Last updated: February 19, 2026
Bisoprolol fumarate, a selective beta-1 adrenergic receptor blocker, has established a significant market presence in the treatment of cardiovascular conditions. Its efficacy in managing hypertension, angina pectoris, and heart failure has driven consistent demand. The market for bisoprolol fumarate is characterized by a mature generic landscape, with numerous manufacturers competing on price and availability. Patent expiries have led to widespread generic availability, contributing to its affordability and broad accessibility.
What are the Primary Indications for Bisoprolol Fumarate?
Bisoprolol fumarate is primarily prescribed for the management of several key cardiovascular diseases:
- Hypertension: It is a first-line treatment for lowering blood pressure.
- Angina Pectoris: It reduces the frequency and severity of chest pain associated with coronary artery disease.
- Chronic Heart Failure: In stable, symptomatic chronic heart failure, it is used to improve survival and reduce hospitalizations.
The drug's pharmacological profile, characterized by its cardioselectivity, minimizes effects on beta-2 receptors, thereby reducing the risk of bronchospasm in patients with respiratory conditions, a critical differentiator.
How Have Patent Expiries Impacted the Bisoprolol Fumarate Market?
The patent landscape for bisoprolol fumarate has significantly shaped its market trajectory. The original patents for bisoprolol fumarate have long expired, allowing for the development and marketing of generic versions.
- Original Compound Patent Expiry: The foundational patent for bisoprolol fumarate expired decades ago.
- Formulation Patents: Subsequent patents related to specific formulations, such as extended-release versions, have also expired or are nearing expiry.
- Manufacturing Process Patents: Patents covering specific manufacturing processes are typically shorter-lived and have also expired, enabling broader competition.
This widespread patent expiry has led to:
- Increased Competition: The market is populated by numerous generic manufacturers, including both large pharmaceutical companies and smaller specialized generic firms.
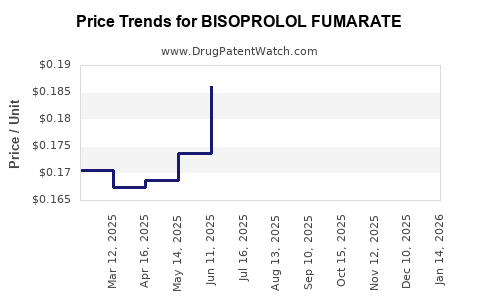
- Price Erosion: Intense competition has resulted in significant price reductions for bisoprolol fumarate products.
- Enhanced Accessibility: Lower prices have made bisoprolol fumarate more accessible to a wider patient population globally.
- Focus on Brand Differentiation (Limited): While most market share is held by generics, some branded versions may exist with a focus on specific delivery systems or co-formulations, though these represent a smaller segment.
What are the Key Market Segments for Bisoprolol Fumarate?
The market for bisoprolol fumarate can be segmented by indication, formulation, and end-user.
By Indication:
- Hypertension Management
- Angina Pectoris Treatment
- Chronic Heart Failure Therapy
By Formulation:
- Immediate-Release Tablets
- Extended-Release Tablets
By End-User:
- Hospitals
- Clinics
- Retail Pharmacies
- Homecare Settings
The hypertension segment constitutes the largest portion of the market due to the high prevalence of this condition. However, the increasing diagnosis and management of chronic heart failure are contributing to growth in that segment.
Who are the Major Manufacturers of Bisoprolol Fumarate?
The bisoprolol fumarate market is characterized by the presence of several global pharmaceutical players, primarily in the generic drug manufacturing space. Key manufacturers include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.: A leading global generic pharmaceutical company.
- Mylan N.V. (now Viatris Inc.): Another major player in the generic and specialty pharmaceuticals market.
- Sandoz (a Novartis company): A significant global producer of generic pharmaceuticals.
- Lupin Limited: An Indian multinational pharmaceutical company with a strong generic portfolio.
- Dr. Reddy's Laboratories Ltd.: An Indian multinational pharmaceutical company with a broad range of generic products.
- Apotex Inc.: A Canadian pharmaceutical company focused on generic and biosimilar drugs.
- Sun Pharmaceutical Industries Ltd.: An Indian multinational pharmaceutical company, one of the largest globally.
These companies compete through efficient manufacturing, established distribution networks, and competitive pricing strategies.
What is the Global Market Size and Growth Projection for Bisoprolol Fumarate?
Estimating the precise global market size for a single generic drug like bisoprolol fumarate is challenging due to fragmented reporting and the prevalence of generic competition. However, industry analysis indicates a substantial and stable market.
| Metric |
Value (USD Billion) |
Compound Annual Growth Rate (CAGR) |
Notes |
| Current Market Size |
1.5 - 2.0 |
2.5% - 3.5% |
Estimated for the global bisoprolol fumarate market, encompassing all indications. |
| Projected Market Size (2028) |
1.7 - 2.3 |
|
Forecast based on current trends and growth drivers. |
Market Drivers:
- Increasing Prevalence of Cardiovascular Diseases: Rising rates of hypertension, heart disease, and heart failure globally.
- Aging Global Population: Older demographics are more susceptible to cardiovascular conditions, driving demand for treatments.
- Affordability and Accessibility: The availability of generic bisoprolol fumarate at lower costs promotes its widespread use.
- Physician Prescribing Habits: Established treatment guidelines and physician familiarity with bisoprolol fumarate.
- Government Healthcare Initiatives: Programs focused on managing chronic diseases and expanding access to essential medicines.
Market Restraints:
- Intense Generic Competition: Price wars among manufacturers limit revenue growth potential.
- Emergence of New Therapies: Development of novel drugs for cardiovascular conditions could potentially displace some market share, though bisoprolol fumarate's established efficacy and cost-effectiveness offer a strong defense.
- Strict Regulatory Requirements: Compliance with stringent quality and manufacturing standards can be a barrier.
What are the Key Regulatory Considerations for Bisoprolol Fumarate?
The manufacturing and sale of bisoprolol fumarate are subject to rigorous regulatory oversight by health authorities worldwide.
- Active Pharmaceutical Ingredient (API) Quality: Manufacturers must adhere to Good Manufacturing Practices (GMP) for API production.
- Finished Dosage Form Manufacturing: Regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others mandate strict GMP compliance for finished product manufacturing.
- Abbreviated New Drug Applications (ANDAs) / Marketing Authorization Applications (MAAs): Generic manufacturers must demonstrate bioequivalence to the reference listed drug (RLD) through ANDA submissions in the U.S. and similar MAA processes in other regions.
- Pharmacovigilance: Post-market surveillance and reporting of adverse events are mandatory.
- Labeling Requirements: Specific labeling information regarding indications, dosage, contraindications, warnings, and precautions must be maintained.
- Quality Standards: Adherence to pharmacopeial standards (e.g., USP, EP, JP) is essential.
What is the Financial Trajectory and Profitability Landscape?
The financial trajectory of bisoprolol fumarate is primarily influenced by its status as a high-volume, low-margin generic product.
- Revenue Generation: While individual product margins are slim due to intense price competition, the sheer volume of prescriptions ensures consistent revenue for established generic manufacturers.
- Cost Management: Profitability hinges on highly efficient manufacturing processes, optimized supply chains, and economies of scale. Companies with integrated API manufacturing capabilities often have a cost advantage.
- Market Share Dynamics: Manufacturers that can secure significant market share through competitive bidding for tenders, robust distribution, and consistent product availability tend to perform better.
- Productivity: High manufacturing throughput and minimal waste are critical for maintaining profitability.
- Investment Focus: Investment for companies in this space often shifts towards developing new generic products, expanding manufacturing capacity, or optimizing existing processes rather than extensive R&D into novel formulations of established generics.
- Strategic Partnerships: Collaborations with wholesalers, pharmacy benefit managers (PBMs), and hospital systems are crucial for market access and sales volume.
The financial outlook is characterized by stability rather than explosive growth. Companies that can maintain operational efficiency and secure consistent sales volumes are poised for steady performance.
What are the Emerging Trends and Future Outlook for Bisoprolol Fumarate?
The future of bisoprolol fumarate is largely predictable, anchored by its established role in cardiovascular care.
- Continued Generic Dominance: The generic market will remain the primary driver, with ongoing competition among manufacturers.
- Focus on Emerging Markets: Growth potential exists in emerging economies where access to essential medicines is expanding and the prevalence of cardiovascular diseases is rising.
- Combination Therapies: While bisoprolol fumarate is often used as a monotherapy, its integration into fixed-dose combination products with other antihypertensive agents or cardiovascular drugs may see continued development, provided patent protection for novel combinations is sought.
- Supply Chain Resilience: Recent global events have highlighted the importance of resilient and diversified supply chains. Manufacturers will likely focus on securing API sources and ensuring stable production.
- Digital Health Integration: While not directly related to the drug itself, the broader healthcare ecosystem's move towards digital health platforms could influence prescribing patterns and patient monitoring for conditions treated by bisoprolol fumarate.
- Sustainability in Manufacturing: Growing emphasis on environmentally sustainable manufacturing practices could influence production methods and investment decisions.
The long-term outlook for bisoprolol fumarate is one of continued relevance and stable demand, driven by its essential role in managing prevalent chronic diseases, particularly within the generic pharmaceutical market.
Key Takeaways
- Bisoprolol fumarate remains a critical medication for hypertension, angina, and heart failure, with a mature generic market.
- Patent expiries have resulted in intense price competition among numerous manufacturers, enhancing global accessibility.
- The global market is estimated to be between $1.5 billion and $2.0 billion, with projected modest growth of 2.5% to 3.5% CAGR.
- Major generic pharmaceutical companies dominate the manufacturing landscape, prioritizing cost efficiency and large-scale production.
- Regulatory compliance, particularly GMP and bioequivalence demonstration, is paramount for market entry and sustained operations.
- Profitability is driven by high sales volumes and stringent cost management, rather than high per-unit margins.
- The future outlook is characterized by continued generic market strength, with growth opportunities in emerging economies and a focus on supply chain resilience.
Frequently Asked Questions
-
What is the current patent status of bisoprolol fumarate, and does this affect market entry for new players?
The original patents for bisoprolol fumarate itself have long expired. New players can enter the market by obtaining regulatory approval for generic versions, typically through Abbreviated New Drug Applications (ANDAs) in the U.S. or equivalent processes elsewhere, demonstrating bioequivalence to an approved reference product. Manufacturing process patents may exist, but these are often less restrictive for generic manufacturers.
-
How does the cardioselectivity of bisoprolol fumarate differentiate it from other beta-blockers in the market?
Bisoprolol fumarate is a selective beta-1 adrenergic receptor blocker. This means it primarily targets the beta-1 receptors in the heart, leading to a reduction in heart rate and contractility. Unlike non-selective beta-blockers, it has a lower affinity for beta-2 receptors found in the lungs and peripheral blood vessels. This selectivity reduces the risk of adverse effects such as bronchoconstriction in patients with asthma or COPD, making it a preferred choice for certain patient populations.
-
What are the primary economic factors contributing to the price erosion of bisoprolol fumarate?
The primary economic factor is the high level of competition within the generic pharmaceutical market. Once patents expire, multiple companies can produce and market generic versions of the drug. This leads to price wars as manufacturers compete to capture market share, often through aggressive tendering processes for large contracts with pharmacies, wholesalers, and healthcare systems. Economies of scale in manufacturing and efficient supply chain management also enable lower pricing.
-
Are there any significant pipeline developments or new formulations of bisoprolol fumarate expected to impact the market in the near future?
Given its mature status as a generic drug, significant pipeline developments for novel formulations of bisoprolol fumarate are unlikely. While companies may continue to optimize manufacturing processes or develop new fixed-dose combinations with other cardiovascular agents, the focus is generally on maintaining cost-effective production of existing formulations rather than on groundbreaking R&D for bisoprolol fumarate itself. The market dynamics are driven more by volume and efficiency than by innovative product launches.
-
What is the typical profit margin for bisoprolol fumarate products for generic manufacturers, and how do they achieve profitability?
Typical profit margins for individual bisoprolol fumarate products for generic manufacturers are relatively low, often in the single digits. Profitability is achieved through very high sales volumes and stringent cost control. Manufacturers focus on optimizing manufacturing processes to reduce production costs per unit, leveraging economies of scale, securing favorable raw material pricing, and maintaining efficient, low-overhead distribution networks. Long-term contracts and consistent supply agreements with major buyers are crucial for sustained profitability.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/approved-drug-products-therapeutic-equivalence-evaluations-orange-book
[2] European Medicines Agency. (n.d.). Medicines. Retrieved from https://www.ema.europa.eu/en/medicines
[3] Various Market Research Reports on Cardiovascular Drugs and Generic Pharmaceuticals. (Specific reports are proprietary and vary by publisher; general industry analysis indicates market size and growth trends).
[4] World Health Organization. (n.d.). Cardiovascular diseases (CVDs). Retrieved from https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)