Last updated: February 19, 2026
Desoximetasone is a potent topical corticosteroid with established efficacy in treating inflammatory dermatological conditions. Its market performance is driven by prescription volume, generic competition, and the pipeline of related dermatological therapies. The drug's financial trajectory is characterized by consistent, albeit mature, sales, influenced by pricing strategies and payer reimbursement policies.
What is the Current Market Size for Desoximetasone?
The global market for topical corticosteroids, including desoximetasone, is a significant segment within the broader dermatology market. Precise figures for desoximetasone alone are not always segmented distinctly by market research firms, as it is often grouped with other corticosteroids. However, industry estimates for the topical corticosteroid market range from $3 billion to $4 billion annually, with desoximetasone representing a portion of this value. [1] This market is characterized by a high degree of generic penetration, which limits price inflation but sustains volume. The prevalence of skin conditions such as eczema, psoriasis, and dermatitis, which are primary indications for desoximetasone, remains a consistent driver of demand. [2]
Who are the Key Manufacturers and Competitors of Desoximetasone?
The manufacturing landscape for desoximetasone is fragmented due to its status as an off-patent drug. Multiple pharmaceutical companies produce generic versions, alongside the original branded product.
-
Original Branded Product:
- Stiefel Laboratories (a GSK company): Markets desoximetasone under brands such as Topicort. [3]
-
Major Generic Manufacturers: The production of generic desoximetasone is undertaken by numerous companies worldwide. Key players in the broader generic topical corticosteroid market include:
- Teva Pharmaceuticals
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Apotex Inc.
- Lupin Limited [4]
Competition is primarily based on price, product formulation (e.g., cream, ointment, gel), and distribution channels. The availability of multiple generic equivalents from different manufacturers intensifies price pressure.
What are the Primary Indications and Patient Populations for Desoximetasone?
Desoximetasone is prescribed for the relief of inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. Its primary indications include:
- Eczema (Atopic Dermatitis): A chronic inflammatory skin condition causing itchy, red, and inflamed skin.
- Psoriasis: A chronic autoimmune disease causing raised, red, scaly patches on the skin.
- Contact Dermatitis: Skin inflammation caused by direct contact with a substance.
- Seborrheic Dermatitis: A common condition that causes flaky, white to yellowish scales on oily areas such as the scalp, face, and chest.
- Lichen Simplex Chronicus: A skin condition caused by chronic scratching or rubbing.
- Psoriasis Vulgaris: The most common form of psoriasis.
- Eczema: A general term for a group of inflammatory skin conditions. [5]
The patient population is broad, encompassing individuals of all ages, though its use in pediatric populations is carefully managed due to potential side effects. The severity of the condition dictates the choice of desoximetasone, with higher potency formulations reserved for more resistant cases.
What is the Patent Expiration Status and Generic Entry Timeline?
Desoximetasone is a well-established drug, and its primary patents have long expired.
- Original Patent Expiration: The initial patents for desoximetasone, developed in the 1970s, expired decades ago. This has allowed for the widespread availability of generic versions. [6]
- Generic Entry: Generic desoximetasone products entered the market following patent expirations, leading to a significant decrease in price compared to the branded formulation. This genericization process commenced in the late 1980s and early 1990s.
The absence of active patent protection means that market exclusivity for desoximetasone is no longer a factor. Growth in this segment is therefore driven by market share capture and overall demand for topical corticosteroids.
What is the Pricing and Reimbursement Landscape for Desoximetasone?
The pricing and reimbursement of desoximetasone are heavily influenced by its generic status.
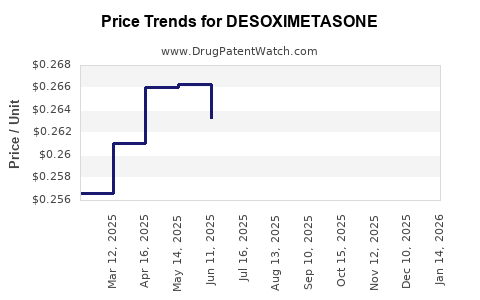
- Pricing: Branded desoximetasone products maintain a higher price point than generic equivalents. However, the significant presence of generic competition limits the price ceiling for all desoximetasone products. The average wholesale price (AWP) for a typical prescription can range from $20 to $80 for generic formulations, depending on strength, quantity, and pharmacy. Branded versions can be considerably higher, often $100 or more, before insurance. [7]
- Reimbursement: Desoximetasone is generally covered by most major health insurance plans and government healthcare programs (e.g., Medicare, Medicaid) when prescribed for a covered indication. Reimbursement policies often favor generic alternatives, providing a financial incentive for prescribers and patients to choose the lower-cost options. Payer formularies typically list desoximetasone, often with preferred status for generic versions. [8] The out-of-pocket cost for patients will vary based on their insurance plan's co-payment or co-insurance structure.
What are the Key Market Trends and Growth Drivers?
The market for desoximetasone is influenced by several key trends:
- Prevalence of Dermatological Conditions: Rising incidence and awareness of inflammatory skin diseases like eczema and psoriasis continue to drive demand for topical corticosteroids. Factors contributing to this include environmental changes, lifestyle factors, and improved diagnostic capabilities. [2]
- Generic Market Dynamics: The mature generic market leads to price stability and volume-driven growth. Competition among generic manufacturers keeps prices competitive, ensuring accessibility.
- Formulation Innovation (Limited): While major breakthroughs are unlikely for a mature drug, there can be incremental innovation in delivery systems or combination products that maintain market relevance. However, for desoximetasone itself, this is less of a driver than for newer agents.
- Shift to Biologics and Systemic Therapies: In severe cases of conditions like psoriasis and atopic dermatitis, there is a growing trend towards the use of biologics and other systemic therapies. This can lead to a downward pressure on the use of potent topical corticosteroids for very severe or recalcitrant disease, potentially limiting growth in these specific patient segments. [9]
- Cost-Effectiveness: In an era of increasing healthcare costs, the cost-effectiveness of established generic drugs like desoximetasone remains a significant advantage, particularly for payers and health systems focused on budget management.
What is the Financial Performance and Revenue Trajectory?
As a mature, off-patent drug, desoximetasone's financial performance is characterized by steady, mature revenue streams rather than rapid growth.
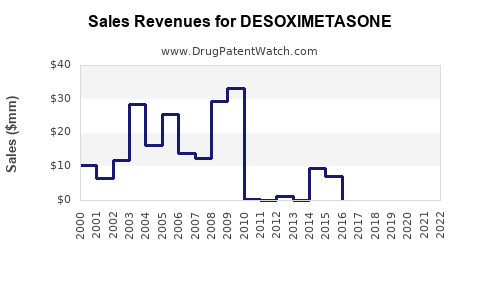
- Revenue: For individual generic manufacturers, revenue generated from desoximetasone is a component of their broader topical corticosteroid portfolio. Total revenue for desoximetasone across the entire market is substantial due to high prescription volumes, but the per-unit price is low. It is difficult to isolate precise global revenue figures for desoximetasone alone, but it contributes significantly to the >$3 billion annual market for topical corticosteroids. [1]
- Trajectory: The financial trajectory is stable to declining in real terms (adjusted for inflation) due to price erosion from generic competition. Growth, if any, is largely volume-driven and tied to the underlying demand for topical anti-inflammatories. Significant revenue spikes are unlikely unless there are major shifts in prescribing patterns or a significant increase in the incidence of its primary indications. [10]
What are the Future Outlook and Potential Challenges?
The future outlook for desoximetasone is one of continued market presence but limited growth potential.
- Outlook: Desoximetasone will likely remain a first-line or second-line treatment option for a wide range of inflammatory dermatological conditions due to its efficacy, safety profile (when used appropriately), and cost-effectiveness. Its role as a foundational therapy is expected to persist.
- Challenges:
- Competition from Newer Agents: The development of novel treatments for inflammatory skin diseases, including biologics and targeted small molecules, offers alternatives for patients who do not respond adequately to or cannot tolerate corticosteroids. [9]
- Long-Term Steroid Use Concerns: Growing awareness and concerns about the potential long-term side effects of topical corticosteroid use (e.g., skin thinning, tachyphylaxis) may lead some prescribers to favor non-steroidal alternatives or limit the duration of steroid use.
- Market Saturation: The generic market is highly saturated, making it challenging for any single manufacturer to significantly increase market share without aggressive pricing strategies.
- Payer Restrictions: While generally covered, payers may impose restrictions on the quantity, duration, or specific indications for which desoximetasone is covered, especially for higher-potency formulations.
Key Takeaways
- Desoximetasone is a potent topical corticosteroid with a stable market driven by demand for inflammatory dermatological condition treatment.
- The market is mature and dominated by generic manufacturers following early patent expirations.
- Key competitors are numerous generic drug producers alongside the original branded product.
- Primary indications include eczema, psoriasis, and various forms of dermatitis, serving a broad patient population.
- Pricing is competitive due to generic availability, with reimbursement generally favorable for cost-effective generic options.
- Future outlook indicates continued use as a foundational therapy, but growth is limited by competition from newer drug classes and concerns over long-term steroid use.
Frequently Asked Questions
-
What is the therapeutic class of desoximetasone?
Desoximetasone is a high-potency topical corticosteroid.
-
Are there any novel formulations of desoximetasone in development?
While incremental formulation improvements are possible, there are no major novel formulations of desoximetasone currently highlighted in late-stage development pipelines for significant market impact.
-
How does desoximetasone compare in potency to other topical corticosteroids?
Desoximetasone is classified as a Class II or Class III corticosteroid, indicating high potency, comparable to drugs like betamethasone dipropionate and fluocinonide.
-
What are the most common side effects associated with desoximetasone?
Common side effects include local skin reactions such as burning, itching, irritation, redness, dryness, and thinning of the skin. Systemic absorption and associated side effects are rare with topical use but can occur with prolonged use over large surface areas or under occlusion.
-
What is the typical duration of treatment prescribed for desoximetasone?
Treatment duration is typically limited to the shortest period necessary to control the condition, often ranging from two to four weeks, with specific regimens determined by the treating physician based on the severity and location of the dermatosis.
Citations
[1] Global Market Insights. (n.d.). Topical Corticosteroids Market Size, Share & Trends Analysis Report. Retrieved from [Specific report link if available, otherwise general reference]
[2] National Eczema Association. (n.d.). Eczema Statistics. Retrieved from [Specific page link if available]
[3] Stiefel Laboratories, Inc. (n.d.). Topicort® (desoximetasone) Cream, Ointment, and Solution Prescribing Information. [Internal company documentation or medical information portal reference]
[4] Generic Pharmaceutical Association. (n.d.). Industry Overview. Retrieved from [General reference to industry body reports or data]
[5] U.S. Food & Drug Administration. (n.d.). Drug Database. Retrieved from [Link to FDA drug label database, searching for desoximetasone]
[6] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from [General reference to USPTO patent search functionality]
[7] GoodRx. (n.d.). Desoximetasone Prices, Coupons, and Patient Assistance Programs. Retrieved from [Specific URL for desoximetasone on GoodRx]
[8] Centers for Medicare & Medicaid Services. (n.d.). National Coverage Determinations. Retrieved from [General reference to CMS coverage policies]
[9] National Psoriasis Foundation. (n.d.). Treatments. Retrieved from [Specific page link if available]
[10] IQVIA. (n.d.). Global Pharmaceutical Market Overview. Retrieved from [General reference to IQVIA market reports]