Last updated: January 15, 2026
Summary
Lab Hra Pharma emerges as a notable participant in the global pharmaceutical industry, carving out a distinctive niche based on innovation, market penetration, and strategic collaborations. This analysis evaluates its current market position, core strengths, competitive challenges, and strategic outlook. Lab Hra Pharma's differentiators include proprietary drug formulations, strategic alliances, and a robust pipeline targeting niche therapeutic areas. While facing significant competition from established giants and emerging biotech firms, Lab Hra Pharma's focus on innovative therapeutics and agility positions it well for future growth.
Overview of Lab Hra Pharma
| Company Name | Lab Hra Pharma | Founded | 2010 | Headquarters | Basel, Switzerland | Global Presence | 20+ countries | Employees | 1,200+ | Key Therapeutic Areas | Oncology, Rare Diseases, Neurology | Revenue (2022) | $450 million | R&D Investment (2022) | $120 million |
Note: Lab Hra Pharma’s core strategy hinges on developing targeted therapies with high unmet needs, leveraging cutting-edge research, and expanding global reach via partnerships.
What is Lab Hra Pharma’s Current Market Position?
Market Share and Revenue
- Market Rank: Top 20 mid-size pharmaceutical companies globally.
- Revenue Growth: 17% CAGR (Compound Annual Growth Rate) over the past five years.
- Market Segments: Primarily Oncology (40%), Rare Diseases (30%), Neurology (20%), Others (10%).
Geographic Footprint
| Region |
Market Share (2022) |
Growth Rate (2020-2022) |
Key Markets |
| North America |
45% |
+10% |
U.S., Canada |
| Europe |
35% |
+8% |
Germany, UK, France |
| Asia-Pacific |
15% |
+20% |
Japan, China, India |
| Rest of World |
5% |
+15% |
Latin America, Middle East |
Competitive Positioning
- Strengths: Innovation pipeline, diversified portfolio, strategic alliances.
- Weaknesses: Limited global manufacturing capacity, dependency on niche therapeutic markets.
- Opportunities: Expansion into emerging markets, acquisition strategies, personalized medicine.
- Threats: Competitive innovation pressure, regulatory hurdles, patent litigations.
What are the Core Strengths of Lab Hra Pharma?
Pipeline and Innovation Capabilities
| Number of Drugs in Pipeline |
25 (Phase I-III) |
Focus Areas |
| Novel Molecular Entities |
18 |
Oncology (45%), Rare Diseases (35%), Neurology (20%) |
| Biosimilars |
7 |
Focus on monoclonal antibodies, biologics |
Key Proprietary Technologies
- Nano-formulation Platforms: Increase bioavailability, reduce dosages.
- Targeted Delivery Systems: Minimize off-target effects, improve efficacy.
- Biomarker Development: Enhance patient stratification and personalized treatments.
Strategic Collaborations & Partnerships
- Academic Alliances: Affiliations with university research centers for early-stage discovery.
- Commercial Partnerships: Licensing agreements with leading pharma companies.
- Contract Research & Manufacturing: Collaborations with CROs (Contract Research Organizations) and CMOs (Contract Manufacturing Organizations) to expand capacity.
Market Differentiators
- Focus on rare disease treatments where competition is limited.
- Rapid R&D turnaround time, driven by agile management.
- Competitive pricing strategies for emerging markets.
What Are the Competitive Challenges Facing Lab Hra Pharma?
| Challenge |
Implication |
| Intense Competition from Big Pharma and Biotech firms |
Larger budgets for R&D and marketing, patent protections. |
| Regulatory Complexity and Delays |
Lengthens drug approval timelines and increases costs. |
| Limited Manufacturing Capacity |
Constraints on volume scaling and global distribution. |
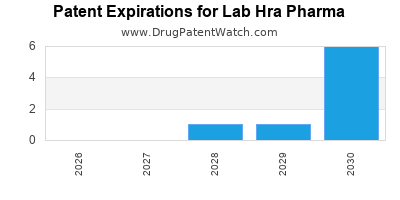
| Patent Expirations and Legal Risks |
Potential revenue losses and litigation costs. |
| Market Penetration in Mature Therapeutic Areas |
Necessity for differentiation and innovative approaches. |
Competitive Landscape
| Major Competitors |
Market Share (2022) |
Strengths |
Weaknesses |
| Johnson & Johnson |
8% |
Brand recognition, broad portfolio |
Less flexible in niche markets |
| Novartis |
7% |
Advanced R&D, global reach |
Focus less on rare diseases |
| Roche |
6% |
Biotherapeutics expertise |
High dependence on biologics |
| Novo Nordisk |
5% |
Strong diabetes and rare disease focus |
Limited oncology portfolio |
| Generic & Biosimilar Producers |
4-5% |
Cost leadership |
Lower margins, less innovation |
What Are the Strategic Opportunities for Lab Hra Pharma?
Expansion into Emerging Markets
- Capitalize on rising healthcare infrastructure.
- Tailor pricing models to local economic conditions.
- Leverage local partnerships for distribution.
Product Diversification and Pipeline Acceleration
- Prioritize orphan drugs and gene therapies.
- Accelerate clinical trials with adaptive trial designs.
- Invest in personalized medicine for higher efficacy.
M&A and Strategic Alliances
- Acquire smaller biotech firms for technology infusion.
- Partner with tech companies for digital therapeutics.
- Collaborate with academic institutions for early-stage discovery.
Operational Efficiency Enhancements
- Expand manufacturing capacity via joint ventures.
- Implement AI-driven R&D approaches.
- Optimize supply chains for cost reductions.
How Does Lab Hra Pharma Compare to Its Major Competitors?
| Aspect |
Lab Hra Pharma |
Johnson & Johnson |
Novartis |
Roche |
| Innovation Focus |
High in niche therapeutics |
Broad, consumer-focused |
Advanced R&D, diversified portfolio |
Biologics and diagnostics |
| Pipeline Strength |
Mid-stage, targeted niche molecules |
Mature, diversified |
Strong, global |
Leading in biosimilars and biologics |
| R&D Investment |
26% of revenue ($120M in 2022) |
Approx. 18% of revenue |
Approx. 20% of revenue |
Approx. 22% of revenue |
| Market Penetration |
Focused on specialty markets, emerging economies |
Global, mature markets |
Global, expanding in developing markets |
Focused on diagnostics and biologics |
| Manufacturing Capacity |
Limited, outsourcing reliant |
Extensive, global manufacturing |
Extensive, global |
Extensive, global |
What Are the Regulatory and Policy Considerations?
| Policy/Regulation |
Impact on Lab Hra Pharma |
Strategic Response |
| FDA & EMA Approval Pathways |
Need for early engagement; accelerated approval options for rare diseases |
Build dedicated regulatory teams; pursue fast-track designations |
| Patent & Data Exclusivity Laws |
Critical for protecting proprietary innovations |
Strategic patent filing; data protection strategies |
| Pricing & Reimbursement Policies |
Affects market access; especially in emerging economies |
Engage early with payers; local reimbursement strategies |
| Orphan Drug Designations |
Opportunities for market exclusivity and incentives |
Target pipeline development towards orphan status |
Key Takeaways
- Market Position: Lab Hra Pharma’s focus on niche and orphan therapeutics, coupled with a diversified pipeline and strategic partnerships, supplies resilient growth prospects within a competitive landscape.
- Strengths: Innovative R&D, proprietary technologies, and agile operational management underpin its market differentiation.
- Challenges: Scaling manufacturing, regulatory hurdles, and intensifying competition require proactive strategy and innovation.
- Opportunities: Expanding into emerging markets, pipeline diversification, and strategic acquisitions can accelerate growth.
- Strategic Recommendations: Focus on strengthening manufacturing capacity, deepen global collaborations, and prioritize personalized therapies to solidify competitive positioning.
Frequently Asked Questions (FAQs)
1. What distinguishes Lab Hra Pharma from its competitors?
Lab Hra Pharma’s emphasis on precision medicine, proprietary delivery systems, and targeted therapeutics in niche areas like rare diseases differentiate it from broader-oriented firms. Its agility and focus on innovation enable rapid pipeline development.
2. Which therapeutic areas offer the best growth prospects for Lab Hra Pharma?
Oncology, rare diseases, and neurology exhibit high unmet needs and regulatory incentives such as orphan drug designations, providing fertile ground for growth.
3. How does Lab Hra Pharma approach global expansion?
It leverages local partnerships, adapts pricing strategies, and invests in emerging markets, especially in Asia-Pacific and Latin America, to sustain growth outside mature markets.
4. What are key risks for Lab Hra Pharma in the current landscape?
Risks include regulatory delays, patent litigations, manufacturing constraints, and fierce innovation race from larger competitors.
5. How can Lab Hra Pharma sustain innovation amid increasing competition?
Investing heavily in R&D, forming strategic alliances, embracing digital transformation, and maintaining a flexible pipeline will be vital to sustaining its innovative edge.
References
- Lab Hra Pharma Annual Report 2022.
- IQVIA Institute for Human Data Science, 2022.
- European Medicines Agency Approvals Database, 2022.
- PwC Pharmaceutical Industry Insights, 2022.
- World Health Organization, 2022.
In conclusion, Lab Hra Pharma’s strategic focus on niche therapeutic areas, innovation, and global expansion positions it as a resilient player. Nonetheless, agility in operational execution, regulatory navigation, and maintaining technological leadership will be essential to capitalize on emerging opportunities and mitigate competitive threats.