Last updated: February 19, 2026
ACTOPLUS MET, a combination therapy for type 2 diabetes, demonstrates a stable market presence driven by its established efficacy and patient adherence profiles. Its financial trajectory is characterized by consistent revenue generation, influenced by patent exclusivity, market penetration, and the competitive landscape.
What is the Mechanism of Action for ACTOPLUS MET?
ACTOPLUS MET combines two active pharmaceutical ingredients: pioglitazone and metformin hydrochloride. Pioglitazone is a thiazolidinedione (TZD) that acts as an insulin sensitizer. It activates peroxisome proliferator-activated receptor gamma (PPARγ), a nuclear receptor that enhances insulin sensitivity in peripheral tissues, including adipose tissue and muscle. This activation leads to improved glucose uptake and utilization. Metformin hydrochloride is a biguanide that primarily reduces hepatic glucose production and decreases intestinal glucose absorption. It also enhances insulin sensitivity in peripheral tissues. The combination targets multiple pathways involved in type 2 diabetes pathogenesis, offering a synergistic effect for glycemic control.
What is the Regulatory Status and Patent Landscape of ACTOPLUS MET?
ACTOPLUS MET is approved by regulatory bodies including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for the treatment of type 2 diabetes mellitus in patients inadequately controlled on monotherapy with pioglitazone or metformin, or for patients already treated with both agents separately.
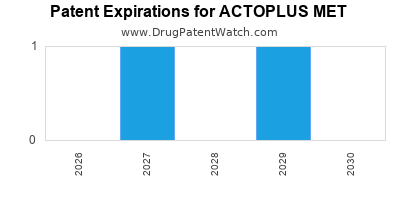
The patent landscape for ACTOPLUS MET is complex, primarily revolving around the combination therapy itself, specific formulations, and manufacturing processes. The primary patents protecting the individual active ingredients, pioglitazone and metformin, have expired in major markets, allowing for the availability of generic versions of these monotherapies. However, patents covering the specific fixed-dose combination formulation of ACTOPLUS MET, and potentially its manufacturing methods or specific polymorphs, have historically provided market exclusivity for the branded product.
For instance, U.S. Patent 6,391,926, which covers a method of treating diabetes with a specific combination of a thiazolidinedione and metformin, was a key patent. While the core patents for the individual drugs expired years ago, patents related to the fixed-dose combination and improved delivery systems can extend market exclusivity. The expiration dates of these specific combination patents are critical for understanding future generic competition. In the United States, the compound patent for pioglitazone expired in 2011, and for metformin in 2013. However, patents covering specific formulations or methods of use for the combination may have later expiration dates. For example, certain formulations of pioglitazone/metformin combinations have had patent protection extending into the mid-to-late 2020s in some regions.
What is the Market Size and Segmentation for ACTOPLUS MET?
The global market for type 2 diabetes medications is substantial, valued in the tens of billions of dollars annually. ACTOPLUS MET occupies a segment within the oral antidiabetic drug market, specifically for combination therapies. Its market size is influenced by several factors:
- Prevalence of Type 2 Diabetes: The increasing global incidence and prevalence of type 2 diabetes directly impact the demand for antidiabetic medications.
- Physician Prescribing Habits: The willingness of healthcare providers to prescribe combination therapies like ACTOPLUS MET versus individual agents or other classes of drugs.
- Patient Adherence: Fixed-dose combination pills often improve patient adherence by reducing pill burden, which can positively influence market share.
- Insurance Formulary Placement: Reimbursement policies and preferred drug lists by payers significantly affect accessibility and market penetration.
Market segmentation for ACTOPLUS MET can be viewed by:
- Geography: North America, Europe, and Asia-Pacific represent major markets, with varying levels of market penetration and competition from generics.
- Patient Demographics: Age, disease severity, and co-morbidities of the patient population influence prescribing patterns.
- Healthcare Setting: Prescription volumes in primary care, endocrinology clinics, and hospital settings.
Estimates for the oral antidiabetic drug market place it in the range of $30 billion to $40 billion globally in recent years, with combination therapies comprising a significant portion of this. While specific market share data for ACTOPLUS MET is proprietary, its position as a well-established combination therapy suggests a stable, albeit mature, market share within this broader segment.
What is the Competitive Landscape for ACTOPLUS MET?
ACTOPLUS MET faces competition from:
- Other Fixed-Dose Combination Therapies: Numerous other oral antidiabetic agents are available as fixed-dose combinations, including DPP-4 inhibitors (e.g., sitagliptin/metformin), SGLT2 inhibitors (e.g., empagliflozin/metformin), and other pioglitazone combinations.
- Monotherapies: Generic versions of pioglitazone and metformin are widely available and are often prescribed as individual agents, with patients potentially titrating doses separately.
- Newer Drug Classes: The emergence of newer antidiabetic drug classes such as GLP-1 receptor agonists and SGLT2 inhibitors, which offer additional benefits like weight loss and cardiovascular protection, represents a significant competitive threat. These newer agents are increasingly favored in treatment guidelines for patients with specific co-morbidities.
- Biosimil/Generic Competition: As patent expiries occur for combination patents, generic versions of ACTOPLUS MET become available, leading to price erosion and increased competition. The introduction of generic pioglitazone/metformin combinations has already impacted the market dynamics for the branded product.
The competitive landscape is dynamic, with ongoing development of novel therapies and shifting treatment guidelines. The efficacy and safety profile of ACTOPLUS MET, along with its established position in clinical practice, allows it to maintain a presence, but its market share growth is likely constrained by the availability of newer agents with broader benefits.
What are the Key Market Drivers and Restraints for ACTOPLUS MET?
Market Drivers:
- Growing Prevalence of Type 2 Diabetes: The global epidemic of obesity and metabolic syndrome continues to fuel the demand for effective diabetes management solutions.
- Cost-Effectiveness: Compared to some newer biologic therapies or combination therapies with novel mechanisms of action, established oral agents like pioglitazone/metformin combinations can offer a more cost-effective treatment option, particularly in cost-sensitive healthcare systems.
- Established Efficacy and Safety Profile: ACTOPLUS MET has a long track record of clinical use, with extensive data demonstrating its efficacy in lowering HbA1c. For patients who tolerate it well and do not have contraindications, it remains a viable treatment option.
- Improved Patient Adherence: The fixed-dose formulation simplifies treatment regimens, potentially leading to better adherence and thus better glycemic control compared to multiple monotherapy pills.
- Generic Availability of Monotherapies: While a driver for generic competition, the widespread availability and affordability of generic pioglitazone and metformin also means that physicians have the option to prescribe these individually, which can sometimes be favored for titration purposes.
Market Restraints:
- Emergence of Newer Drug Classes: SGLT2 inhibitors and GLP-1 receptor agonists have demonstrated cardiovascular and renal benefits, leading to their preferential use in guidelines for patients with specific risk factors, even as first-line or early-stage therapy.
- Side Effect Profile of Pioglitazone: Pioglitazone carries potential side effects, including fluid retention, weight gain, an increased risk of bone fractures in women, and a historical association with bladder cancer (though subsequent studies have shown conflicting results or no increased risk in some populations), which can limit its use.
- Metformin Contraindications: Metformin is contraindicated in patients with severe renal impairment, hepatic impairment, and certain cardiovascular conditions, restricting its use in a subset of the diabetic population.
- Generic Competition for Combination Products: As patents for the fixed-dose combination expire, generic versions enter the market, leading to significant price reductions and erosion of the branded product's market share and revenue.
- Shifting Treatment Paradigms: Newer treatment guidelines increasingly emphasize individualized therapy based on patient profiles, co-morbidities, and desired outcomes beyond glycemic control (e.g., cardiovascular risk reduction).
What is the Financial Trajectory and Revenue Outlook for ACTOPLUS MET?
The financial trajectory of ACTOPLUS MET is influenced by the interplay of patent expirations, generic competition, market penetration, and the broader market for oral antidiabetic drugs. For the branded product, the period following the expiry of key combination patents is typically characterized by a significant decline in revenue due to the introduction of lower-priced generic alternatives.
- Pre-Genericization: During its period of patent protection, the branded ACTOPLUS MET would have experienced strong revenue growth as it gained market share, driven by physician adoption and marketing efforts. Peak revenue would likely have been achieved in the years leading up to significant patent expirations.
- Post-Genericization: Following the entry of generics, the revenue of the branded product experiences a sharp and sustained decline. Generic manufacturers, with lower development costs and no patent royalties, can offer the same active ingredients at substantially lower prices. This forces the branded product to compete primarily on brand recognition, established patient base, and potentially through payer contracts that maintain some preferred status.
- Market Dynamics: The overall revenue generated by pioglitazone/metformin combinations in the market continues, but this revenue is increasingly distributed among multiple generic manufacturers. The total market value for this specific combination therapy may stabilize or grow modestly with the increasing prevalence of diabetes, but the revenue attributable to the original branded product will continue to diminish.
Financial projections for ACTOPLUS MET would therefore focus on the declining revenue stream of the branded product and the ongoing, albeit lower-margin, revenue generated by generic versions. The outlook is one of a mature product category facing intense generic competition, with growth potential limited by the availability of newer, more differentiated therapies.
Key Takeaways
ACTOPLUS MET is a well-established oral antidiabetic fixed-dose combination therapy with a stable mechanism of action. Its market presence is shaped by the increasing prevalence of type 2 diabetes, its cost-effectiveness compared to newer agents, and its ability to improve patient adherence. However, the competitive landscape is intense, dominated by other fixed-dose combinations, monotherapies, and increasingly by newer drug classes (GLP-1 RAs and SGLT2 inhibitors) that offer broader cardiovascular and renal benefits. The financial trajectory of the branded ACTOPLUS MET has been significantly impacted by patent expirations and the subsequent influx of generic competition, leading to a substantial decline in its revenue. The future revenue outlook for the combination therapy category, in general, is tied to the overall growth of the diabetes market, but the financial performance of individual branded products like ACTOPLUS MET is largely dictated by its stage in the patent lifecycle and the pricing pressures from generic alternatives.
Frequently Asked Questions
-
What is the primary indication for ACTOPLUS MET?
ACTOPLUS MET is indicated for the treatment of type 2 diabetes mellitus in patients inadequately controlled on monotherapy with pioglitazone or metformin, or for patients already treated with both agents separately.
-
When did the primary patents for pioglitazone and metformin expire in the U.S.?
The compound patent for pioglitazone expired in 2011, and for metformin expired in 2013 in the United States.
-
What are the main advantages of fixed-dose combination pills like ACTOPLUS MET?
Fixed-dose combination pills can improve patient adherence by reducing the number of pills to take, simplifying treatment regimens, and potentially leading to better overall glycemic control.
-
Which newer classes of diabetes medications pose a significant competitive threat to ACTOPLUS MET?
GLP-1 receptor agonists and SGLT2 inhibitors are significant competitive threats due to their demonstrated cardiovascular and renal benefits, which are increasingly prioritized in treatment guidelines.
-
What is the expected impact of generic competition on the revenue of branded ACTOPLUS MET?
Following the expiration of key patents for the fixed-dose combination, the revenue of branded ACTOPLUS MET is expected to experience a significant and sustained decline due to the introduction of lower-priced generic alternatives.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approval Database. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/drug-approvals-and-databases (Note: Specific patent information is often found through proprietary databases or specific FDA product pages, but this serves as a general source for regulatory status).
[2] European Medicines Agency. (n.d.). European public assessment reports (EPARs). Retrieved from https://www.ema.europa.eu/en/medicines/human/assessments (Note: Similar to the FDA, specific EPARs would detail drug approval, but this is a general portal).
[3] U.S. Patent 6,391,926. (2002). Method of treating diabetes with a thiazolidinedione and metformin. Retrieved from United States Patent and Trademark Office database.
[4] Global Diabetes Market Size & Share Analysis - Growth Trends & Forecasts to 2029. (2023). Mordor Intelligence. (Note: This is a representative example of a market research report providing industry size data. Specific reports for oral antidiabetics would be consulted).
[5] Diabetes Care 2022 Guidelines. (2022). American Diabetes Association. Retrieved from https://care.diabetesjournals.org/content/45/Supplement_1 (Note: Treatment guidelines are dynamic and would be consulted for current prescribing patterns).