ANNOVERA Drug Patent Profile
✉ Email this page to a colleague
When do Annovera patents expire, and what generic alternatives are available?
Annovera is a drug marketed by Mayne Pharma and is included in one NDA. There are nine patents protecting this drug.
This drug has eighteen patent family members in seventeen countries.
The generic ingredient in ANNOVERA is ethinyl estradiol; segesterone acetate. There are twenty-six drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the ethinyl estradiol; segesterone acetate profile page.
DrugPatentWatch® Generic Entry Outlook for Annovera
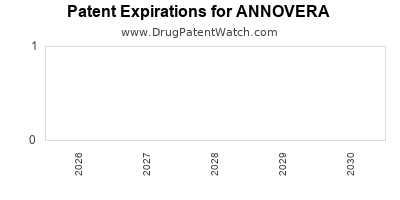
Annovera was eligible for patent challenges on August 10, 2022.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 21, 2039. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ANNOVERA?
- What are the global sales for ANNOVERA?
- What is Average Wholesale Price for ANNOVERA?
Summary for ANNOVERA
| International Patents: | 18 |
| US Patents: | 9 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Clinical Trials: | 2 |
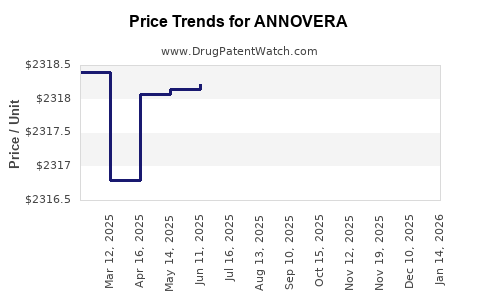
| Drug Prices: | Drug price information for ANNOVERA |
| What excipients (inactive ingredients) are in ANNOVERA? | ANNOVERA excipients list |
| DailyMed Link: | ANNOVERA at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ANNOVERA
Generic Entry Date for ANNOVERA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
RING;VAGINAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ANNOVERA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| TherapeuticsMD | Phase 1 |
Pharmacology for ANNOVERA
| Drug Class | Estrogen Progestin |
| Mechanism of Action | Estrogen Receptor Agonists |
US Patents and Regulatory Information for ANNOVERA
ANNOVERA is protected by twelve US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ANNOVERA is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
International Patents for ANNOVERA
When does loss-of-exclusivity occur for ANNOVERA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 0976
Patent: SISTEMA PARA PROPORCIONAR UN CONTROL DE LA NATALIDAD
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 20294780
Patent: System for providing birth control
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2021025853
Patent: Sistema para fornecimento de controle de natalidade
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 41077
Patent: SYSTEME DE FOURNITURE DE CONTRACEPTION (SYSTEM FOR PROVIDING BIRTH CONTROL)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 21003410
Patent: Sistema para proporcionar control de la natalidad
Estimated Expiration: ⤷ Start Trial
China
Patent: 4364369
Patent: 用于提供节育的系统 (System for providing birth control)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 21017509
Patent: Sistema para proporcionar control de la natalidad
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 021000267
Patent: SISTEMA PARA PROPORCIONAR CONTROL DE LA NATALIDAD
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 86375
Patent: SYSTÈME DE FOURNITURE DE CONTRACEPTION (SYSTEM FOR PROVIDING BIRTH CONTROL)
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 9135
Patent: מערכת למתן אמצעי מניעה (System for providing birth control)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 80954
Estimated Expiration: ⤷ Start Trial
Patent: 22536836
Patent: 受胎調節を提供するためのシステム
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 22000047
Patent: SISTEMA PARA PROPORCIONAR CONTROL DE LA NATALIDAD. (SYSTEM FOR PROVIDING BIRTH CONTROL.)
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 021553181
Patent: SYSTEM FOR PROVIDING BIRTH CONTROL
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 2110288
Patent: SYSTEM FOR PROVIDING BIRTH CONTROL
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 220027979
Patent: 산아 제한을 제공하기 위한 시스템
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 9131
Patent: СИСТЕМА ДЛЯ ЗАПОБІГАННЯ ВАГІТНОСТІ (SYSTEM FOR PROVIDING BIRTH CONTROL)
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ANNOVERA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 114364369 | 用于提供节育的系统 (System for providing birth control) | ⤷ Start Trial |
| Japan | 7780954 | ⤷ Start Trial | |
| Israel | 289135 | מערכת למתן אמצעי מניעה (System for providing birth control) | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2020257544 | ⤷ Start Trial | |
| Japan | 2022536836 | 受胎調節を提供するためのシステム | ⤷ Start Trial |
| Argentina | 120976 | SISTEMA PARA PROPORCIONAR UN CONTROL DE LA NATALIDAD | ⤷ Start Trial |
| Dominican Republic | P2021000267 | SISTEMA PARA PROPORCIONAR CONTROL DE LA NATALIDAD | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ANNOVERA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0771217 | 07C0001 | France | ⤷ Start Trial | PRODUCT NAME: ETHINYLESTRADIOL BETADEX CLATHRATE; NAT. REGISTRATION NO/DATE: NL 32343 20060710; FIRST REGISTRATION: NL - RVG 31781 20050804 |
| 1214076 | 49/2008 | Austria | ⤷ Start Trial | PRODUCT NAME: WIRKSTOFFKOMBINATION VON ETHINYLESTRADIOL UND DROSPIRENON; REGISTRATION NO/DATE: 1-27586 20080612 |
| 1453521 | 39/2015 | Austria | ⤷ Start Trial | PRODUCT NAME: ETHINYLESTRADIOL UND EINE KOMBINATION VON LEVONORGESTREL UND ETHINYLESTRADIOL; NAT. REGISTRATION NO/DATE: 136021 20150224; FIRST REGISTRATION: SK 17/0017/15-S 20150211 |
| 1214076 | SZ 49/2008 | Austria | ⤷ Start Trial | PRODUCT NAME: WIRKSTOFFKOMBINATION VON ETHINYLESTRADIOL UND DROSPIRENON |
| 0136011 | 2000C/027 | Belgium | ⤷ Start Trial | PRODUCT NAME: ETHINYLESTRADIOLUM / NORETHISTERONI ACETAS; NAT. REGISTRATION NO/DATE: 19 IS 106 F3 20000911; FIRST REGISTRATION: NL RVG 23909 19991124 |
| 1453521 | 93156 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: LEVONORGESTREL ET ETHINYLESTRADIOL; FIRST REGISTRATION DATE: 20150211 |
| 1214076 | C01214076/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: DROSPIRENONE + ETHINYLESTRADIOL; REGISTRATION NUMBER/DATE: SWISSMEDIC 57946 13.06.2008 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
ANNOVERA: Market Dynamics and Financial Trajectory
More… ↓
DrugChatter Q&A for ANNOVERA
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.