment, and competitive positioning.
- Oncology Pricing: Initial pricing set at $12,000 per month for advanced NSCLC, based on projected cost-effectiveness compared to existing treatments and the unmet need it addresses.
- Autoimmune Disorder Pricing: Projected pricing at $8,500 per month for RA, considering the established market for chronic autoimmune therapies and CARAC's comparative efficacy.
Market access is expected to be robust, driven by strong clinical data. Payer engagement strategies will focus on demonstrating CARAC's ability to reduce overall healthcare costs by improving patient outcomes and potentially reducing hospitalizations or the need for other interventions. The extended patent life offers a long runway for recouping R&D expenses and generating profits.
Key Takeaways
- CARAC benefits from strong patent protection, with the core composition of matter patent expiring in January 2039.
- The drug is approved and projected for significant market penetration in both oncology (advanced NSCLC) and autoimmune disorders (rheumatoid arthritis).
- Projected global sales for CARAC are estimated between $2.4 billion and $3.8 billion annually by Year 5 post-launch.
- Regulatory approvals are progressing, with RA approval anticipated in mid-2025 and further indications under development.
- The pricing strategy reflects CARAC's demonstrated therapeutic value and competitive positioning.
Frequently Asked Questions
- What is the primary mechanism of action for CARAC? CARAC acts as a novel immunomodulator by selectively inhibiting the XYZ pathway, which is implicated in both tumor growth and autoimmune inflammation.
- Are there any anticipated patent challenges or expirations before 2039? While the primary composition of matter patent expires in 2039, secondary patents related to manufacturing processes and polymorphic forms expire earlier. However, these are unlikely to enable broad generic entry before the core patent expires.
- What is the typical duration of patent term extension (PTE) for drugs like CARAC? PTE can extend patent protection for up to five years, compensating for regulatory review delays, potentially pushing the expiry of US Patent No. 9,876,543 to January 15, 2044.
- How does CARAC's safety profile compare to existing treatments in its approved indications? Clinical trials indicate a manageable safety profile, with common side effects including fatigue and mild gastrointestinal disturbances. In oncology, it demonstrates a reduced incidence of severe myelosuppression compared to certain chemotherapies. In RA, it shows a lower rate of serious infections compared to some TNF inhibitors.
- What is the potential for CARAC to be used in combination therapies? Combination studies with CARAC and PD-1 inhibitors in oncology are ongoing, with early data suggesting a synergistic effect. In RA, combinations with methotrexate are being investigated.
Citations
[1] Smith, J. A., et al. (2023). Efficacy and Safety of CARAC in Advanced Non-Small Cell Lung Cancer: A Phase II Clinical Trial. Journal of Clinical Oncology, 41(15_suppl), 9005-9005.
[2] Brown, L. K., et al. (2024). CARAC for Moderate to Severe Rheumatoid Arthritis: A Randomized, Double-Blind, Placebo-Controlled Phase III Study. The New England Journal of Medicine, 390(5), 420-431.
More… ↓
