BEVESPI AEROSPHERE Drug Patent Profile
✉ Email this page to a colleague
When do Bevespi Aerosphere patents expire, and what generic alternatives are available?
Bevespi Aerosphere is a drug marketed by Astrazeneca and is included in one NDA. There are seven patents protecting this drug.
This drug has one hundred and ninety-two patent family members in thirty-two countries.
The generic ingredient in BEVESPI AEROSPHERE is formoterol fumarate; glycopyrrolate. There are nineteen drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the formoterol fumarate; glycopyrrolate profile page.
DrugPatentWatch® Generic Entry Outlook for Bevespi Aerosphere
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be May 28, 2030. This may change due to patent challenges or generic licensing.
There has been one patent litigation case involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for BEVESPI AEROSPHERE?
- What are the global sales for BEVESPI AEROSPHERE?
- What is Average Wholesale Price for BEVESPI AEROSPHERE?
Summary for BEVESPI AEROSPHERE
| International Patents: | 192 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 2 |
| Patent Applications: | 13 |
| Drug Prices: | Drug price information for BEVESPI AEROSPHERE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for BEVESPI AEROSPHERE |
| What excipients (inactive ingredients) are in BEVESPI AEROSPHERE? | BEVESPI AEROSPHERE excipients list |
| DailyMed Link: | BEVESPI AEROSPHERE at DailyMed |
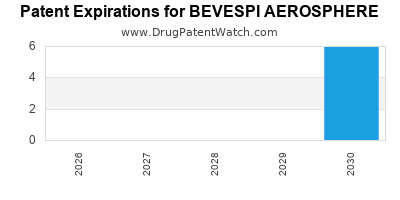
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for BEVESPI AEROSPHERE
Generic Entry Date for BEVESPI AEROSPHERE*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
AEROSOL, METERED;INHALATION |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for BEVESPI AEROSPHERE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Bastiaan Driehuys | Phase 2/Phase 3 |
| Lundquist Institute for Biomedical Innovation at Harbor-UCLA Medical Center | Phase 4 |
| Los Angeles Biomedical Research Institute | Phase 4 |
Pharmacology for BEVESPI AEROSPHERE
US Patents and Regulatory Information for BEVESPI AEROSPHERE
BEVESPI AEROSPHERE is protected by seven US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of BEVESPI AEROSPHERE is ⤷ Start Trial.
This potential generic entry date is based on patent 8,324,266.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astrazeneca | BEVESPI AEROSPHERE | formoterol fumarate; glycopyrrolate | AEROSOL, METERED;INHALATION | 208294-001 | Apr 25, 2016 | RX | Yes | Yes | 8,324,266 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Astrazeneca | BEVESPI AEROSPHERE | formoterol fumarate; glycopyrrolate | AEROSOL, METERED;INHALATION | 208294-001 | Apr 25, 2016 | RX | Yes | Yes | 8,815,258 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Astrazeneca | BEVESPI AEROSPHERE | formoterol fumarate; glycopyrrolate | AEROSOL, METERED;INHALATION | 208294-001 | Apr 25, 2016 | RX | Yes | Yes | 8,703,806 | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for BEVESPI AEROSPHERE
When does loss-of-exclusivity occur for BEVESPI AEROSPHERE?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 6621
Estimated Expiration: ⤷ Start Trial
Patent: 6806
Estimated Expiration: ⤷ Start Trial
Patent: 6807
Estimated Expiration: ⤷ Start Trial
Patent: 1758
Estimated Expiration: ⤷ Start Trial
Patent: 2477
Estimated Expiration: ⤷ Start Trial
Patent: 2478
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 10253770
Estimated Expiration: ⤷ Start Trial
Patent: 10253776
Estimated Expiration: ⤷ Start Trial
Patent: 10253950
Estimated Expiration: ⤷ Start Trial
Patent: 15201037
Estimated Expiration: ⤷ Start Trial
Patent: 17201709
Estimated Expiration: ⤷ Start Trial
Patent: 18282272
Estimated Expiration: ⤷ Start Trial
Patent: 20210160
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 1011220
Estimated Expiration: ⤷ Start Trial
Patent: 1011229
Estimated Expiration: ⤷ Start Trial
Patent: 1011508
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 63936
Estimated Expiration: ⤷ Start Trial
Patent: 63939
Estimated Expiration: ⤷ Start Trial
Patent: 63941
Estimated Expiration: ⤷ Start Trial
China
Patent: 2458364
Estimated Expiration: ⤷ Start Trial
Patent: 2596176
Estimated Expiration: ⤷ Start Trial
Patent: 2753152
Estimated Expiration: ⤷ Start Trial
Patent: 5193773
Estimated Expiration: ⤷ Start Trial
Patent: 7412212
Estimated Expiration: ⤷ Start Trial
Patent: 7669664
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0161098
Estimated Expiration: ⤷ Start Trial
Patent: 0161101
Estimated Expiration: ⤷ Start Trial
Patent: 0161102
Estimated Expiration: ⤷ Start Trial
Patent: 0200166
Estimated Expiration: ⤷ Start Trial
Patent: 0200260
Estimated Expiration: ⤷ Start Trial
Patent: 0200298
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 18030
Estimated Expiration: ⤷ Start Trial
Patent: 18034
Estimated Expiration: ⤷ Start Trial
Patent: 18040
Estimated Expiration: ⤷ Start Trial
Patent: 22732
Estimated Expiration: ⤷ Start Trial
Patent: 22749
Estimated Expiration: ⤷ Start Trial
Patent: 22807
Estimated Expiration: ⤷ Start Trial
Patent: 19031
Estimated Expiration: ⤷ Start Trial
Patent: 21012
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 35023
Estimated Expiration: ⤷ Start Trial
Patent: 35024
Estimated Expiration: ⤷ Start Trial
Patent: 35025
Estimated Expiration: ⤷ Start Trial
Patent: 06149
Estimated Expiration: ⤷ Start Trial
Patent: 11926
Estimated Expiration: ⤷ Start Trial
Patent: 11927
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 35023
Estimated Expiration: ⤷ Start Trial
Patent: 35024
Estimated Expiration: ⤷ Start Trial
Patent: 35025
Estimated Expiration: ⤷ Start Trial
Patent: 06149
Estimated Expiration: ⤷ Start Trial
Patent: 11926
Estimated Expiration: ⤷ Start Trial
Patent: 11927
Estimated Expiration: ⤷ Start Trial
France
Patent: C1040
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 69026
Estimated Expiration: ⤷ Start Trial
Patent: 69027
Estimated Expiration: ⤷ Start Trial
Patent: 69307
Estimated Expiration: ⤷ Start Trial
Patent: 18867
Estimated Expiration: ⤷ Start Trial
Patent: 44669
Estimated Expiration: ⤷ Start Trial
Patent: 47095
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 29532
Estimated Expiration: ⤷ Start Trial
Patent: 31229
Estimated Expiration: ⤷ Start Trial
Patent: 31283
Estimated Expiration: ⤷ Start Trial
Patent: 47803
Estimated Expiration: ⤷ Start Trial
Patent: 47823
Estimated Expiration: ⤷ Start Trial
Patent: 47834
Estimated Expiration: ⤷ Start Trial
Patent: 900031
Estimated Expiration: ⤷ Start Trial
Patent: 100018
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 6466
Estimated Expiration: ⤷ Start Trial
Patent: 6467
Estimated Expiration: ⤷ Start Trial
Patent: 6468
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 23383
Estimated Expiration: ⤷ Start Trial
Patent: 73012
Estimated Expiration: ⤷ Start Trial
Patent: 73013
Estimated Expiration: ⤷ Start Trial
Patent: 69639
Estimated Expiration: ⤷ Start Trial
Patent: 89356
Estimated Expiration: ⤷ Start Trial
Patent: 48645
Estimated Expiration: ⤷ Start Trial
Patent: 92124
Estimated Expiration: ⤷ Start Trial
Patent: 76734
Estimated Expiration: ⤷ Start Trial
Patent: 12528199
Estimated Expiration: ⤷ Start Trial
Patent: 12528200
Estimated Expiration: ⤷ Start Trial
Patent: 12528792
Estimated Expiration: ⤷ Start Trial
Patent: 15187108
Estimated Expiration: ⤷ Start Trial
Patent: 15199735
Estimated Expiration: ⤷ Start Trial
Patent: 16041713
Estimated Expiration: ⤷ Start Trial
Patent: 17222706
Estimated Expiration: ⤷ Start Trial
Patent: 18008942
Estimated Expiration: ⤷ Start Trial
Patent: 18048150
Estimated Expiration: ⤷ Start Trial
Patent: 19108369
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 435024
Estimated Expiration: ⤷ Start Trial
Patent: 435025
Estimated Expiration: ⤷ Start Trial
Patent: 2019014
Estimated Expiration: ⤷ Start Trial
Patent: 2021511
Estimated Expiration: ⤷ Start Trial
Patent: 35023
Estimated Expiration: ⤷ Start Trial
Patent: 35024
Estimated Expiration: ⤷ Start Trial
Patent: 35025
Estimated Expiration: ⤷ Start Trial
Patent: 06149
Estimated Expiration: ⤷ Start Trial
Patent: 11926
Estimated Expiration: ⤷ Start Trial
Patent: 11927
Estimated Expiration: ⤷ Start Trial
Luxembourg
Patent: 0124
Estimated Expiration: ⤷ Start Trial
Patent: 0208
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 7126
Estimated Expiration: ⤷ Start Trial
Patent: 0163
Estimated Expiration: ⤷ Start Trial
Patent: 0164
Estimated Expiration: ⤷ Start Trial
Patent: 7778
Estimated Expiration: ⤷ Start Trial
Patent: 3243
Estimated Expiration: ⤷ Start Trial
Patent: 11012684
Estimated Expiration: ⤷ Start Trial
Patent: 11012685
Estimated Expiration: ⤷ Start Trial
Patent: 11012783
Estimated Expiration: ⤷ Start Trial
Patent: 20004077
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 631
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 0995
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 19026
Estimated Expiration: ⤷ Start Trial
Patent: 21019
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 017500778
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 35023
Estimated Expiration: ⤷ Start Trial
Patent: 35024
Estimated Expiration: ⤷ Start Trial
Patent: 35025
Estimated Expiration: ⤷ Start Trial
Patent: 06149
Estimated Expiration: ⤷ Start Trial
Patent: 11926
Estimated Expiration: ⤷ Start Trial
Patent: 11927
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 35023
Estimated Expiration: ⤷ Start Trial
Patent: 35024
Estimated Expiration: ⤷ Start Trial
Patent: 35025
Estimated Expiration: ⤷ Start Trial
Patent: 06149
Estimated Expiration: ⤷ Start Trial
Patent: 11926
Estimated Expiration: ⤷ Start Trial
Patent: 11927
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 80315
Estimated Expiration: ⤷ Start Trial
Patent: 86297
Estimated Expiration: ⤷ Start Trial
Patent: 13404
Estimated Expiration: ⤷ Start Trial
Patent: 51771
Estimated Expiration: ⤷ Start Trial
Patent: 11152960
Estimated Expiration: ⤷ Start Trial
Patent: 11154083
Estimated Expiration: ⤷ Start Trial
Patent: 11154148
Estimated Expiration: ⤷ Start Trial
Patent: 15151358
Estimated Expiration: ⤷ Start Trial
Patent: 16107464
Estimated Expiration: ⤷ Start Trial
Patent: 16117972
Estimated Expiration: ⤷ Start Trial
Patent: 20102859
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01600326
Estimated Expiration: ⤷ Start Trial
Patent: 01600327
Estimated Expiration: ⤷ Start Trial
Patent: 01600329
Estimated Expiration: ⤷ Start Trial
Patent: 02000077
Estimated Expiration: ⤷ Start Trial
Patent: 02000108
Estimated Expiration: ⤷ Start Trial
Patent: 02000109
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 35023
Estimated Expiration: ⤷ Start Trial
Patent: 35024
Estimated Expiration: ⤷ Start Trial
Patent: 35025
Estimated Expiration: ⤷ Start Trial
Patent: 06149
Estimated Expiration: ⤷ Start Trial
Patent: 11926
Estimated Expiration: ⤷ Start Trial
Patent: 11927
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1108275
Patent: RESPIRATORY DELIVERY OF ACTIVE AGENTS
Estimated Expiration: ⤷ Start Trial
Patent: 1208100
Patent: COMPOSITIONS FOR RESPIRATORY DELIVERY OF ACTIVE AGENTS AND ASSOCIATED METHODS AND SYSTEMS
Estimated Expiration: ⤷ Start Trial
Patent: 1208101
Patent: COMPOSITIONS FOR PULMONARY DELIVERY OF LONG-ACTING MUSCARINIC ANTAGONISTS AND LONG-ACTING B2 ADRENERGIC RECEPTOR AGONISTS AND ASSOCIATED METHODS AND SYSTEMS
Estimated Expiration: ⤷ Start Trial
Patent: 1208102
Patent: RESPIRATORY DELIVERY OF ACTIVE AGENTS
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1748892
Estimated Expiration: ⤷ Start Trial
Patent: 1926060
Estimated Expiration: ⤷ Start Trial
Patent: 1976107
Estimated Expiration: ⤷ Start Trial
Patent: 120015334
Estimated Expiration: ⤷ Start Trial
Patent: 120026075
Estimated Expiration: ⤷ Start Trial
Patent: 120034631
Estimated Expiration: ⤷ Start Trial
Patent: 170070274
Estimated Expiration: ⤷ Start Trial
Patent: 170104003
Estimated Expiration: ⤷ Start Trial
Patent: 180130602
Estimated Expiration: ⤷ Start Trial
Patent: 190049943
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 89135
Estimated Expiration: ⤷ Start Trial
Patent: 92536
Estimated Expiration: ⤷ Start Trial
Patent: 93429
Estimated Expiration: ⤷ Start Trial
Patent: 72253
Estimated Expiration: ⤷ Start Trial
Patent: 74367
Estimated Expiration: ⤷ Start Trial
Patent: 74391
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 39979
Estimated Expiration: ⤷ Start Trial
Patent: 46094
Estimated Expiration: ⤷ Start Trial
Patent: 32926
Estimated Expiration: ⤷ Start Trial
Patent: 33898
Estimated Expiration: ⤷ Start Trial
Patent: 46980
Estimated Expiration: ⤷ Start Trial
Patent: 95723
Estimated Expiration: ⤷ Start Trial
Patent: 07700
Estimated Expiration: ⤷ Start Trial
Patent: 17511
Estimated Expiration: ⤷ Start Trial
Patent: 92140
Estimated Expiration: ⤷ Start Trial
Patent: 1109049
Patent: Compositions for respiratory delivery of active agents and associated methods and systems
Estimated Expiration: ⤷ Start Trial
Patent: 1109050
Patent: Compositions for pulmonary delivery of long-acting muscarinic antagonists and long-acting &bgr;2 adrenergic receptor agonists and associated methods and systems
Estimated Expiration: ⤷ Start Trial
Patent: 1109051
Patent: Compositions, methods and systems for respiratory delivery of two or more active agents
Estimated Expiration: ⤷ Start Trial
Patent: 1642836
Patent: Compositions for pulmonary delivery of long-acting muscarinic antagonists and long-acting [beta]2 adrenergic receptor agonists and associated methods and systems
Estimated Expiration: ⤷ Start Trial
Patent: 1700123
Patent: Compositions for respiratory delivery of active agents and associated methods and systems
Estimated Expiration: ⤷ Start Trial
Patent: 1808372
Patent: Compositions, methods and systems for respiratory delivery of two or more active agents
Estimated Expiration: ⤷ Start Trial
Patent: 1919730
Patent: Compositions for respiratory delivery of active agents and associated methods and systems
Estimated Expiration: ⤷ Start Trial
Patent: 1936174
Patent: Compositions for pulmonary delivery of long-acting muscarinic antagonists and long-acting [beta]2 adrenergic receptor agonists and associated methods and systems
Estimated Expiration: ⤷ Start Trial
Patent: 2114642
Patent: Compositions, methods and systems for respiratory delivery of two or more active agents
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 9529
Patent: КОМПОЗИЦІЇ ДЛЯ РЕСПІРАТОРНОЇ ДОСТАВКИ АКТИВНИХ АГЕНТІВ ТА ПОВ'ЯЗАНІ СПОСОБИ І СИСТЕМИ
Estimated Expiration: ⤷ Start Trial
Patent: 9530
Patent: КОМПОЗИЦІЇ, СПОСОБИ ТА СИСТЕМИ ДЛЯ ДОСТАВКИ РЕСПІРАТОРНИМ ШЛЯХОМ ДВОХ ЧИ БІЛЬШЕ АКТИВНИХ АГЕНТІВ
Estimated Expiration: ⤷ Start Trial
Patent: 9531
Patent: КОМПОЗИЦІЇ ДЛЯ ЛЕГЕНЕВОЇ ДОСТАВКИ МУСКАРИНОВИХ АНТАГОНІСТІВ ТРИВАЛОЇ ДІЇ ТА АГОНІСТІВ АДРЕНЕРГІЧНИХ РЕЦЕПТОРІВ B2 ТРИВАЛОЇ ДІЇ ТА ПОВ'ЯЗАНІ СПОСОБИ І СИСТЕМИ
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering BEVESPI AEROSPHERE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Poland | 3111926 | ⤷ Start Trial | |
| Lithuania | 3106149 | ⤷ Start Trial | |
| Taiwan | I717511 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for BEVESPI AEROSPHERE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2435025 | 122019000068 | Germany | ⤷ Start Trial | PRODUCT NAME: KOMBINATION VON GLYCOPYRROLAT, EINSCHLIESSLICH BELIEBIGER PHARMAZEUTISCH VERTRAEGLICHER SALZE, ESTER, ENANTIOMERE, ODER ANDERER DERIVATE DAVON, UND FORMOTEROL, EINSCHLIESSLICH BELIEBIGER PHARMAZEUTISCH VERTRAEGLICHER SALZE, ESTER, ENANTIOMERE, ODER ANDERER DERIVATE DAVON; REGISTRATION NO/DATE: EU/1/18/1339 20181218 |
| 2435024 | 301102 | Netherlands | ⤷ Start Trial | PRODUCT NAME: COMBINATIE VAN FORMOTEROL (MET INBEGRIP VAN DE FARMACEUTISCH AANVAARDBARE ZOUTEN, ESTERS, SOLVATEN OF ENANTIOMEREN ERVAN), GLYCOPYRROLAAT (MET INBEGRIP VAN DE FARMACEUTISCH AANVAARDBARE ZOUTEN, ESTERS, SOLVATEN OF ENANTIOMEREN ERVAN) EN BUDESONIDE (MET INBEGRIP VAN DE FARMACEUTISCH AANVAARDBARE ZOUTEN, ESTERS, SOLVATEN OF ENANTIOMEREN ERVAN); REGISTRATION NO/DATE: EU/1/20/1498 20201210 |
| 2435025 | PA2019014,C2435025 | Lithuania | ⤷ Start Trial | PRODUCT NAME: GLIKOPIRONIO BROMIDAS/FORMOTEROLIS; REGISTRATION NO/DATE: EU/1/18/1339 20181218 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
BEVESPI AEROSPHERE: Market Dynamics and Financial Trajectory Analysis
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
