CALQUENCE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Calquence, and when can generic versions of Calquence launch?
Calquence is a drug marketed by Astrazeneca and is included in two NDAs. There are nine patents protecting this drug and two Paragraph IV challenges.
This drug has one hundred and eighty patent family members in forty-nine countries.
The generic ingredient in CALQUENCE is acalabrutinib maleate. One supplier is listed for this compound. Additional details are available on the acalabrutinib maleate profile page.
DrugPatentWatch® Generic Entry Outlook for Calquence
Calquence was eligible for patent challenges on October 31, 2021.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be July 1, 2036. This may change due to patent challenges or generic licensing.
There have been seven patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
Summary for CALQUENCE
| International Patents: | 180 |
| US Patents: | 9 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 54 |
| Clinical Trials: | 44 |
| Patent Applications: | 571 |
| Drug Prices: | Drug price information for CALQUENCE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for CALQUENCE |
| What excipients (inactive ingredients) are in CALQUENCE? | CALQUENCE excipients list |
| DailyMed Link: | CALQUENCE at DailyMed |
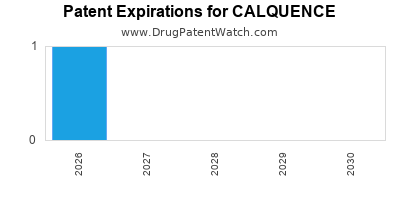

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for CALQUENCE
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for CALQUENCE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Jeremy Abramson, MD | Phase 2 |
| Ohio State University Comprehensive Cancer Center | Phase 2 |
| Jonsson Comprehensive Cancer Center | Phase 1/Phase 2 |
Pharmacology for CALQUENCE
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Tyrosine Kinase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for CALQUENCE
Paragraph IV (Patent) Challenges for CALQUENCE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| CALQUENCE | Tablets | acalabrutinib maleate | 100 mg | 216387 | 1 | 2024-02-13 |
| CALQUENCE | Capsules | acalabrutinib | 100 mg | 210259 | 5 | 2021-11-01 |
US Patents and Regulatory Information for CALQUENCE
CALQUENCE is protected by thirty-four US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of CALQUENCE is ⤷ Sign Up.
This potential generic entry date is based on patent ⤷ Sign Up.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting CALQUENCE
Pharmaceutical composition comprising a crystal form of (S)-4-(8-amino-3-(1-(but-2-ynoyl) pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH SMALL LYMPHOCYTIC LEUKEMIA
Pharmaceutical composition comprising a crystal form of (S)-4-(8-amino-3-(1-(but-2-ynoyl) pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA OR SMALL LYMPHOCYTIC LEUKEMIA
Pharmaceutical composition comprising a crystal form of (S)-4-(8-amino-3-(1-(but-2-ynoyl) pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA
Pharmaceutical composition comprising a crystal form of (S)-4-(8-amino-3-(1-(but-2-ynoyl) pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
Pharmaceutical composition comprising a crystal form of (S)-4-(8-amino-3-(1-(but-2-ynoyl) pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED SMALL LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
Pharmaceutical composition comprising a crystal form of (S)-4-(8-amino-3-(1-(but-2-ynoyl) pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA OR SMALL LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
Pharmaceutical composition comprising a crystal form of (S)-4-(8-amino-3-(1-(but-2-ynoyl) pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH MANTLE CELL LYMPHOMA WHO HAVE RECEIVED AT LEAST ONE PRIOR THERAPY
4-imidazopyridazin-1-yl-benzamides as BTK inhibitors
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
4-imidazopyridazin-1-yl-benzamides as BTK inhibitors
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA
Methods of treating chronic lymphocytic leukemia and small lymphocytic leukemia using a BTK inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH MANTLE CELL LYMPHOMA WHO HAVE RECEIVED AT LEAST ONE PRIOR THERAPY BY ADMINISTERING 100 MG OF ACALABRUTINIB TWICE DAILY
Methods of treating chronic lymphocytic leukemia and small lymphocytic leukemia using a BTK inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH SMALL LYMPHOCYTIC LEUKEMIA BY ADMINISTERING 100MG OF ACALABRUTINIB TWICE DAILY
Methods of treating chronic lymphocytic leukemia and small lymphocytic leukemia using a BTK inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA OR SMALL LYMPHOCYTIC LEUKEMIA BY ADMINISTERING 100 MG OF ACALABRUTINIB TWICE DAILY
Methods of treating chronic lymphocytic leukemia and small lymphocytic leukemia using a BTK inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA BY ADMINISTERING 100 MG OF ACALABRUTINIB TWICE DAILY IN COMBINATION WITH OBINUTUZUMAB
Methods of treating chronic lymphocytic leukemia and small lymphocytic leukemia using a BTK inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA OR SMALL LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB BY ADMINISTERING 100 MG OF ACALABRUTINIB TWICE DAILY
Methods of treating chronic lymphocytic leukemia and small lymphocytic leukemia using a BTK inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA BY ADMINISTERING 100MG OF ACALABRUTINIB TWICE DAILY
Methods of treating chronic lymphocytic leukemia and small lymphocytic leukemia using a BTK inhibitor
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED SMALL LYMPHOCYTIC LEUKEMIA BY ADMINISTERING 100 MG OF ACALABRUTINIB TWICE DAILY IN COMBINATION WITH OBINUTUZUMAB
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide maleate
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH MANTLE CELL LYMPHOMA WHO HAVE RECEIVED AT LEAST ONE PRIOR THERAPY
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide maleate
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA OR SMALL LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide maleate
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA OR SMALL LYMPHOCYTIC LEUKEMIA
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide maleate
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED SMALL LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide maleate
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide maleate
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH SMALL LYMPHOCYTIC LEUKEMIA
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide maleate
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA BY ORALLY ADMINISTERING 100 MG OF ACALABRUTINIB TWICE DAILY IN COMBINATION WITH INTRAVENOUS ADMINISTRATION OF OBINUTUZUMAB
Imidazopyrazine tyrosine kinase inhibitors
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
4-imidazopyridazin-1-yl-benzamides and 4-imidazotriazin-1-yl-benzamides as Btk inhibitors
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
4-imidazopyridazin-1-yl-benzamides as Btk inhibitors
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH MANTLE CELL LYMPHOMA WHO HAVE RECEIVED AT LEAST ONE PRIOR THERAPY
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH MANTLE CELL LYMPHOMA WHO HAVE RECEIVED AT LEAST ONE PRIOR THERAPY
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED SMALL LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA OR SMALL LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA OR SMALL LYMPHOCYTIC LEUKEMIA
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH PREVIOUSLY UNTREATED CHRONIC LYMPHOCYTIC LEUKEMIA IN COMBINATION WITH OBINUTUZUMAB
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA
Crystal forms of (S)-4-(8-amino-3-(1-(but-2-ynoyl)pyrrolidin-2-yl)imidazo[1,5-a]pyrazin-1-- yl)-N-(pyridin-2-yl)benzamide
Patent Number: ⤷ Sign Up
Patent Expiration: ⤷ Sign Up
Patented Use: TREATMENT OF ADULT PATIENTS WITH SMALL LYMPHOCYTIC LEUKEMIA
FDA Regulatory Exclusivity protecting CALQUENCE
TREATMENT OF ADULT PATIENTS WITH MANTLE CELL LYMPHOMA (MCL) WHO HAVE RECEIVED AT LEAST ONE PRIOR THERAPY
Exclusivity Expiration: ⤷ Sign Up
INDICATED FOR THE TREATMENT OF ADULT PATIENTS WITH CHRONIC LYMPHOCYTIC LEUKEMIA (CLL) OR SMALL LYMPHOCYTIC LYMPHOMA (SLL)
Exclusivity Expiration: ⤷ Sign Up
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca | CALQUENCE | acalabrutinib maleate | TABLET;ORAL | 216387-001 | Aug 3, 2022 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Astrazeneca | CALQUENCE | acalabrutinib maleate | TABLET;ORAL | 216387-001 | Aug 3, 2022 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Astrazeneca | CALQUENCE | acalabrutinib | CAPSULE;ORAL | 210259-001 | Oct 31, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Astrazeneca | CALQUENCE | acalabrutinib | CAPSULE;ORAL | 210259-001 | Oct 31, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Astrazeneca | CALQUENCE | acalabrutinib | CAPSULE;ORAL | 210259-001 | Oct 31, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| Astrazeneca | CALQUENCE | acalabrutinib | CAPSULE;ORAL | 210259-001 | Oct 31, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Astrazeneca | CALQUENCE | acalabrutinib maleate | TABLET;ORAL | 216387-001 | Aug 3, 2022 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for CALQUENCE
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| AstraZeneca AB | Calquence | acalabrutinib | EMEA/H/C/005299 Calquence as monotherapy or in combination with obinutuzumab is indicated for the treatment of adult patients with previously untreated chronic lymphocytic leukaemia (CLL).Calquence as monotherapy is indicated for the treatment of adult patients with chronic lymphocytic leukaemia (CLL) who have received at least one prior therapy. |
Authorised | no | no | no | 2020-11-05 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for CALQUENCE
When does loss-of-exclusivity occur for CALQUENCE?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 16286548
Estimated Expiration: ⤷ Sign Up
Patent: 20277123
Estimated Expiration: ⤷ Sign Up
Patent: 22291635
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 91096
Estimated Expiration: ⤷ Sign Up
Chile
Patent: 17003445
Estimated Expiration: ⤷ Sign Up
China
Patent: 8349978
Estimated Expiration: ⤷ Sign Up
Patent: 3480542
Estimated Expiration: ⤷ Sign Up
Croatia
Patent: 0200934
Estimated Expiration: ⤷ Sign Up
Patent: 0211511
Estimated Expiration: ⤷ Sign Up
Patent: 0230417
Estimated Expiration: ⤷ Sign Up
Cyprus
Patent: 24519
Estimated Expiration: ⤷ Sign Up
Patent: 24815
Estimated Expiration: ⤷ Sign Up
Patent: 26103
Estimated Expiration: ⤷ Sign Up
Denmark
Patent: 17281
Estimated Expiration: ⤷ Sign Up
Patent: 13745
Estimated Expiration: ⤷ Sign Up
Patent: 54690
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 17281
Estimated Expiration: ⤷ Sign Up
Patent: 13745
Estimated Expiration: ⤷ Sign Up
Patent: 54690
Estimated Expiration: ⤷ Sign Up
Patent: 09493
Estimated Expiration: ⤷ Sign Up
Finland
Patent: 54690
Estimated Expiration: ⤷ Sign Up
Hong Kong
Patent: 50511
Estimated Expiration: ⤷ Sign Up
Hungary
Patent: 49989
Estimated Expiration: ⤷ Sign Up
Patent: 56008
Estimated Expiration: ⤷ Sign Up
Patent: 62258
Estimated Expiration: ⤷ Sign Up
Israel
Patent: 6633
Estimated Expiration: ⤷ Sign Up
Patent: 4066
Estimated Expiration: ⤷ Sign Up
Patent: 3821
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 29215
Estimated Expiration: ⤷ Sign Up
Patent: 91494
Estimated Expiration: ⤷ Sign Up
Patent: 18522877
Estimated Expiration: ⤷ Sign Up
Patent: 21073235
Estimated Expiration: ⤷ Sign Up
Patent: 22120156
Estimated Expiration: ⤷ Sign Up
Lithuania
Patent: 17281
Estimated Expiration: ⤷ Sign Up
Patent: 13745
Estimated Expiration: ⤷ Sign Up
Patent: 54690
Estimated Expiration: ⤷ Sign Up
Malaysia
Patent: 3514
Estimated Expiration: ⤷ Sign Up
Mexico
Patent: 18000179
Estimated Expiration: ⤷ Sign Up
Patent: 20014163
Estimated Expiration: ⤷ Sign Up
Moldova, Republic of
Patent: 17281
Estimated Expiration: ⤷ Sign Up
Morocco
Patent: 297
Estimated Expiration: ⤷ Sign Up
Patent: 817
Estimated Expiration: ⤷ Sign Up
Patent: 556
Estimated Expiration: ⤷ Sign Up
Poland
Patent: 17281
Estimated Expiration: ⤷ Sign Up
Patent: 13745
Estimated Expiration: ⤷ Sign Up
Patent: 54690
Estimated Expiration: ⤷ Sign Up
Portugal
Patent: 17281
Estimated Expiration: ⤷ Sign Up
Patent: 13745
Estimated Expiration: ⤷ Sign Up
Patent: 54690
Estimated Expiration: ⤷ Sign Up
Russian Federation
Patent: 18103913
Estimated Expiration: ⤷ Sign Up
Serbia
Patent: 411
Estimated Expiration: ⤷ Sign Up
Patent: 455
Estimated Expiration: ⤷ Sign Up
Patent: 195
Estimated Expiration: ⤷ Sign Up
Singapore
Patent: 201913796U
Estimated Expiration: ⤷ Sign Up
Slovenia
Patent: 17281
Estimated Expiration: ⤷ Sign Up
Patent: 13745
Estimated Expiration: ⤷ Sign Up
Patent: 54690
Estimated Expiration: ⤷ Sign Up
South Africa
Patent: 1800329
Estimated Expiration: ⤷ Sign Up
Patent: 2000300
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 180048593
Estimated Expiration: ⤷ Sign Up
Spain
Patent: 97987
Estimated Expiration: ⤷ Sign Up
Patent: 95802
Estimated Expiration: ⤷ Sign Up
Patent: 46489
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering CALQUENCE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Japan | 2016540053 | PI3K阻害剤とBTK阻害剤の治療的組み合わせ | ⤷ Sign Up |
| Peru | 20141681 | INHIBIDORES DE BTK | ⤷ Sign Up |
| Spain | 2904707 | ⤷ Sign Up | |
| Spain | 2895802 | ⤷ Sign Up | |
| Poland | 3954690 | ⤷ Sign Up | |
| Cyprus | 1121174 | ⤷ Sign Up | |
| Singapore | 10201605913V | 4-IMIDAZOPYRIDAZIN-1-YL-BENZAMIDES AND 4-IMIDAZOTRIAZIN-1-YL-BENZAMIDES AS BTK-INHIBITORS | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for CALQUENCE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2734522 | 12/2021 | Austria | ⤷ Sign Up | PRODUCT NAME: 4-IMIDAZOPYRIDAZIN-1-YL-BENZAMIDE UND 4-IMIDAZOTRIAZIN-1-YL-BENZAMIDE ALS BTK-INHIBITOREN; REGISTRATION NO/DATE: EU/1/20/1479 (MITTEILUNG) 20201106 |
| 2734522 | CA 2021 00007 | Denmark | ⤷ Sign Up | PRODUCT NAME: ACALABRUTINIB ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/20/1479 20201106 |
| 2734522 | 301097 | Netherlands | ⤷ Sign Up | PRODUCT NAME: ACALABRUTINIB OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; REGISTRATION NO/DATE: EU/1/20/1479 20201106 |
| 2734522 | C202130014 | Spain | ⤷ Sign Up | PRODUCT NAME: ACALABRUTINIB O UNA SAL FARMACEUTICAMENTE ACEPTABLE DEL MISMO; NATIONAL AUTHORISATION NUMBER: EU/1/20/1479; DATE OF AUTHORISATION: 20201105; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/20/1479; DATE OF FIRST AUTHORISATION IN EEA: 20201105 |
| 2734522 | 21C1014 | France | ⤷ Sign Up | PRODUCT NAME: ACALABRUTINIB OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI; REGISTRATION NO/DATE: EU/1/20/1479 20201106 |
| 2734522 | C20210006 00368 | Estonia | ⤷ Sign Up | PRODUCT NAME: AKALABRUTINIIB;REG NO/DATE: EU/1/20/1479 06.11.2020 |
| 2734522 | 2021C/512 | Belgium | ⤷ Sign Up | PRODUCT NAME: ACALABRUTINIB OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI; AUTHORISATION NUMBER AND DATE: EU/1/20/1479 20201106 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


