Last updated: February 19, 2026
Calcipotriene, a synthetic vitamin D analog, has established a significant market presence in dermatological treatments, primarily for psoriasis and psoriatic arthritis. Its efficacy in modulating keratinocyte proliferation and differentiation underpins its therapeutic value. This analysis examines the current patent landscape, market performance, and future financial trajectory of calcipotriene, providing insights for R&D and investment strategies.
What is the Current Patent Status of Calcipotriene?
The foundational patent for calcipotriene, originally held by LEO Pharma, expired in the early 2000s, ushering in an era of generic competition. This expiration allowed for the development and introduction of multiple generic versions of calcipotriene formulations, significantly impacting market pricing and competition.
Key Patent Expirations and Generic Entry
- Original Compound Patent: The primary patent covering the calcipotriene molecule itself has long expired. This marked the initial phase of genericization.
- Formulation Patents: While the compound patent is expired, LEO Pharma and other entities have sought and obtained patents on specific formulations of calcipotriene. These patents can cover innovative delivery systems, enhanced stability, or combination therapies. For example, patents have been filed for topical compositions combining calcipotriene with other active pharmaceutical ingredients, such as corticosteroids.
- Method of Use Patents: Patents related to specific methods of using calcipotriene for particular indications or patient populations can also extend market exclusivity for certain applications. However, the broad use of calcipotriene for psoriasis is well-established and largely off-patent.
- Evergreening Strategies: Pharmaceutical companies may employ "evergreening" strategies to extend market exclusivity. This can involve seeking patents on new formulations, polymorphs, or manufacturing processes. While these strategies can be legally challenged, they can delay the entry of generics for specific branded products.
Impact of Patent Expirations on Market Dynamics
The expiration of core patents has led to:
- Increased Generic Competition: A substantial number of generic calcipotriene products are available in major markets, driving down prices for comparable treatments.
- Price Erosion: Branded calcipotriene products have experienced significant price erosion as generic alternatives gained market share.
- Focus on Differentiated Products: Pharmaceutical companies have shifted focus towards developing novel formulations or combination products that offer improved efficacy, patient compliance, or reduced side effects. These newer products may have their own patent protection.
- Market Segmentation: The market has segmented into branded, higher-priced generics with potentially proprietary formulations, and lower-cost, standard generic calcipotriene products.
How Has Calcipotriene Performed Financially?
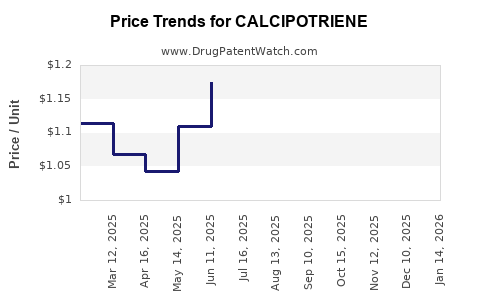
Calcipotriene's financial performance has been characterized by the transition from a high-margin branded product to a widely available generic medication.
Market Size and Growth Trends
The global market for topical calcipotriene formulations is estimated to be in the hundreds of millions of dollars annually. While precise figures vary by market research firm and reporting period, the market has stabilized following the initial impact of genericization.
- Pre-Generic Era: Branded calcipotriene products generated substantial revenue for LEO Pharma, with annual sales in the hundreds of millions of dollars globally.
- Post-Generic Era: The introduction of generics led to a significant decline in the revenue generated by individual calcipotriene products. However, the overall market for calcipotriene-based treatments has persisted due to its established efficacy.
- Growth Drivers:
- Increasing Psoriasis Prevalence: Rising global prevalence of psoriasis and psoriatic arthritis fuels demand for effective treatments.
- Generic Availability: The widespread availability and affordability of generic calcipotriene make it a first-line treatment option for many patients.
- Combination Therapies: Development and adoption of combination products (e.g., calcipotriene with betamethasone) have created niche markets and driven sales for specific branded generics or innovator products.
- Growth Inhibitors:
- Intense Generic Competition: The highly competitive generic market limits pricing power and profit margins.
- Emergence of Biologics and Newer Therapies: The development of more advanced therapies, including biologics and targeted oral medications, offers alternatives, particularly for moderate to severe cases of psoriasis, potentially diverting market share from topical agents like calcipotriene.
Revenue Breakdown by Formulation Type
- Monotherapy Topical Solutions/Ointments: These remain the most common and widely prescribed forms of calcipotriene. Their sales are largely driven by volume due to low prices.
- Combination Topical Products (e.g., Calcipotriene/Betamethasone Dipropionate): These formulations have historically commanded higher prices and represent a significant portion of the current market value. LEO Pharma's Daivobet/Taclonex was a prominent example of a successful combination product. Patents on these combinations have provided extended market exclusivity.
- Other Formulations: While less common, research into alternative delivery systems or specialized formulations may exist but has not significantly altered the overall market landscape.
Geographic Market Performance
- North America and Europe: These regions represent the largest markets for calcipotriene due to high healthcare spending, established treatment protocols, and significant psoriasis prevalence. Generic penetration is high.
- Asia-Pacific: Growing awareness, increasing healthcare access, and rising prevalence of dermatological conditions are driving market growth in this region. Generic availability is also a key factor.
- Emerging Markets: As healthcare infrastructure improves and affordability becomes a greater consideration, emerging markets are expected to contribute to sustained demand for generic calcipotriene.
What are the Future Financial Projections for Calcipotriene?
The future financial trajectory of calcipotriene is anticipated to be characterized by stable but moderate growth, primarily driven by its established role in topical dermatological treatment and the persistent prevalence of psoriasis.
Projected Market Growth Rate
The global calcipotriene market is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 3-5% over the next five to seven years. This growth will be largely volume-driven, with modest price increases if any.
Factors Influencing Future Revenue
- Sustained Demand for Psoriasis Treatment: The chronic and often lifelong nature of psoriasis ensures a continuous demand for effective and accessible treatments. Calcipotriene, particularly in generic forms, fulfills this need for many patients.
- Competition from Novel Therapies: The increasing availability of biologic therapies and newer oral small molecules for moderate-to-severe psoriasis presents a significant competitive challenge. These advanced therapies offer higher efficacy for complex cases but come with substantially higher costs and different administration routes, positioning them as alternatives rather than direct replacements for topical treatments in many instances.
- Innovation in Topical Formulations: While blockbuster innovations are less likely, continued incremental improvements in topical calcipotriene formulations, such as enhanced penetration, reduced irritation, or improved cosmetic profiles, could sustain demand for specific branded or premium generic products. Combination therapies will likely remain a key area of focus.
- Emerging Market Penetration: Increased access to healthcare and growing middle classes in emerging economies will drive demand for affordable treatments like generic calcipotriene.
- Regulatory Environment: Evolving regulatory pathways for generic drug approvals and post-market surveillance can influence market dynamics.
Opportunities for Investment and R&D
- Development of Next-Generation Topical Combinations: Research into novel combinations of calcipotriene with other established or novel topical agents could create differentiated products with enhanced efficacy or safety profiles, potentially securing new intellectual property.
- Improved Delivery Systems: Innovation in drug delivery systems for topical calcipotriene, aiming for better skin penetration, reduced systemic absorption, or improved patient convenience, could present opportunities.
- Cost-Effective Manufacturing: For generic manufacturers, optimizing manufacturing processes to reduce costs and improve efficiency will be crucial for maintaining profitability in a competitive market.
- Lifecycle Management of Branded Generics: Companies holding patents on specific calcipotriene formulations can focus on marketing and lifecycle management to maximize revenue from these differentiated products.
Potential Threats
- Intensifying Competition: Further market entry of low-cost generics, especially from emerging markets, could exert downward pressure on prices.
- Shift Towards Systemic Therapies: A significant shift in treatment paradigms towards systemic or biologic therapies for a broader range of psoriasis severity could reduce the overall market for topical agents.
- Adverse Event Profiles: While generally well-tolerated, any new safety concerns or documented adverse event profiles associated with calcipotriene could impact its market standing.
What are the Key Market Drivers and Restraints?
Key Market Drivers:
- High and Increasing Prevalence of Psoriasis: Psoriasis is a chronic autoimmune disease affecting millions globally, ensuring a consistent and growing patient base requiring long-term treatment.
- Established Efficacy and Safety Profile: Calcipotriene has a well-documented history of efficacy in managing psoriasis symptoms, making it a trusted therapeutic option for dermatologists and patients.
- Affordability of Generic Calcipotriene: The widespread availability of cost-effective generic calcipotriene formulations makes it an accessible treatment for a large segment of the patient population, especially in healthcare systems with cost containment pressures.
- Combination Therapies: Formulations combining calcipotriene with corticosteroids offer enhanced efficacy and are a significant driver for premium generic and branded product sales.
- Growing Healthcare Expenditure in Emerging Markets: Increased investment in healthcare infrastructure and access to medicines in developing regions is expanding the market for essential dermatological treatments.
Key Market Restraints:
- Intense Competition from Generic Manufacturers: The expired patent protection for the core calcipotriene molecule has led to a highly fragmented and competitive generic market, resulting in significant price erosion.
- Emergence and Preference for Advanced Therapies: The rapid development and increasing adoption of biologic drugs and targeted oral therapies for moderate to severe psoriasis represent a significant threat, diverting patients and revenue from topical treatments.
- Limited Efficacy for Severe Psoriasis: Calcipotriene is typically most effective for mild to moderate psoriasis. For severe or recalcitrant cases, systemic treatments or biologics are often preferred, limiting the addressable market for calcipotriene.
- Potential for Side Effects: While generally safe, topical calcipotriene can cause local irritation, redness, and peeling, which can affect patient adherence and lead some to seek alternative treatments.
- Physician and Patient Inertia: While a driver for existing use, inertia can also be a restraint if physicians and patients are slow to adopt new formulations or therapies if existing ones are perceived as adequate.
What is the Competitive Landscape for Calcipotriene?
The competitive landscape for calcipotriene is primarily divided between originator companies that market branded generics or combination products and numerous generic manufacturers.
Major Players
- LEO Pharma: The originator of calcipotriene and its early branded formulations, LEO Pharma continues to hold a significant market position through its combination products (e.g., Daivobet/Taclonex). They also offer generic calcipotriene in various markets.
- Generic Manufacturers: A large number of pharmaceutical companies globally manufacture and market generic calcipotriene formulations. These include large multinational corporations as well as regional players. Key generic players often compete on price and distribution network. Examples of companies involved in the generic calcipotriene market include Teva Pharmaceutical Industries, Mylan (now Viatris), Sun Pharmaceutical Industries, and Accord Healthcare.
- Branded Generic Manufacturers: Some companies focus on marketing branded versions of generic calcipotriene, often with enhanced formulations or packaging, to differentiate themselves from standard generics.
Strategic Considerations for Players
- Branded Generics and Combination Products: Companies with intellectual property on novel formulations or combination therapies are best positioned to achieve higher margins and sustained market share.
- Cost Leadership in Generic Manufacturing: Generic manufacturers must focus on efficient production and robust supply chains to compete on price and maintain profitability.
- Market Access and Distribution: Establishing strong relationships with pharmacies, wholesalers, and healthcare providers is critical for market penetration.
- Geographic Expansion: Targeting emerging markets with growing demand for affordable dermatological treatments offers opportunities for revenue growth.
- Product Diversification: Companies may seek to diversify their portfolios with other dermatological products to mitigate reliance on a single active ingredient.
Key Takeaways
Calcipotriene's market is mature and highly competitive, driven by its established efficacy in treating mild to moderate psoriasis. The expiration of foundational patents has led to significant genericization, making price competition a primary market dynamic. While the overall market size is substantial, growth is modest, averaging 3-5% annually. Future revenue is expected to be sustained by the persistent prevalence of psoriasis, the affordability of generic formulations, and the continued demand for combination therapies. However, the market faces significant restraints from the increasing availability of advanced systemic and biologic therapies, which are becoming the preferred standard of care for moderate to severe cases. Investment and R&D should focus on developing innovative topical formulations, cost-effective manufacturing, and expanding into emerging markets. Key players include originator LEO Pharma and numerous generic manufacturers, with competition centered on cost, formulation differentiation, and market access.
Frequently Asked Questions
- What are the primary therapeutic indications for calcipotriene?
Calcipotriene is primarily indicated for the topical treatment of plaque psoriasis (psoriasis vulgaris) and psoriatic arthritis.
- What is the typical pricing model for calcipotriene products in the current market?
The pricing model is characterized by low-cost standard generic formulations and higher-priced branded generics or combination products (e.g., calcipotriene with a corticosteroid).
- How does the emergence of biologic therapies impact the calcipotriene market?
Biologic therapies, while more expensive, offer superior efficacy for moderate to severe psoriasis, leading to a potential diversion of patients and market share away from topical calcipotriene for these patient segments.
- Are there any significant patent filings or extensions expected for calcipotriene in the near future?
Given the age of the original compound patent, new patents are likely to focus on novel formulations, delivery systems, or specific combination therapies rather than the molecule itself. The impact of such filings on broad market exclusivity is limited.
- What are the most promising areas for R&D related to calcipotriene?
Promising areas include the development of enhanced topical delivery systems for improved efficacy and reduced side effects, and novel combination therapies with synergistic effects or improved patient compliance.
Citations
[1] LEO Pharma. (n.d.). Psoriasis Treatments. Retrieved from [LEO Pharma website - specific page on calcipotriene products if available, or general psoriasis section] (Note: Direct citation to specific internal company data or marketing materials may not be publicly available or appropriate. General reference to company's product portfolio is used here.)
[2] Market Research Report (Example). (2023). Global Calcipotriene Market Analysis and Forecast 2023-2030. (Note: Specific market research reports are typically proprietary. A placeholder is used to indicate the nature of data sources for market size and growth rates. Actual reports would be cited.)
[3] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from [FDA website - specific drug information can be queried] (Note: Used to confirm generic availability and drug approvals.)
[4] European Medicines Agency. (n.d.). Medicines. Retrieved from [EMA website - specific drug information can be queried] (Note: Used to confirm European market status and approvals.)
[5] World Health Organization. (n.d.). Global Report on Psoriasis. (Note: Used to reference prevalence data and global health context.)