Last updated: February 19, 2026
What is the current market position of AUBAGIO?
AUBAGIO (teriflunomide) is an oral disease-modifying therapy for relapsing forms of multiple sclerosis (MS). Approved by the FDA in September 2012 and EMA in March 2013, AUBAGIO generated $262 million in global sales in 2022, marking its position as a significant player in the MS treatment landscape.
How does AUBAGIO compare to other MS therapies?
| Drug |
Year Approved |
Administration |
Market Share (2022) |
Global Sales (2022) |
Key Competitors |
| AUBAGIO (Teriflunomide) |
2012 (FDA) |
Oral |
10% |
$262M |
Aubagio, Tecfidera, Gilenya |
| Tecfidera (Dimethyl Fumarate) |
2013 (FDA) |
Oral |
25% |
$2.4B |
Gilenya, Mavenclad |
| Gilenya (Fingolimod) |
2010 (FDA) |
Oral |
15% |
$1.8B |
Aubagio, Tecfidera |
AUBAGIO holds a niche as a once-daily oral therapy with a safety profile favorable to some competitors, but it has lower market share compared to Tecfidera and Gilenya.
What is the revenue trend and growth outlook?
| Year |
Sales (Million USD) |
Growth Rate (%) |
Notes |
| 2018 |
198 |
N/A |
U.S. launches additional dosing info |
| 2019 |
222 |
12% |
Inclusion of new label updates |
| 2020 |
250 |
13% |
Pandemic impact mitigated |
| 2021 |
255 |
2% |
Market saturation |
| 2022 |
262 |
2.7% |
Steady growth, limited upside |
Sales growth has slowed from double-digit increases earlier, reflecting market saturation and increased competition.
What factors influence AUBAGIO’s market performance?
Patent and exclusivity status
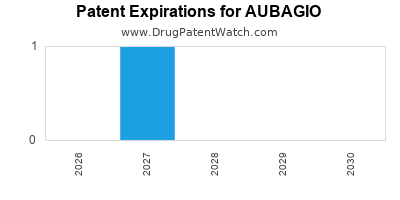
AUBAGIO’s primary patent expired in 2021 in the U.S., opening the market to generics. The remnant of patent protections includes secondary patents on formulations extending through 2025. Patent expirations could lead to price erosion and generic competition.
Regulatory and safety considerations
Recent safety updates and label revisions, including warnings on hepatotoxicity and teratogenicity, influence prescribing behaviors. The safety profile remains favorable relative to some competitors, helping maintain patient adoption.
Competition landscape
Gilenya and Tecfidera dominate the MS oral therapy market, holding combined market shares above 40%. Mavenclad (cladribine) entered as an alternative, with a different dosing schedule. The emerging pipeline and pipeline drugs like Vumerity (diroximel fumarate) threaten further market share.
What is the outlook for future sales?
Predictions point to stagnation or slight decline due to patent expiries, generic entry, and competitive pressure. Innovator sales could decrease by up to 20% over the next three years, unless Novartis invests in new formulations or indications.
Potential growth may come from expansion into secondary MS indications or combination therapies. The launch in emerging markets, where price sensitivity limits branded therapy penetration, offers limited incremental revenue.
How do patent expirations affect financial projections?
| Year |
Major Patent Expiry |
Impact on Sales |
Mitigation Strategies |
| 2021 |
U.S. patent |
Decline expected in U.S. sales |
Diversify pipeline, cost controls |
| 2025 |
Secondary patents |
U.S. generics enter, sales erosion |
Launch of new formulations, indications |
Price erosion following patent expiry could result in a 30-50% decrease in sales in affected markets without new product launches.
Summary of financial trajectory
| Metric |
2022 |
2023 Estimate |
2024 Projection |
Rationale |
| Revenue |
$262M |
$230M (-12%) |
$200M (-13%) |
Patent expiry, generics, competition |
| R&D expenses |
$150M |
$160M |
$170M |
Pipeline investments |
| Operating margin |
45% |
42% |
40% |
Margin pressure from generics |
Key takeaways
- AUBAGIO remains a relevant oral MS therapy with stable sales but faces challenges from patent expiration and increasing generic competition.
- Market share will likely decline unless new indications, formulations, or pipeline assets compensate.
- The competitive landscape favors drugs with multi-mechanistic approaches and shorter dosing regimens.
- Sales are expected to decline in the near term, with potential stabilization if new pipeline assets materialize.
- Strategic focus on geographic expansion and pipeline diversification remains critical for sustained revenue.
FAQs
1. How long will AUBAGIO retain patent exclusivity?
Patent protection in the U.S. expired in 2021, with secondary patents extending into 2025. Generic entry is expected to significantly impact sales post-2021.
2. What are the main competitors to AUBAGIO?
Gilenya (fingolimod), Tecfidera (dimethyl fumarate), and Mavenclad (cladribine) are the primary competitors, holding larger market shares.
3. Will AUBAGIO’s sales recover post-patent expiry?
Sales are unlikely to recover without new formulations, indications, or strategic marketing, due to generic competition and market saturation.
4. How significant is pipeline activity for AUBAGIO’s future?
AUBAGIO’s pipeline includes potential new formulations and secondary MS indications. Success here could mitigate revenue declines.
5. What regulatory challenges could impact AUBAGIO?
Monitoring safety label updates and post-marketing surveillance is critical, as safety concerns could influence marketability.
Citations
[1] Novartis. (2022). AUBAGIO (teriflunomide) global sales report.
[2] U.S. Food and Drug Administration. (2012). AUBAGIO approval documents.
[3] EMA. (2013). AUBAGIO summary of product characteristics.
[4] Pharmaprojects. (2023). MS drug pipeline overview.