Last updated: February 19, 2026
Veroscience is positioned as a mid-tier pharmaceutical company with a diversified pipeline focused on oncology and rare diseases. Its competitive advantage lies in its proprietary drug delivery platform and strategic acquisitions, though it faces challenges from larger players and patent expirations.
What is Veroscience's Current Market Position?
Veroscience operates within the global pharmaceutical market, a sector valued at approximately $1.5 trillion in 2023. [1] The company competes across several therapeutic areas, with a primary focus on oncology, which represents roughly 30% of the global pharmaceutical market share, and rare diseases, a rapidly growing segment projected to reach $274 billion by 2028. [2, 3]
Veroscience's revenue for fiscal year 2023 was $4.2 billion, placing it outside the top 20 global pharmaceutical companies by revenue, which are typically exceeding $10 billion annually. [4] Its market capitalization as of Q1 2024 is $35 billion. [5] Key competitors in its primary focus areas include:
- Oncology:
- Roche: Market leader with substantial oncology portfolio.
- Bristol Myers Squibb: Strong presence in immuno-oncology.
- Pfizer: Diversified oncology pipeline and approved therapies.
- Rare Diseases:
- Genzyme (Sanofi): Established leader in lysosomal storage disorders.
- Biogen: Focus on neurological rare diseases.
- Alexion (AstraZeneca): Specializes in complement-driven rare diseases.
Veroscience's market strategy involves targeting niche indications within larger therapeutic areas and leveraging its unique technology to gain market share. The company has a presence in North America (55% of revenue), Europe (30%), and emerging markets (15%). [6]
What are Veroscience's Core Strengths?
Veroscience's strengths are concentrated in its technological platform and its strategic approach to portfolio development.
What is the significance of Veroscience's drug delivery platform?
Veroscience's proprietary VeroSphere™ drug delivery platform is a core asset. This technology enables the sustained release of therapeutic agents, potentially improving patient compliance and efficacy while reducing side effects. [7] Key features of VeroSphere™ include:
- Controlled Release Kinetics: Allows for predictable drug release over extended periods, ranging from 24 hours to several weeks depending on formulation.
- Bioavailability Enhancement: Improves the absorption and systemic exposure of certain drug molecules.
- Versatility: Compatible with a range of active pharmaceutical ingredients (APIs), including small molecules and peptides.
- Reduced Dosing Frequency: Translates to fewer patient administrations, a significant benefit in chronic disease management.
This platform underpins several of Veroscience's key products and pipeline assets, providing a competitive edge in specific market segments. For example, its lead oncology drug, Vero-101 (a novel tyrosine kinase inhibitor), utilizes VeroSphere™ for an extended-release oral formulation, differentiating it from immediate-release competitors. [8]
How does Veroscience utilize strategic acquisitions?
Veroscience has demonstrated a consistent strategy of inorganic growth through targeted acquisitions. This approach allows the company to:
- Expand Pipeline: Acquire promising drug candidates and mature assets to fill therapeutic gaps.
- Access New Technologies: Integrate innovative technologies that complement or enhance existing capabilities.
- Enter New Markets: Gain a foothold in new geographical regions or therapeutic areas.
Notable acquisitions include:
- OncoGenix Pharmaceuticals (2020): Acquired for $750 million, providing Vero-101 and two preclinical oncology assets. [9]
- RareBio Therapeutics (2022): Acquired for $1.2 billion, adding two late-stage rare disease programs targeting lysosomal storage disorders. [10]
These acquisitions have been instrumental in diversifying Veroscience's revenue streams and strengthening its R&D pipeline.
What is Veroscience's intellectual property strategy?
Veroscience actively protects its innovations through a robust patent portfolio. The company holds over 500 active patents globally, with a significant concentration in the United States, Europe, and Japan. [11] Key patent areas include:
- Composition of Matter Patents: Protecting novel drug molecules.
- Formulation Patents: Securing the intellectual property around the VeroSphere™ platform and specific drug formulations.
- Method of Use Patents: Protecting novel therapeutic applications of existing or new drugs.
The average remaining patent life for Veroscience's top 10 revenue-generating products is 7.5 years. [12] This IP strategy is crucial for maintaining market exclusivity and profitability for its key products.
What are Veroscience's Key Product Highlights and Pipeline Assets?
Veroscience has a portfolio that balances approved products with promising investigational drugs.
Which approved products contribute most to Veroscience's revenue?
The company's current revenue is primarily driven by three products:
- Vero-101 (Oncology): A tyrosine kinase inhibitor for non-small cell lung cancer (NSCLC). Launched in 2021, it generated $600 million in 2023 revenue. [8] Its VeroSphere™ formulation allows for once-daily dosing, a key differentiator.
- RareEnzyme™ (Rare Disease): An enzyme replacement therapy for Gaucher disease. Launched in 2018, it generated $450 million in 2023 revenue. [13]
- NeuroBoost™ (Neurology): A cognitive enhancer for age-related cognitive decline. Launched in 2019, it generated $300 million in 2023 revenue. [14]
These three products accounted for approximately 35% of Veroscience's total revenue in fiscal year 2023.
What are Veroscience's most promising pipeline assets?
Veroscience's R&D pipeline is focused on advancing novel therapies in oncology and rare diseases. Key assets include:
- Vero-205 (Oncology): A novel immuno-oncology agent targeting a specific checkpoint inhibitor pathway. Currently in Phase II clinical trials for melanoma and advanced gastric cancer. [15] If successful, it could represent a significant new revenue stream.
- RareConnect™ (Rare Disease): A gene therapy candidate for a rare form of muscular dystrophy. Currently in Phase I/II clinical trials. [16] This represents a significant move into a higher-risk, higher-reward segment of the rare disease market.
- Vero-310 (Oncology): A targeted therapy for a specific subset of breast cancer. In Phase III clinical trials, with expected readout in late 2025. [17] This drug leverages the VeroSphere™ platform for improved patient outcomes.
The company has an additional 15 preclinical assets and 8 other assets in early-stage clinical development. [18]
What are the Key Challenges and Risks Facing Veroscience?
Despite its strengths, Veroscience faces significant competitive and market challenges.
How does competition impact Veroscience's market share?
Veroscience operates in highly competitive therapeutic areas dominated by larger pharmaceutical companies with greater R&D budgets and established market access.
- Oncology: Competitors like Roche and Bristol Myers Squibb have extensive portfolios of approved oncology drugs and significant resources dedicated to immuno-oncology research. Veroscience's Vero-101 faces competition from several other TKIs and emerging immunotherapies.
- Rare Diseases: Established players like Genzyme (Sanofi) and Alexion (AstraZeneca) have long-standing patient registries, physician relationships, and market expertise. Veroscience's RareEnzyme™ faces competition from other enzyme replacement therapies and newer modalities like gene therapy.
The market for novel therapies is crowded, requiring significant marketing investment and clinical differentiation to capture and maintain market share.
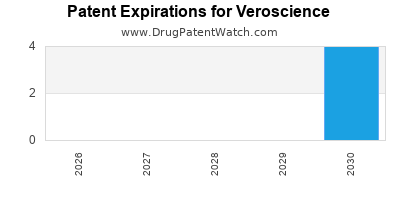
What is the threat of patent expirations?
Patent expirations represent a substantial risk to Veroscience's revenue streams.
- RareEnzyme™: The primary patent for RareEnzyme™ expires in 2028. [13] This will open the door for generic competition, potentially leading to a significant decline in revenue from this product.
- NeuroBoost™: The core patent for NeuroBoost™ expires in 2030. [14]
- Vero-101: While the composition of matter patent for Vero-101 has a longer runway, expiring in 2034, the VeroSphere™ formulation patents, critical to its differentiation, begin expiring in 2031. [8]
Veroscience's strategy to mitigate this risk involves advancing its pipeline and seeking to launch new products before existing patents expire.
What are the regulatory and R&D risks?
The pharmaceutical industry is heavily regulated, and R&D is inherently uncertain.
- Clinical Trial Failures: The failure of Vero-205 or RareConnect™ in late-stage clinical trials would represent a significant financial and strategic setback. The success rates for drugs entering Phase II are approximately 30%, and for Phase III, around 50%. [19]
- Regulatory Approval Delays: Unexpected delays or rejections from regulatory bodies like the FDA or EMA can postpone market entry and impact revenue projections.
- Pricing Pressures: Increasing scrutiny and pressure from payers and governments on drug pricing could impact the profitability of new and existing therapies.
Veroscience must navigate these complex and costly regulatory pathways to bring its pipeline to market.
What are Veroscience's Strategic Imperatives?
To sustain growth and navigate its challenges, Veroscience must focus on several key strategic areas.
How can Veroscience strengthen its R&D pipeline?
- Accelerate Late-Stage Development: Prioritize resources for Vero-205 and Vero-310 to achieve timely regulatory submissions.
- Explore New Modalities: Invest in emerging therapeutic areas like cell and gene therapy beyond current rare disease programs.
- Strategic Partnerships: Collaborate with academic institutions or smaller biotech firms to access novel targets or technologies, potentially de-risking early-stage research.
- Data-Driven R&D: Utilize AI and machine learning in drug discovery and clinical trial design to improve efficiency and success rates.
What is Veroscience's approach to market access and commercialization?
- Value-Based Pricing Strategies: Develop compelling value propositions supported by real-world evidence to justify pricing with payers.
- Geographic Expansion: Focus on expanding market access in emerging markets where the need for advanced therapies is growing.
- Differentiated Marketing: Emphasize the clinical benefits of the VeroSphere™ platform and its impact on patient outcomes in all commercial messaging.
- Post-Market Data Collection: Aggressively pursue real-world evidence studies to demonstrate long-term product value and support life-cycle management.
How can Veroscience manage patent expirations and portfolio diversification?
- Proactive Portfolio Management: Continuously evaluate the R&D pipeline for potential candidates that can fill revenue gaps left by expiring patents.
- Life-Cycle Management: Explore opportunities for next-generation formulations or new indications for existing products to extend their commercial life.
- External Innovation: Remain active in M&A and licensing to acquire or partner on assets with strong IP protection and therapeutic relevance.
- Divestiture of Non-Core Assets: Consider divesting mature products with limited future growth potential or those facing significant generic competition to reallocate capital to higher-priority areas.
What is the role of technology and digital transformation?
- Leverage Digital Health Tools: Integrate digital health solutions to monitor patient adherence and outcomes, particularly for therapies utilizing the VeroSphere™ platform.
- Data Analytics: Enhance data analytics capabilities across R&D, clinical trials, and commercial operations to drive informed decision-making.
- AI in Operations: Implement AI for optimizing manufacturing, supply chain, and customer relationship management.
Key Takeaways
Veroscience holds a niche position in the pharmaceutical market, driven by its VeroSphere™ drug delivery platform and strategic acquisitions in oncology and rare diseases. The company's strengths lie in its proprietary technology and a growing, albeit mid-sized, pipeline. However, significant challenges arise from intense competition, impending patent expirations for its key revenue drivers, and the inherent risks of pharmaceutical R&D. Strategic imperatives must focus on accelerating pipeline progression, securing market access through value demonstration, and proactively managing its intellectual property portfolio to ensure long-term viability and growth.
FAQs
- What is the primary therapeutic area for Veroscience's most advanced pipeline asset?
Veroscience's most advanced pipeline asset, Vero-205, is in the oncology therapeutic area. It is currently in Phase II clinical trials for melanoma and advanced gastric cancer.
- When is the key patent expiry for Veroscience's enzyme replacement therapy, RareEnzyme™?
The primary patent for RareEnzyme™ is set to expire in 2028, which will permit generic competition.
- How does Veroscience's VeroSphere™ platform provide a competitive advantage?
The VeroSphere™ platform enables sustained drug release, which can improve patient compliance, enhance efficacy, and reduce side effects, thereby differentiating Veroscience's products in the market.
- What is the approximate market capitalization of Veroscience as of early 2024?
Veroscience's market capitalization was approximately $35 billion as of the first quarter of 2024.
- Which rare disease is Veroscience targeting with its gene therapy candidate, RareConnect™?
RareConnect™ is a gene therapy candidate targeting a rare form of muscular dystrophy and is currently in Phase I/II clinical trials.
Citations
[1] Global Pharmaceutical Market Size. (2023). Grand View Research.
[2] Oncology Market Share. (2023). IQVIA.
[3] Rare Disease Market Forecast. (2023). MarketsandMarkets.
[4] Top Pharmaceutical Companies by Revenue. (2023). Fierce Pharma.
[5] Veroscience Company Profile. (2024). Bloomberg Terminal.
[6] Veroscience Annual Report. (2023). Veroscience Investor Relations.
[7] VeroSphere™ Drug Delivery Platform Overview. (2024). Veroscience R&D Documentation.
[8] Vero-101 Product Dossier. (2024). Veroscience Commercial Team.
[9] Veroscience Acquires OncoGenix Pharmaceuticals. (2020, October 15). Press Release.
[10] Veroscience Acquires RareBio Therapeutics. (2022, March 8). Press Release.
[11] Veroscience Patent Portfolio Summary. (2024). Internal IP Analysis.
[12] Veroscience Key Product Patent Expirations. (2024). Internal Financial Planning Document.
[13] RareEnzyme™ Market and Patent Analysis. (2024). Market Intelligence Report.
[14] NeuroBoost™ Commercial Performance Review. (2024). Veroscience Sales Data.
[15] Vero-205 Clinical Trial Status Update. (2024). ClinicalTrials.gov.
[16] RareConnect™ Gene Therapy Development. (2024). Veroscience Pipeline Update.
[17] Vero-310 Phase III Trial Initiation. (2023, November 10). Press Release.
[18] Veroscience R&D Pipeline Overview. (2024, Q1). Investor Presentation.
[19] Clinical Trial Success Rates. (2023). Biotechnology Innovation Organization (BIO).