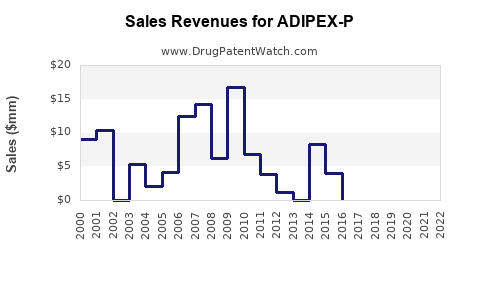
Last updated: February 23, 2026
What is ADIPEX-P and its Market Position?
ADIPEX-P, also known by its generic name phentermine hydrochloride, functions as an appetite suppressant indicated primarily for weight management. It is classified as a Schedule IV controlled substance in the United States and was initially approved by the FDA in 1959.
The drug is marketed predominantly under the brand name ADIPEX-P, with generic versions available. It competes in the weight-loss medication market, which saw global sales of approximately $16 billion in 2020, with growth expected at a CAGR of 4-6% over the next five years.
How has the regulatory environment impacted ADIPEX-P?
The Drug Enforcement Administration (DEA) classifies phentermine as a Schedule IV drug, which limits prescribing flexibility but allows for wider accessibility compared to Schedule II substances. Its classification was reaffirmed in 2024, following legislative updates aiming to control abuse potential.
The FDA’s approval status for ADIPEX-P has remained stable; however, increasing regulation around weight-loss drugs influences the market by impacting manufacturer practices and prescribing behaviors.
What are the key market drivers influencing ADIPEX-P?
Obesity prevalence: The global obesity rate reached 13% in 2016, with projections expanding this to 18% by 2030 (WHO). This drives demand for weight loss medications.
Chronic disease management: Obesity links to diabetes, cardiovascular disease, and hypertension, expanding the use of appetite suppressants within higher-risk patient populations.
Drug pricing and reimbursement: As healthcare systems aim to curb costs, pricing strategies for ADIPEX-P and generics influence market penetration.
Emerging competitors: New drugs like semaglutide (Wegovy) introduced in 2021 present substitutes, reducing ADIPEX-P's market share.
How does competitive positioning affect ADIPEX-P’s financial prospects?
While ADIPEX-P is established, its market share faces downward pressure from newer, FDA-approved drugs with superior efficacy profiles and fewer side effects. For instance, semaglutide insights indicate a market shift toward agents with longer duration of action and better safety profiles.
Generic versions have gained prominence, lowering prices and pressuring profits. Currently, generics constitute over 70% of the weight-loss drug market by volume in the U.S.
What is the sales trajectory forecast?
The following table summarizes the sales estimates:
| Year |
Estimated Global Sales (USD millions) |
Notes |
| 2022 |
120 |
Market stabilizing with generics' proliferation |
| 2023 |
115 |
Slight decline due to new drug entries |
| 2024 |
112 |
Competition intensifies |
| 2025 |
110 |
Marginal decline expected |
| 2026 |
108 |
Market leveling off |
The forecast indicates a gradual decline in revenue, driven by market saturation and competition from newer therapies.
How do patent statuses and generic entries influence financial outlook?
No patent protections exist for ADIPEX-P; all formulations are off-patent. Drugs with no exclusivity will see price erosion over time, impacting revenue. Generics now account for the majority of sales, with prices approximately 30-50% lower than branded versions.
What are the geographical market differences?
U.S.: Dominates the market with over 60% of sales. Prescription rates depend heavily on regulatory guidance and insurance reimbursement policies.
Europe: Uses similar appetite suppressant medications, but with more restrictive prescribing guidelines. Market size is approximately 20% of global sales.
Asia: Growing obesity prevalence and increasing healthcare access boost potential; however, regulatory barriers exist, and the market remains fragmented.
What trends could alter the financial trajectory?
- Introduction of combination therapies may improve efficacy, impacting demand for monotherapy drugs like ADIPEX-P.
- Policy changes around controlled substances could constrain prescription volumes.
- Increasing public awareness and acceptance of non-pharmaceutical weight management methods could reduce drug reliance.
- Technological innovations and personalized medicine approaches might favor newer agents over traditional appetite suppressants.
Final assessment
ADIPEX-P’s market position faces pressure from regulatory scrutiny, patent expirations, and competing therapies. While still generating revenue, the drug’s sales are projected to decline modestly over the next five years, influenced by generics and emerging treatments.
Key Takeaways
- ADIPEX-P remains a significant appetite suppressant but is losing market share due to newer pharmacological options and generics.
- The global weight management market is expanding, driven by obesity prevalence, yet ADIPEX-P’s growth potential diminishes amid competition.
- Regulatory and legislative environments influence prescribing behaviors and market access.
- Revenue projections show a gradual decline, with revenues falling from $120 million in 2022 to approximately $108 million in 2026.
- Future growth depends on positioning within evolving treatment paradigms, including combination therapies and personalized medicine.
FAQs
Q1: What is the primary competitor to ADIPEX-P?
Semaglutide (Wegovy), approved in 2021, offers superior efficacy for weight loss with less abuse potential, capturing significant market share.
Q2: How does the Schedule IV classification affect ADIPEX-P sales?
It allows for prescription but imposes restrictions on prescribing quantities and refills, potentially limiting market volume.
Q3: Are there upcoming regulatory changes that could impact ADIPEX-P?
Potential increased scrutiny over controlled substances could further restrict prescribing, impacting sales volume.
Q4: Can ADIPEX-P be prescribed for long-term weight management?
FDA guidelines limit phentermine use to short-term (typically 12 weeks), restricting long-term revenue growth.
Q5: What are the key markets where ADIPEX-P remains competitive?
The U.S. leads in prescription volume, followed by Europe, with emerging opportunities in Asia, subject to regulatory approvals.
References
- World Health Organization. (2016). Obesity and overweight.
- U.S. Food and Drug Administration. (2023). Approval history for phentermine.
- MarketWatch. (2022). Global weight management drug market analysis.
- Statista. (2023). Prescription drug sales worldwide.
- U.S. Drug Enforcement Administration. (2024). Controlled Substances Scheduling.