Last updated: February 26, 2026
What is the Current Market Size and Growth Rate?
Teriparatide (brand name Forteo) is an anabolic agent approved for osteoporosis treatment in postmenopausal women and men at high fracture risk. The global osteoporosis therapeutics market, including Teriparatide, was valued at approximately $10 billion in 2022. It is projected to grow at a compounded annual growth rate (CAGR) of 5.2% between 2023 and 2030, reaching an estimated $16.2 billion.
In 2022, Teriparatide accounted for roughly 8% of the osteoporosis segment revenue, driven by its targeted efficacy in high-risk populations. North America led sales, comprising 60% of revenue, supported by high awareness, reimbursement coverage, and advanced healthcare infrastructure.
How Does the Competitive Landscape Affect Market Dynamics?
Key competitors include Abaloparatide (Tymlos), Romosozumab (Evenity), and alternative osteoporosis therapies like bisphosphonates. The competitive advantage of Teriparatide remains its anabolic mechanism, providing benefits in severe cases resistant to anti-resorptive agents.
However, new entrants and biosimilars threaten its market share. The patent for Forteo expired in the US in 2022, opening opportunities for biosimilar versions, which may reduce prices by 30-50%, impacting revenue streams.
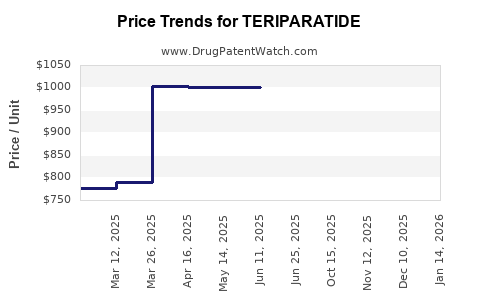
What Are the Pricing and Reimbursement Trends?
In the US, Teriparatide’s list price exceeds $4,000 per vial. Reimbursement policies, favoring high-cost therapies in high-risk patients, support its sales, though rebates and negotiations influence net pricing.
European markets exhibit variable reimbursement policies, with some countries imposing cost-effectiveness thresholds, affecting uptake. Emerging markets often face affordability challenges, restraining market expansion.
What Regulatory Developments Influence the Market?
The FDA approved a once-weekly subcutaneous formulation of Teriparatide in 2022, aiming to improve patient adherence. Regulatory pathways favor indicating expanded uses, such as severe osteoporosis with comorbidities or fracture healing, potentially broadening market scope.
In 2021, the EMA authorized generic or biosimilar versions, increasing competition. Elsewhere, regulatory approvals in China, Japan, and India expand the drug’s global footprint, though access depends on local pricing policies.
What Are the Financial Projections?
Analysts forecast a steady revenue decline for proprietary Teriparatide post-patent expiry, estimating a 20-30% decrease in US sales by 2025. Biosimilar entry could accelerate this decline.
Long-term, revenue could stabilize as biosimilar versions capture 60-70% of the market, with price erosion accounting for reduced per-unit revenue. Conversely, pipeline advancements or expanded indications could offset declines.
How Do R&D Pipelines and Pipeline Products Impact Future Market Trajectory?
Research focuses on oral formulations, combination therapies, and extended-use indications. For instance, a phase 3 trial for an oral PTH analog aims to improve administration compliance. Positive trial results could catalyze future sales.
Several biosimilar candidates aim to accelerate market share reduction, with some anticipated to launch by 2024, depending on regulatory timelines.
Summary of Market Factors
| Factor |
Impact |
Timeline |
| Patent expiry of Forteo |
Revenue decline, biosimilar entry |
2022-2024 |
| Biosimilar market entry |
Price erosion, volume increase |
2023-2025 |
| New formulations & indications |
Potential revenue growth, market expansion |
2023-2026 |
| Regulatory approvals |
Broader patient access and uptake |
2022-2027 |
Key Takeaways
- The global osteoporosis drug market is projected to grow at 5.2% CAGR through 2030.
- Post-patent, biosimilars will drive pricing pressure, likely reducing Forteo revenue by 20-30% in the US.
- New formulations and expanded indications could offset market declines.
- Reimbursement policies heavily influence regional sales strength.
- Competitive innovations and pipeline products will shape the upcoming market landscape.
FAQs
1. How will biosimilar entry affect Teriparatide’s revenue?
Biosimilars could reduce proprietary drug sales by up to 50%, depending on market penetration and pricing strategies.
2. Are there opportunities in emerging markets?
Yes; China, India, and Southeast Asia present growth avenues, provided pricing and access barriers are managed.
3. What is the impact of new formulations?
Once-weekly injections and oral options could improve patient adherence and increase market penetration.
4. What are the key regulatory considerations?
Approval of biosimilars and new indications influence competitive dynamics and potential revenue.
5. How does the market for osteoporosis drugs evolve with aging populations?
An aging global population sustains demand; innovations remain critical to market share and growth.
References:
- Allied Market Research. (2022). Osteoporosis therapeutics market. https://www.alliedmarketresearch.com/osteoporosis-therapeutics-market
- MarketsandMarkets. (2023). Osteoporosis drugs market. https://www.marketsandmarkets.com
- U.S. Food and Drug Administration. (2022). FDA approves weekly Teriparatide formulation. https://www.fda.gov
- European Medicines Agency. (2021). Biosimilar approvals for osteoporosis drugs. https://www.ema.europa.eu
- IQVIA Institute. (2022). Global Outlook for Osteoporosis Treatment. https://www.iqvia.com