Last updated: February 19, 2026
BACTROBAN, a topical antibiotic, holds a stable position in the antibacterial market, primarily indicated for the treatment of impetigo and nasal staphylococcal colonization. Its market dynamics are shaped by established efficacy, generic competition, and evolving treatment guidelines for skin infections. The financial trajectory is characterized by mature product revenue, limited pipeline expansion, and reliance on market access in key regions.
What is BACTROBAN's Primary Therapeutic Use and Mechanism of Action?
BACTROBAN is a prescription topical antibiotic. Its active pharmaceutical ingredient is mupirocin. Mupirocin is an inhibitor of bacterial protein synthesis. It achieves this by reversibly binding to and inhibiting bacterial isoleucyl-tRNA synthetase [1]. This enzyme is crucial for the incorporation of isoleucine into polypeptide chains. By blocking this process, mupirocin effectively halts bacterial protein production, leading to bacterial death.
BACTROBAN is indicated for the treatment of:
- Impetigo: A highly contagious superficial bacterial skin infection, most commonly caused by Staphylococcus aureus or Streptococcus pyogenes. BACTROBAN is effective against the strains of these bacteria responsible for impetigo.
- Nasal Staphylococcal Colonization: This refers to the presence of Staphylococcus aureus bacteria in the nasal passages without causing an active infection. BACTROBAN is used to eradicate these colonizing bacteria, particularly in individuals at risk of transmitting the bacteria or developing invasive infections, such as healthcare workers or those undergoing certain surgical procedures [2].
The drug is available in different formulations, including an ointment and a nasal ointment. The topical application targets the site of infection or colonization, minimizing systemic exposure and associated side effects.
How Has BACTROBAN's Market Penetration Evolved?
BACTROBAN, first approved in the United States in 1988, has achieved significant market penetration within its defined therapeutic areas due to its well-established efficacy and safety profile. Its initial market entry was driven by the need for effective topical treatments for common bacterial skin infections, offering an alternative to systemic antibiotics which carry a higher risk of side effects and contribute to broader antimicrobial resistance.
The market for topical antibiotics is characterized by:
- Established Brand Loyalty: For healthcare providers and patients familiar with BACTROBAN's performance, it maintains a degree of brand loyalty.
- Treatment Guidelines: Inclusion in clinical practice guidelines for managing impetigo and staphylococcal nasal colonization has reinforced its use. For instance, the Infectious Diseases Society of America (IDSA) guidelines have historically recognized mupirocin as a first-line option for impetigo and nasal decolonization [3].
- Generic Availability: The expiration of original patents led to the introduction of generic mupirocin products. This has broadened accessibility and reduced pricing, but also intensified competition, impacting branded BACTROBAN's market share and pricing power. Generic mupirocin now constitutes a substantial portion of the overall mupirocin market.
The market penetration can be segmented by indication and region. In the United States and Europe, BACTROBAN and its generic equivalents are widely prescribed for impetigo. Nasal decolonization use is more prominent in specific healthcare settings and for certain patient populations.
What are the Key Competitive Forces Affecting BACTROBAN?
The competitive landscape for BACTROBAN is multifaceted, influenced by both direct and indirect competitors, as well as evolving therapeutic approaches.
Direct Competitors:
- Generic Mupirocin: The most significant competitive force is the widespread availability of generic mupirocin products. These generics offer the same active ingredient and therapeutic benefits at a lower cost, directly eroding the market share and pricing flexibility of branded BACTROBAN. Manufacturers of generic mupirocin, such as Teva Pharmaceuticals, Mylan, and Sandoz, contribute to this intensified competition.
- Other Topical Antibiotics: While mupirocin is a key player, other topical antibiotic agents compete for the treatment of bacterial skin infections. These include:
- Retapamulin (Altabax): A topical antibiotic from GlaxoSmithKline, approved for impetigo.
- Bacitracin/Neomycin/Polymyxin B (e.g., Neosporin): Combination topical antibiotics that are widely available over-the-counter (OTC) and by prescription. While their spectrum of activity differs and resistance patterns are a concern, they are frequently used for minor cuts and scrapes.
- Clindamycin and Erythromycin: Topical formulations of these antibiotics are also prescribed for certain skin infections.
Indirect Competitors and Market Trends:
- Antimicrobial Resistance (AMR): The increasing prevalence of antibiotic-resistant bacteria poses a significant challenge. While mupirocin resistance is reported, it is generally considered lower than for some other topical antibiotics [4]. However, the emergence of mupirocin-resistant Staphylococcus aureus strains (particularly High-Level Mupirocin Resistance - HMLR) is a growing concern, especially in clinical settings [5]. This trend necessitates careful stewardship and can influence prescribing patterns, potentially favoring agents with different mechanisms of action or limiting the duration of mupirocin use.
- Shifting Treatment Paradigms: Clinical guidelines and research continue to evolve. There is an ongoing effort to optimize antibiotic use, de-escalate treatment where appropriate, and explore non-antibiotic or adjunct therapies for certain skin conditions.
- Over-the-Counter (OTC) Availability: Some topical antibiotic combinations are available OTC, leading to self-treatment for minor skin issues, potentially bypassing prescription-based options like BACTROBAN.
The competitive intensity is high, driven by pricing pressures from generics and the availability of alternative therapeutic options.
What is BACTROBAN's Revenue Generation and Financial Performance?
BACTROBAN's financial performance is indicative of a mature pharmaceutical product. As a branded product facing significant generic competition, its revenue trajectory is characterized by a gradual decline or stabilization, with growth limited to specific market expansions or niche indications.
Key Financial Aspects:
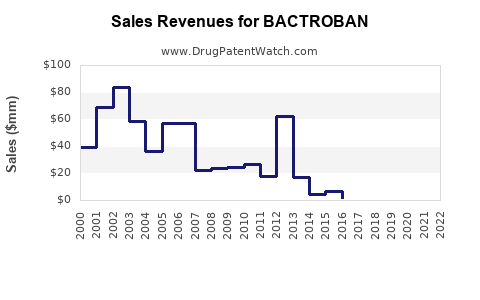
- Sales Revenue: Precise, up-to-the-minute global sales figures for branded BACTROBAN are typically proprietary information held by the marketing authorization holder, GSK. However, based on market analysis of topical antibiotics and the impact of generic erosion, it is understood that the revenue generated by branded BACTROBAN has likely plateaued or is in a declining phase. Global sales of topical antibiotics are estimated to be in the hundreds of millions of dollars annually, with mupirocin generics capturing a significant share.
- Pricing Dynamics: The price of branded BACTROBAN is considerably higher than that of generic mupirocin. This price differential is a primary driver for the shift towards generics. The pricing strategy for branded BACTROBAN would focus on maintaining value proposition in specific markets or for specific physician segments, while generic pricing is driven by cost-effectiveness and market volume.
- Profit Margins: While specific margin data is not publicly available, branded pharmaceuticals generally command higher profit margins than their generic counterparts due to established brand equity, marketing investment, and research and development recovery. However, with declining sales volumes due to generic competition, the overall profit contribution from branded BACTROBAN may be reduced.
- Market Share: Branded BACTROBAN's market share within the overall mupirocin market has diminished significantly since the advent of generics. It now competes with a multitude of generic manufacturers. Its share within the broader topical antibacterial market is also impacted by other active ingredients and product types.
- Investment and R&D: Given its mature status, significant new investment in R&D for BACTROBAN is unlikely. Focus would likely be on maintaining existing market access, supporting post-marketing surveillance, and potentially exploring minor line extensions or label expansions if economically viable. The bulk of R&D investment in the antibacterial space is directed towards novel antibiotics to address the growing AMR crisis.
The financial trajectory is largely determined by its ability to retain a segment of the market that values the branded product, alongside the overall market size for topical mupirocin.
What is the Patent Landscape and Exclusivity Status of BACTROBAN?
The patent landscape for BACTROBAN (mupirocin) has largely evolved from a period of strong patent protection to one of widespread generic availability.
Original Patents and Expiration:
- The original patents covering the composition of matter and methods of use for mupirocin expired decades ago. The initial US patent for mupirocin expired in the early 2000s.
- This expiration opened the door for numerous generic manufacturers to enter the market. Companies like Teva Pharmaceuticals, Mylan, and Dr. Reddy's Laboratories have historically been significant players in the generic mupirocin market.
Current Exclusivity:
- As a molecule with expired composition of matter patents, there is no further market exclusivity for the active pharmaceutical ingredient itself.
- Any remaining exclusivity would likely be tied to specific formulations, manufacturing processes, or new method-of-use patents that may have been pursued over time. However, these are less impactful than core composition patents.
- Regulatory Exclusivity: In some jurisdictions, there may be a limited period of regulatory data exclusivity or market exclusivity tied to specific product approvals or label extensions, but these are typically short-lived and do not provide broad market protection.
- Trademarks: The brand name "BACTROBAN" is protected by trademark law, which prevents competitors from using the identical brand name. However, it does not prevent the marketing of generic equivalents under different names.
The absence of strong patent protection means that BACTROBAN operates in a highly competitive genericized market. Its market position is sustained through brand recognition, established physician preference, and potentially specific supply agreements or formulary placements, rather than through patent-protected market exclusivity.
What are the Regulatory Considerations and Market Access Challenges for BACTROBAN?
BACTROBAN, like all pharmaceutical products, is subject to stringent regulatory oversight and faces ongoing market access challenges.
Regulatory Considerations:
- Prescription Status: BACTROBAN is a prescription-only medication in most major markets, including the United States and European Union. This requires a healthcare professional to diagnose and prescribe the medication.
- Labeling and Indications: Regulatory approvals dictate the specific indications for which BACTROBAN can be marketed and prescribed. Any expansion of indications would require rigorous clinical trials and subsequent regulatory review and approval.
- Pharmacovigilance: Post-marketing surveillance for adverse events and drug safety is an ongoing regulatory requirement. GSK, as the marketing authorization holder, is responsible for monitoring and reporting any safety concerns.
- Manufacturing Standards: Production facilities must adhere to Good Manufacturing Practices (GMP) to ensure product quality, purity, and consistency. Regulatory bodies conduct inspections to verify compliance.
- Antimicrobial Stewardship: Regulatory bodies and public health organizations are increasingly emphasizing antimicrobial stewardship to combat resistance. This can influence prescribing guidelines and potentially lead to recommendations for judicious use of topical antibiotics like mupirocin, including limits on duration of treatment or use in specific patient populations.
Market Access Challenges:
- Pricing and Reimbursement: In many healthcare systems, particularly in Europe and government-funded programs in the US, pricing and reimbursement negotiations are critical. The presence of cheaper generic alternatives places downward pressure on the pricing of branded BACTROBAN. Payers may favor generics or require significant justification for the higher cost of the branded product.
- Generic Substitution: Pharmacy benefit managers (PBMs) and healthcare systems often promote or mandate generic substitution, where a generic version of a prescribed drug is dispensed unless the prescriber specifically requests the branded product. This directly impacts branded BACTROBAN's sales volume.
- Formulary Placement: Inclusion on hospital and insurance formularies is essential for market access. Competition from generics can make it challenging for branded BACTROBAN to secure preferred formulary status, especially if cost-effectiveness is a primary determinant.
- Antibiotic Resistance Data: Evidence of increasing mupirocin resistance in relevant bacterial pathogens can impact prescribing decisions and potentially lead to reluctance from payers to reimburse for the branded product if effective and cheaper alternatives with lower resistance rates emerge or are preferred.
- Competition from Non-Antibiotic Therapies: In some dermatological conditions, non-antibiotic therapies or approaches are gaining traction, potentially reducing the overall market size for topical antibiotics.
Navigating these regulatory and market access hurdles is crucial for the continued commercial viability of BACTROBAN, even in its mature phase.
What is the Future Outlook for BACTROBAN?
The future outlook for BACTROBAN is primarily defined by its status as a mature, genericized product. Significant growth is not anticipated, with its trajectory largely dependent on maintaining its existing market share within specific niches and regions.
Key Outlook Factors:
- Continued Generic Dominance: The market for mupirocin will remain dominated by generic products. Branded BACTROBAN will likely continue to represent a smaller, value-added segment, potentially catering to specific physician preferences or healthcare systems with different purchasing dynamics.
- Stable Demand for Impetigo Treatment: Impetigo remains a common childhood infection, ensuring a baseline demand for effective topical treatments like mupirocin. BACTROBAN will continue to be a recognized option, albeit one among many generics.
- Niche Use in Nasal De-colonization: Its role in nasal staphylococcal de-colonization, particularly in healthcare settings or for specific patient groups, may provide a steady, albeit limited, revenue stream.
- Impact of Antimicrobial Resistance: The ongoing challenge of AMR will influence prescribing patterns. While mupirocin has a relatively favorable resistance profile compared to some older topical antibiotics, the emergence of mupirocin-resistant strains could, over time, lead to reduced utilization or preference for alternative agents, especially in high-prevalence settings.
- Limited Pipeline Expansion: It is highly improbable that significant new R&D investment will be directed towards BACTROBAN for novel indications or formulations. The focus for GSK, if any, would be on lifecycle management for the existing product.
- Competitive Pressure: Ongoing price pressure from generic manufacturers and competition from other topical antibiotics and potentially novel therapeutic approaches will persist.
- Geographic Variations: Market dynamics will vary geographically. Developed markets may see a greater shift towards generics, while emerging markets might still present opportunities for branded products if specific distribution channels or marketing efforts are in place.
In summary, BACTROBAN's future is one of sustained, but not growing, presence in its established therapeutic areas. Its financial trajectory will be characterized by mature, stable-to-declining revenues for the branded product, overshadowed by the larger market volume of its generic counterparts.
Key Takeaways
- BACTROBAN (mupirocin) is a topical antibiotic for impetigo and nasal staphylococcal colonization, inhibiting bacterial protein synthesis.
- Its market penetration is established but challenged by widespread generic mupirocin availability and competition from other topical antibiotics.
- Key competitive forces include generic erosion, other topical antibacterial agents, and the growing concern of antimicrobial resistance.
- Financial performance reflects a mature product, with branded BACTROBAN revenue likely stable or declining due to genericization, contrasting with the overall mupirocin market volume.
- Original composition of matter patents have expired, leading to a highly competitive generic market with no significant ongoing patent-protected exclusivity for the active ingredient.
- Regulatory considerations include prescription status and pharmacovigilance, while market access challenges involve pricing, reimbursement, generic substitution, and formulary placement.
- The future outlook for BACTROBAN involves continued generic dominance, stable demand for its primary indications, ongoing competitive pressure, and a limited potential for significant growth.
Frequently Asked Questions
What is the difference between branded BACTROBAN and generic mupirocin?
Branded BACTROBAN is the original product marketed by GSK. Generic mupirocin products contain the same active ingredient, mupirocin, and are therapeutically equivalent. The primary difference lies in the manufacturer, branding, and typically, the price, with generics being significantly less expensive.
How does antimicrobial resistance affect the use of BACTROBAN?
The development of antibiotic resistance, including resistance to mupirocin, is a global health concern. While mupirocin resistance is less common than for some other antibiotics, the emergence of resistant strains can influence prescribing practices, leading to more cautious use or a preference for alternative agents in certain situations, particularly in settings with high rates of resistance.
Is BACTROBAN available over-the-counter (OTC)?
No, BACTROBAN is a prescription-only medication in most countries. A healthcare professional must diagnose the condition and prescribe the medication.
What are the most common side effects of BACTROBAN?
Common side effects of topical mupirocin include local skin reactions such as burning, stinging, itching, redness, dryness, or mild pain at the application site. Systemic side effects are rare due to limited absorption.
Can BACTROBAN be used to treat acne?
BACTROBAN is not indicated for the treatment of acne. Its approved uses are for impetigo and nasal staphylococcal colonization. Acne treatment typically involves different classes of medications targeting the underlying causes of acne.