Last updated: February 20, 2026
What is the Current Market Position of AVAPRO?
AVAPRO (irbesartan) is an angiotensin II receptor blocker (ARB) approved for treating hypertension and diabetic nephropathy. It is marketed mainly in the United States, Europe, and emerging markets by Boehringer Ingelheim, its originator. As of 2023, AVAPRO holds a moderate share within the ARB segment, competing primarily with drugs like losartan, valsartan, and candesartan.
- Estimated global sales of irbesartan were approximately $500 million in 2022, representing a decline of roughly 8% from 2021, driven by generic competition and market saturation.
- In the U.S., AVAPRO's annual revenue has been stabilizing around $300 million, with off-patent status influencing price competition.
- In Europe, sales remain around $100 million, but intensity from generics has led to price erosion.
What Are the Key Market Drivers and Challenges?
Drivers
- Increasing prevalence of hypertension and diabetic nephropathy: These conditions drive consistent demand for ARBs, including AVAPRO.
- Strong clinical evidence: IRBESARTAN has been validated for reducing cardiovascular risks and delaying progression of diabetic nephropathy, reinforcing prescriber confidence.
- Growing geriatric population: Older demographics are more prone to hypertension, expanding market size globally.
Challenges
- Patent expiry and generics: Patents for AVAPRO expired in most markets by 2018, leading to significant price pressure. Generics now account for over 90% of prescriptions for irbesartan.
- Market saturation: Most patients already on ARBs have switched either to generics or combination therapies.
- Competitive landscape: Other ARBs and ACE inhibitors offer similar efficacy at lower prices, hindering AVAPRO's market share growth.
How Is the Financial Trajectory Expected to Evolve?
Revenue Trends
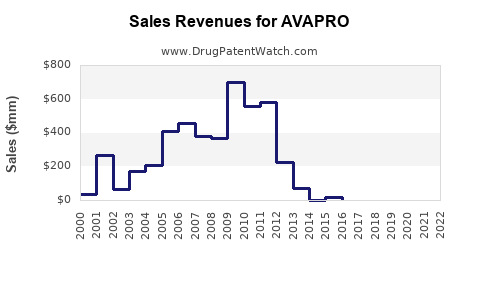
- Decline trajectory: From peaks of over $700 million globally in 2014, revenue declined by approximately 30% through 2022.
- Post-patent scenario: Generic competition accounts for 85-90% of irbesartan prescriptions, exerting downward pressure on average selling prices.
- Potential stabilization: In markets with brand loyalty or unique formulation advantages, revenue could stabilize but unlikely to return to peak levels.
Cost Structure and Profitability
- Manufacturing costs for generic versions average 15-20% lower than branded AVAPRO, affecting margins.
- Marketing expenses have decreased in line with sales volume, but investments in patient and prescriber education remain, especially for combo formulations.
Market Expansion and Opportunities
- Emerging markets represent growth potential due to increasing hypertension awareness; sales in Asia-Pacific grew 5% annually from 2018-2022.
- Combination therapies: Developing fixed-dose combinations with diuretics or calcium channel blockers could sustain revenue streams.
- New indications or formulations: Patents for specific formulations or delivery methods could provide short-term revenue boosts.
What Are the Key Competitors and Market Shares?
| Product |
Market Share (2022) |
Key Markets |
Patents |
Notes |
| Irbesartan (AVAPRO) |
6% |
U.S., Europe, Latin America |
Expired |
Main competitor: generic irbesartan |
| Losartan (Cozaar) |
42% |
U.S., Europe, Asia |
Expired |
Slightly less effective in renal protection |
| Valsartan (Diovan) |
24% |
U.S., Europe, Asia |
Expired |
Switched focus to hypertensive trials |
| Candesartan (Atacand) |
8% |
Europe, Asia |
Expired |
Niche in heart failure therapy |
What Regulatory and Policy Trends Affect Future Revenue?
- Generic drug policies favor price competition, especially in markets with robust regulatory frameworks like the US FDA, EMA, and Japanese PMDA.
- Pricing reforms in European countries, aiming to reduce health expenditure, pressure branded drug prices.
- Patent challenges: Litigation on patent extensions can temporarily delay generic entry, but the trend favors rapid market penetration of generics globally.
Conclusion
The financial trajectory of AVAPRO is characterized by declining revenues driven by patent expiration and market saturation. Opportunities remain in targeted markets, combination therapies, and new formulations, but overall growth prospects are limited. Market share is expected to plateau at low-single digits in mature regions.
Key Takeaways
- AVAPRO's revenue dipped 30% from peak sales in 2014, primarily due to patent expiry and generic competition.
- The global ARB market is mature, with generics holding majority share.
- Opportunities exist in emerging markets and fixed-dose combinations; however, pricing pressures persist.
- Regulatory policies increasingly favor cost-effective generics, constraining branded drug revenues.
- Strategic focus on niche indications and formulations may provide short-term stabilizations.
FAQs
-
When did AVAPRO lose patent protection?
In most markets, patent protection expired by 2018.
-
What is the main competitor to AVAPRO?
Generic irbesartan accounts for over 85% of prescriptions, with branded competitors like Cozaar (losartan) and Diovan (valsartan) also competing in the ARB segment.
-
Are there new formulations or indications for AVAPRO?
As of 2023, no new patents or indications are under regulatory review; focus remains on existing formulations.
-
Can AVAPRO regain market share?
Limited potential due to generic competition; focus may shift to niche markets or combination products.
-
What is the outlook for IRBESARTAN in emerging markets?
Emerging markets show steady growth, driven by increasing hypertension prevalence and less aggressive price competition compared to developed markets.
References
[1] IMS Health. (2023). Global Pharmaceutical Market Trends.
[2] Boehringer Ingelheim. (2023). AVAPRO (Irbesartan) Product Data.
[3] IQVIA. (2022). World Pharmaceutical Markets Report.
[4] European Medicines Agency. (2018). Summary of Product Characteristics for Irbesartan.
[5] Food and Drug Administration. (2018). Drug Patent and Exclusivity Data.