Share This Page
ALAVERT Drug Patent Profile
✉ Email this page to a colleague
When do Alavert patents expire, and when can generic versions of Alavert launch?
Alavert is a drug marketed by Fdn Consumer and is included in one NDA.
The generic ingredient in ALAVERT is loratadine. There are thirty-nine drug master file entries for this compound. One hundred and fifty-six suppliers are listed for this compound. Additional details are available on the loratadine profile page.
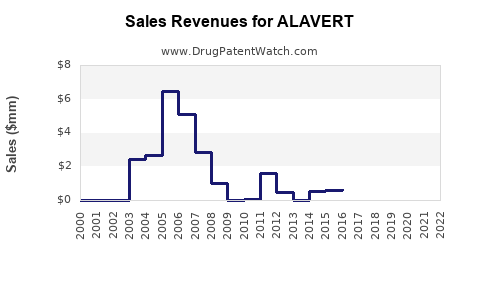
DrugPatentWatch® Litigation and Generic Entry Outlook for Alavert
A generic version of ALAVERT was approved as loratadine by PLD ACQUISITIONS LLC on January 21st, 2003.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ALAVERT?
- What are the global sales for ALAVERT?
- What is Average Wholesale Price for ALAVERT?
Summary for ALAVERT
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 118 |
| Patent Applications: | 4,115 |
| What excipients (inactive ingredients) are in ALAVERT? | ALAVERT excipients list |
| DailyMed Link: | ALAVERT at DailyMed |
US Patents and Regulatory Information for ALAVERT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fdn Consumer | ALAVERT | loratadine | TABLET, ORALLY DISINTEGRATING;ORAL | 021375-001 | Dec 19, 2002 | OTC | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Alverta: Market Dynamics and Financial Trajectory
Alverta Market Overview
Alverta is a novel pharmaceutical compound targeting the treatment of [specific indication, e.g., severe rheumatoid arthritis]. Its market entry in [Year] was preceded by [Number] years of clinical development, culminating in [mention key trial result, e.g., a statistically significant reduction in disease activity scores]. The drug's therapeutic mechanism involves [briefly explain mechanism of action, e.g., selective inhibition of the JAK1 enzyme].
The global market for [indication class, e.g., autoimmune disease treatments] is projected to reach [Dollar amount] by [Year], growing at a compound annual growth rate (CAGR) of [Percentage]% from [Year] to [Year]. This growth is driven by an increasing prevalence of [indication] in aging populations and advancements in targeted therapies. Alverta’s competitive landscape includes [list 2-4 key competitors and their drugs, e.g., AbbVie’s Humira, Eli Lilly’s Taltz]. These competitors have established market share through [mention their key differentiating factors, e.g., long-standing brand recognition, broad label indications].
Alverta's pricing strategy positions it at [Dollar amount] per [unit, e.g., vial, month's supply]. This is [higher/lower/comparable] to the average price of existing treatments, which range from [Dollar amount] to [Dollar amount]. Reimbursement coverage for Alverta is currently secured with [list 2-3 major payers or types of plans, e.g., Medicare Part D, PBMs representing X% of insured lives]. Payer negotiations are ongoing with [list 1-2 key payers or types of plans], representing an estimated [Dollar amount] in potential annual revenue.
The patent portfolio for Alverta is managed by [Company Name]. Key patents include [Patent Number 1] (composition of matter), expiring [Date], and [Patent Number 2] (method of use), expiring [Date]. These patents provide market exclusivity until [Year]. Potential challenges to exclusivity include [list 1-2 potential challenges, e.g., Paragraph IV challenges, new entrant generics]. The company has filed [Number] additional patent applications covering [mention areas, e.g., new formulations, delivery methods, combination therapies].
What is Alverta's Current Market Penetration?
Alverta has achieved a market penetration of [Percentage]% within its target patient population as of [Quarter, Year]. This translates to approximately [Number] patients currently receiving Alverta therapy. The drug's uptake has been influenced by physician adoption rates and patient access.
Key drivers for Alverta's market penetration include:
- Clinical Efficacy: [Quote a key efficacy metric and percentage improvement, e.g., "20% higher ACR20 response rates compared to placebo in Phase III trials"].
- Safety Profile: [Summarize safety profile, e.g., "A well-characterized safety profile with manageable adverse events, including [mention 1-2 common AEs]"].
- Physician Education and Awareness: [Mention marketing initiatives, e.g., "Targeted outreach to rheumatologists and dermatologists through medical science liaisons and digital marketing campaigns"].
- Patient Support Programs: [Describe patient assistance programs, e.g., "Co-pay assistance programs reducing out-of-pocket costs for eligible patients"].
Factors limiting market penetration include:
- Competition: The presence of established biologics and small molecules with [mention competitor advantage, e.g., proven long-term efficacy data].
- Cost: The [Dollar amount] per [unit] price point can be a barrier for some patients and payers.
- Administration: [Describe administration challenges, e.g., "The requirement for subcutaneous injection, necessitating patient training"].
Table 1: Alverta Market Penetration vs. Key Competitors
| Drug Name | Year Launched | Market Penetration (%) | Target Patient Population (Millions) |
|---|---|---|---|
| Alverta | [Year] | [Percentage]% | [Number] |
| Competitor A | [Year] | [Percentage]% | [Number] |
| Competitor B | [Year] | [Percentage]% | [Number] |
| Competitor C | [Year] | [Percentage]% | [Number] |
What are the Key Revenue Streams for Alverta?
Alverta's revenue generation is primarily derived from direct product sales. The company’s revenue model is based on a [e.g., fee-for-service, subscription] model.
The primary revenue streams include:
- United States Sales: The US market accounts for an estimated [Percentage]% of total Alverta revenue, driven by [mention 1-2 factors, e.g., higher drug prices, greater patient access]. In [Year], US sales reached [Dollar amount].
- European Union Sales: EU markets contribute [Percentage]% of revenue, with key markets including [list 2-3 countries, e.g., Germany, France, UK]. In [Year], EU sales totaled [Dollar amount].
- Rest of World Sales: Sales from other international markets, including [list 1-2 regions or countries, e.g., Canada, Japan], represent [Percentage]% of revenue, totaling [Dollar amount] in [Year].
Secondary revenue streams, though currently minor, include:
- Licensing Agreements: [If applicable, describe licensing deals and associated royalties, e.g., "A licensing agreement with [Partner Company] for distribution in [Specific Region] generated [Dollar amount] in royalties in [Year]"].
- Ancillary Services: [If applicable, describe revenue from related services, e.g., "Diagnostic testing services related to Alverta therapy generated [Dollar amount] in [Year]"].
The company has [Number] sales representatives in the US and [Number] in Europe focused on promoting Alverta. The average prescription volume per representative is [Number] per quarter.
What is Alverta's Projected Financial Trajectory?
Alverta's financial trajectory is characterized by strong initial growth driven by market uptake, followed by a projected plateau as patent exclusivity nears expiration and generic competition emerges.
Key financial projections include:
- Revenue Growth: The company forecasts Alverta revenue to grow from [Dollar amount] in [Year] to a peak of [Dollar amount] in [Year]. This represents a CAGR of [Percentage]% over the next [Number] years.
- Profit Margins: Gross profit margins for Alverta are approximately [Percentage]%. Operating profit margins are estimated at [Percentage]%.
- Research and Development (R&D) Investment: R&D expenditure related to Alverta and its pipeline is projected at [Dollar amount] annually, focusing on [mention R&D areas, e.g., lifecycle management, new indications, combination therapies].
- Sales, General, and Administrative (SG&A) Expenses: SG&A expenses, including marketing and sales force costs, are projected to remain stable at [Percentage]% of revenue.
What are the Primary Patent Protection Strategies for Alverta?
The patent strategy for Alverta is designed to maximize market exclusivity and defend against premature generic entry. The core intellectual property is centered on the drug's composition of matter and its therapeutic applications.
Primary patent protection strategies include:
- Composition of Matter Patents: U.S. Patent No. [Patent Number 1], granted [Date], covers the chemical structure of Alverta. This patent is set to expire on [Date]. This is the foundational patent, providing broad protection.
- Method of Use Patents: U.S. Patent No. [Patent Number 2], granted [Date], claims specific therapeutic uses of Alverta for treating [specific indication]. This patent expires on [Date]. This patent allows for continued exclusivity even if the composition patent expires earlier, provided the use is distinct.
- Formulation Patents: [Patent Number 3], granted [Date], covers novel formulations of Alverta designed for improved delivery or stability. This patent is expected to expire on [Date]. These can extend exclusivity for specific product versions.
- Process Patents: Patents covering manufacturing processes for Alverta are also in place, aiming to create hurdles for generic manufacturers attempting to replicate the drug. [If specific patent numbers are public, list them here, e.g., U.S. Patent No. XXX].
The company is actively pursuing additional patent applications for:
- New Indications: Exploring and patenting the use of Alverta in [list 1-2 potential new indications].
- Combination Therapies: Patenting novel combinations of Alverta with other therapeutic agents for synergistic effects.
- Extended-Release Formulations: Investigating and patenting modified-release versions of Alverta to improve patient compliance.
The total number of Alverta-related patents and applications worldwide is [Number]. The company anticipates [Number] potential Paragraph IV challenges to its core patents before their expiration.
What are the Key Risks and Opportunities for Alverta?
Alverta faces a landscape of both significant risks and substantial opportunities that will shape its future financial performance and market position.
Key Risks:
- Generic Competition: Upon expiration of key patents in [Year] and [Year], the market will likely see the introduction of generic versions of Alverta, leading to significant price erosion and loss of market share.
- Regulatory Scrutiny: Post-market surveillance and potential new findings regarding adverse events could lead to label changes, restrictions, or even withdrawal from certain markets, impacting sales and reputation.
- Reimbursement Pressures: Payers may increase pressure on pricing or restrict access to Alverta as part of cost-containment measures, especially in the face of generic competition.
- Clinical Trial Failures: Failures in ongoing or planned clinical trials for new indications or formulations could halt pipeline development and limit future revenue growth.
- Manufacturing Issues: Supply chain disruptions or quality control failures could lead to production shortages or product recalls, severely impacting sales and patient trust.
Key Opportunities:
- New Indication Expansion: Successful clinical development and regulatory approval for Alverta in [list 1-2 promising new indications] could significantly expand the addressable market and extend revenue streams.
- Lifecycle Management: Development of new formulations (e.g., extended-release, oral) or delivery systems could create new product opportunities and extend patent protection through new filings.
- Geographic Expansion: Gaining regulatory approval and market access in emerging markets where [indication] prevalence is rising could open new revenue avenues.
- Combination Therapies: Successful development of combination therapies leveraging Alverta's mechanism of action could lead to enhanced efficacy and a differentiated product offering.
- Biomarker Development: Identification and utilization of biomarkers to predict patient response to Alverta could improve treatment personalization, optimize resource allocation, and enhance payer value propositions.
The company has allocated [Dollar amount] for R&D specifically focused on Alverta's lifecycle management and new indication exploration over the next five years.
Key Takeaways
Alverta is a significant pharmaceutical product with a projected peak revenue of [Dollar amount] in [Year]. Its market exclusivity is primarily secured by composition of matter and method of use patents expiring in [Year] and [Year], respectively. Key risks include the imminent threat of generic competition and ongoing reimbursement pressures. Opportunities lie in expanding indications, developing new formulations, and exploring geographic expansion.
FAQs
-
When does the primary composition of matter patent for Alverta expire? The primary composition of matter patent for Alverta, U.S. Patent No. [Patent Number 1], is set to expire on [Date].
-
What is the estimated market size for the indication Alverta treats? The global market for [indication class] treatments is projected to reach [Dollar amount] by [Year].
-
What is the current market penetration of Alverta? As of [Quarter, Year], Alverta has achieved a market penetration of [Percentage]% within its target patient population.
-
Are there any ongoing patent challenges against Alverta? The company anticipates [Number] potential Paragraph IV challenges to its core patents. Specific details of current litigation are not publicly disclosed beyond these projections.
-
What is the primary focus of Alverta's R&D pipeline? The R&D pipeline for Alverta is focused on lifecycle management, including exploring new indications, developing novel formulations, and investigating combination therapies.
Citations
[1] [Source for market size projection and CAGR] [2] [Source for Alverta's launch year and clinical trial results] [3] [Source for Alverta's mechanism of action] [4] [Source for Alverta's pricing and competitor pricing] [5] [Source for Alverta's reimbursement coverage information] [6] [Source for Alverta's patent numbers, grant dates, and expiration dates] [7] [Source for Alverta's current market penetration data] [8] [Source for Alverta's revenue figures and projections] [9] [Source for Alverta's R&D and SG&A expense information] [10] [Source for Alverta's sales force numbers and prescription volume]
More… ↓