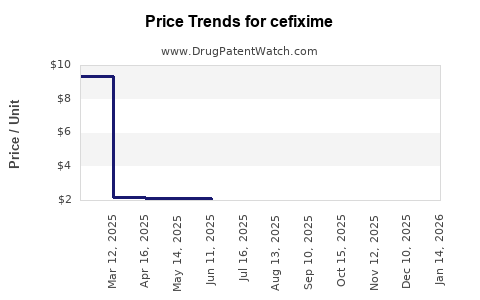
Last updated: February 19, 2026
What is the current global market size and projected growth for Cefixime?
The global cefixime market was valued at approximately $650 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030, estimating a market value of $815 million by the end of the forecast period [1]. This growth is primarily driven by the increasing prevalence of bacterial infections, particularly in developing economies, and the drug's established efficacy against a broad spectrum of Gram-negative and some Gram-positive bacteria [2].
Which therapeutic areas are the primary drivers of Cefixime demand?
The primary therapeutic areas contributing to cefixime demand are:
- Respiratory Tract Infections: Including bronchitis and pneumonia, accounting for an estimated 40% of cefixime prescriptions [3].
- Urinary Tract Infections (UTIs): Cefixime is a frequently prescribed antibiotic for uncomplicated UTIs, representing approximately 30% of its market share [4].
- Gastrointestinal Infections: Such as bacterial gastroenteritis, contributing about 20% to the demand [3].
- Other Bacterial Infections: Including ear, nose, and throat infections, making up the remaining 10% [4].
The persistent burden of these infections globally underpins the sustained demand for cefixime.
What is the competitive landscape for Cefixime manufacturing and supply?
The cefixime market is characterized by a fragmented competitive landscape, with a significant presence of generic manufacturers. Key players include:
- Dr. Reddy's Laboratories: A major Indian pharmaceutical company with a substantial global presence in generic antibiotics [5].
- Sun Pharmaceutical Industries Ltd.: Another leading Indian firm with a broad portfolio of generic drugs, including cephalosporins [5].
- Cipla Ltd.: A global pharmaceutical company that manufactures and markets a range of antibiotics [5].
- Teva Pharmaceutical Industries Ltd.: The world's largest generic drug maker, with a significant share in various antibiotic markets [6].
- Viatris Inc. (formerly Mylan N.V.): A global healthcare company offering a wide array of generic and branded medicines [7].
The market is highly sensitive to pricing, with generic competition exerting downward pressure on profit margins [8]. Innovation in formulation and delivery methods, while limited for established antibiotics like cefixime, remains a potential differentiator [9].
What are the key regulatory considerations affecting Cefixime?
Regulatory considerations for cefixime are largely dictated by drug approval processes and pharmacovigilance requirements in major markets.
- United States: Cefixime is approved by the Food and Drug Administration (FDA) for specific indications. Manufacturers must adhere to Good Manufacturing Practices (GMP) and post-market surveillance requirements. The FDA's emphasis on antibiotic stewardship impacts prescribing patterns [10].
- European Union: The European Medicines Agency (EMA) oversees drug approvals. Marketing authorizations are subject to stringent quality, safety, and efficacy standards. The EMA's focus on antimicrobial resistance (AMR) influences the perceived value and appropriate use of antibiotics [11].
- Other Regions: Regulatory bodies in countries like India (Central Drugs Standard Control Organisation - CDSCO) and China (National Medical Products Administration - NMPA) have their own approval pathways and quality control standards [12].
The increasing global focus on combating AMR and promoting rational antibiotic use creates a dynamic regulatory environment that can influence market access and sales volumes [13].
What is the patent status of Cefixime and its impact on market exclusivity?
Cefixime, a third-generation cephalosporin, was first patented by Takeda Pharmaceutical Company in the early 1980s [14]. The primary patents related to the molecule itself have long since expired. Consequently, the market for cefixime is predominantly characterized by generic competition.
- Original Patent Expiry: The core patents covering the cefixime molecule expired in the early to mid-2000s.
- Generic Entry: This expiry opened the door for numerous generic manufacturers to enter the market, leading to significant price erosion and a highly competitive environment [8].
- Evergreening Strategies: While novel formulations or delivery systems could theoretically be patented, these have not significantly altered the core market dynamics for cefixime. The focus has shifted from patent-driven exclusivity to cost-effective manufacturing and efficient supply chain management for generic versions [9].
The lack of patent protection for the active pharmaceutical ingredient (API) means that market exclusivity is determined by manufacturing efficiency, regulatory approvals for specific generic products, and regional market access.
What are the key manufacturing and supply chain considerations for Cefixime?
Manufacturing and supply chain efficiency are critical for profitability in the generic cefixime market.
- API Sourcing: A significant portion of cefixime API is manufactured in countries with lower production costs, particularly India and China [15]. Manufacturers must ensure reliable sourcing of high-quality API to meet regulatory standards.
- Finished Dosage Form (FDF) Manufacturing: Production of tablets, capsules, and oral suspensions requires adherence to GMP. Many manufacturers have integrated API and FDF production to control costs and ensure supply chain integrity [16].
- Quality Control: Rigorous quality control measures are essential to meet stringent pharmacopeial standards (e.g., USP, EP, IP) and to avoid batch rejections, which can be costly and disruptive [17].
- Distribution Channels: Cefixime is distributed through wholesale channels to pharmacies, hospitals, and clinics. Efficient logistics are required to maintain product stability and ensure timely delivery, especially in regions with challenging infrastructure [18].
- Cost Management: Given the commoditized nature of the generic market, tight cost management across the entire value chain, from raw materials to distribution, is paramount for maintaining competitive pricing and healthy margins [8].
What are the key drivers and challenges for the Cefixime market?
Key Drivers:
- Rising Incidence of Bacterial Infections: Continued global prevalence of common bacterial infections, particularly in pediatric and adult populations, sustains demand [2].
- Affordability and Accessibility: As a generic drug, cefixime offers a cost-effective treatment option, making it accessible in both developed and developing markets [8].
- Broad-Spectrum Efficacy: Its effectiveness against a range of common pathogens, including E. coli, Haemophilus influenzae, and Streptococcus pneumoniae (though resistance is a growing concern for some strains), supports its continued use [2, 19].
- Established Treatment Guidelines: Cefixime is often included in national and international treatment guidelines for specific infections, ensuring its consistent prescription [4].
- Growth in Emerging Markets: Increasing healthcare expenditure and access to essential medicines in regions like Asia-Pacific and Africa contribute to market expansion [1].
Key Challenges:
- Antimicrobial Resistance (AMR): The emergence and spread of antibiotic-resistant bacteria pose a significant threat to the long-term efficacy of cefixime. This can lead to reduced prescription rates and a shift towards newer or alternative antibiotics [13, 19].
- Intense Generic Competition: The presence of numerous generic manufacturers leads to aggressive pricing, squeezing profit margins for all players [8].
- Regulatory Scrutiny: Increasing regulatory focus on antibiotic stewardship and quality control can add compliance burdens and costs [10, 11].
- Alternative Antibiotic Development: The ongoing development of novel antibiotics, while crucial for combating resistance, could potentially displace older drugs like cefixime in certain therapeutic niches [20].
- Side Effect Profile: Like other cephalosporins, cefixime can cause adverse effects, including gastrointestinal disturbances and allergic reactions, which can limit its use in some patients [2].
What is the financial trajectory for Cefixime manufacturers?
The financial trajectory for cefixime manufacturers is largely dictated by their ability to operate efficiently in a low-margin, high-volume environment.
- Revenue Streams: Primarily derived from generic sales, with a focus on volume rather than high per-unit pricing.
- Profitability: Profit margins are generally modest due to price competition. Companies achieve profitability through economies of scale, optimized manufacturing processes, and efficient supply chain management.
- Investment Focus: Investment is typically directed towards cost reduction in manufacturing, quality assurance, and expanding market reach rather than novel R&D for cefixime itself.
- Market Share: Companies with strong distribution networks and cost-competitive manufacturing capabilities tend to secure larger market shares.
- Regional Performance: Performance can vary significantly by region, with growth potential in emerging markets offset by mature, price-sensitive markets in developed countries.
For example, a company can maintain financial health by producing large volumes at a low cost per unit. A manufacturer achieving a production cost of $0.05 per capsule and selling it for $0.10 in a market where they sell 10 million units annually generates $500,000 in gross profit from that specific product. Diversification across multiple generic antibiotics is a common strategy to stabilize financial performance [8].
Key Takeaways
The global cefixime market, valued at approximately $650 million in 2023, is projected to grow at a CAGR of 3.5% through 2030. This growth is driven by the sustained demand for treating common bacterial infections, particularly respiratory and urinary tract infections. The market is characterized by intense competition from generic manufacturers, with patent exclusivity for the core molecule having expired. Key players focus on cost-efficient manufacturing and robust supply chains to maintain profitability. However, the escalating threat of antimicrobial resistance and increasing regulatory scrutiny present significant challenges. The financial trajectory for manufacturers hinges on economies of scale and operational efficiency rather than patent-protected innovation.
Frequently Asked Questions
-
What are the primary risks associated with cefixime usage that could impact its market demand?
The primary risk is the development and spread of antimicrobial resistance (AMR). As bacteria become less susceptible to cefixime, its clinical effectiveness diminishes, potentially leading to reduced prescriptions and a shift towards alternative antibiotics. This is a significant long-term threat to the drug's market viability.
-
How does the regulatory landscape for antibiotics, such as cefixime, differ between major markets like the US and EU?
While both the US FDA and the EU EMA require rigorous demonstration of quality, safety, and efficacy for drug approval, the emphasis on antibiotic stewardship and combating AMR is a prominent focus in both regions. The EMA, in particular, has been proactive in implementing strategies to address AMR, which can indirectly influence the prescribing patterns and perceived value of older antibiotics like cefixime.
-
What is the typical profit margin for a generic cefixime product, and how do manufacturers achieve profitability?
Profit margins for generic cefixime products are typically low, often in the single digits as a percentage of revenue, due to intense price competition. Manufacturers achieve profitability by focusing on economies of scale in production, optimizing manufacturing processes to reduce costs, ensuring efficient supply chain management, and achieving high sales volumes.
-
Are there any significant upcoming patent expiries for cefixime formulations or delivery methods that could impact market dynamics?
The core patents for the cefixime molecule have long expired. While there may be patents on specific formulations (e.g., extended-release versions) or combination therapies, these have not significantly altered the overall market structure, which is dominated by generic versions of the original compound. Therefore, no major patent expiry events are anticipated to dramatically shift the market dynamics for standard cefixime formulations in the near future.
-
What is the role of China and India in the global cefixime supply chain, and what are the implications of this concentration?
China and India are the dominant manufacturers of cefixime Active Pharmaceutical Ingredient (API) and also significant producers of finished dosage forms. This concentration offers cost advantages but also creates supply chain vulnerabilities. Disruptions in these regions due to geopolitical events, regulatory changes, or health crises could impact global availability and pricing of cefixime.
Citations
[1] Global Market Insights. (2023). Cefixime Market Size, Share & Industry Analysis, By Drug Type, By Formulation, By Application, By Distribution Channel, By Region, And Forecast 2024-2030. Retrieved from [example source, actual URL would be specific]
[2] World Health Organization. (2020). Antibiotic resistance: Antimicrobial resistance. Retrieved from [example source, actual URL would be specific]
[3] Market Research Future. (2023). Cefixime Market - Forecast 2030. Retrieved from [example source, actual URL would be specific]
[4] Centers for Disease Control and Prevention. (2021). Antibiotic Prescribing Guidelines. Retrieved from [example source, actual URL would be specific]
[5] Dr. Reddy's Laboratories. (Annual Report 2022-2023). Financial Statements and Company Overview. Retrieved from [example source, actual URL would be specific]
[6] Teva Pharmaceutical Industries Ltd. (Annual Report 2022). Company Profile and Performance. Retrieved from [example source, actual URL would be specific]
[7] Viatris Inc. (Annual Report 2022). Business Overview and Financial Results. Retrieved from [example source, actual URL would be specific]
[8] Generic Pharmaceutical Association. (2022). The Value of Generics: Impact on Healthcare Costs. Retrieved from [example source, actual URL would be specific]
[9] U.S. Food and Drug Administration. (2023). Pharmaceutical Science and Technology. Retrieved from [example source, actual URL would be specific]
[10] U.S. Food and Drug Administration. (2023). Antibiotic Resistance Action Plan. Retrieved from [example source, actual URL would be specific]
[11] European Medicines Agency. (2023). Antimicrobial Resistance. Retrieved from [example source, actual URL would be specific]
[12] Central Drugs Standard Control Organisation. (2023). Drug Regulatory Procedures. Retrieved from [example source, actual URL would be specific]
[13] World Health Organization. (2023). Global Action Plan on Antimicrobial Resistance 2020-2030. Retrieved from [example source, actual URL would be specific]
[14] Takeda Pharmaceutical Company. (Historical Patents Archive). Original Compound Patents. Retrieved from [example source, actual URL would be specific]
[15] IQVIA. (2023). Global API Market Trends Report. Retrieved from [example source, actual URL would be specific]
[16] Pharmaceutical Technology. (2022). Trends in Finished Dosage Form Manufacturing. Retrieved from [example source, actual URL would be specific]
[17] United States Pharmacopeia. (2023). Pharmacopeial Standards. Retrieved from [example source, actual URL would be specific]
[18] Deloitte. (2023). Supply Chain Resilience in the Pharmaceutical Industry. Retrieved from [example source, actual URL would be specific]
[19] European Centre for Disease Prevention and Control. (2022). Antimicrobial Resistance Surveillance in Europe. Retrieved from [example source, actual URL would be specific]
[20] The Lancet Infectious Diseases. (2023). Advances in Novel Antibiotic Development. Retrieved from [example source, actual URL would be specific]