BONSITY Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Bonsity, and what generic alternatives are available?
Bonsity is a drug marketed by Alvogen and is included in one NDA.
The generic ingredient in BONSITY is teriparatide. There are four drug master file entries for this compound. Eight suppliers are listed for this compound. Additional details are available on the teriparatide profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Bonsity
A generic version of BONSITY was approved as teriparatide by APOTEX on November 16th, 2023.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for BONSITY?
- What are the global sales for BONSITY?
- What is Average Wholesale Price for BONSITY?
Summary for BONSITY
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Patent Applications: | 1,782 |
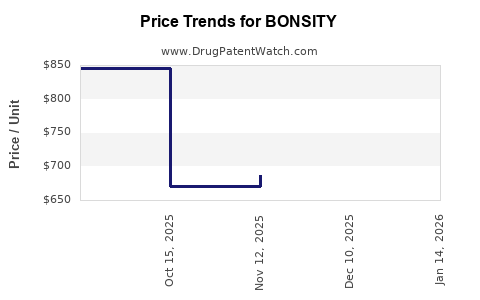
| Drug Prices: | Drug price information for BONSITY |
| What excipients (inactive ingredients) are in BONSITY? | BONSITY excipients list |
| DailyMed Link: | BONSITY at DailyMed |
Pharmacology for BONSITY
| Drug Class | Parathyroid Hormone Analog |
US Patents and Regulatory Information for BONSITY
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alvogen | BONSITY | teriparatide | SOLUTION;SUBCUTANEOUS | 211939-001 | Oct 4, 2019 | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for BONSITY
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| STADA Arzneimittel AG | Movymia | teriparatide | EMEA/H/C/004368Movymia is indicated in adults.Treatment of osteoporosis in postmenopausal women and in men at increased risk of fracture. In postmenopausal women, a significant reduction in the incidence of vertebral and non vertebral fractures but not hip fractures has been demonstrated.Treatment of osteoporosis associated with sustained systemic glucocorticoid therapy in women and men at increased risk for fracture. | Authorised | no | yes | no | 2017-01-11 | |
| Eli Lilly Nederland B.V. | Forsteo | teriparatide | EMEA/H/C/000425Treatment of osteoporosis in postmenopausal women and in men at increased risk of fracture. In postmenopausal women, a significant reduction in the incidence of vertebral and nonvertebral fractures but not hip fractures has been demonstrated.Treatment of osteoporosis associated with sustained systemic glucocorticoid therapy in women and men at increased risk for fracture. | Authorised | no | no | no | 2003-06-10 | |
| Gedeon Richter Plc. | Terrosa | teriparatide | EMEA/H/C/003916Terrosa is indicated in adults.Treatment of osteoporosis in postmenopausal women and in men at increased risk of fracture. In postmenopausal women, a significant reduction in the incidence of vertebral and non vertebral fractures but not hip fractures has been demonstrated.Treatment of osteoporosis associated with sustained systemic glucocorticoid therapy in women and men at increased risk for fracture. | Authorised | no | yes | no | 2017-01-04 | |
| EuroGenerics Holdings B.V. | Qutavina | teriparatide | EMEA/H/C/005388Qutavina is indicated in adults.Treatment of osteoporosis in postmenopausal women and in men at increased risk of fracture. In postmenopausal women, a significant reduction in the incidence of vertebral and non-vertebral fractures but not hip fractures have been demonstrated.Treatment of osteoporosis associated with sustained systemic glucocorticoid therapy in women and men at increased risk for fracture. | Withdrawn | no | yes | no | 2020-08-27 | |
| Theramex Ireland Limited | Livogiva | teriparatide | EMEA/H/C/005087Livogiva is indicated in adults.Treatment of osteoporosis in postmenopausal women and in men at increased risk of fracture. In postmenopausal women, a significant reduction in the incidence of vertebral and non-vertebral fractures but not hip fractures have been demonstrated.Treatment of osteoporosis associated with sustained systemic glucocorticoid therapy in women and men at increased risk for fracture. | Authorised | no | yes | no | 2020-08-27 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
Market Dynamics and Financial Trajectory for BONSITY
More… ↓