Fresenius Kabi Usa Company Profile
✉ Email this page to a colleague
What is the competitive landscape for FRESENIUS KABI USA, and when can generic versions of FRESENIUS KABI USA drugs launch?
FRESENIUS KABI USA has three hundred and three approved drugs.
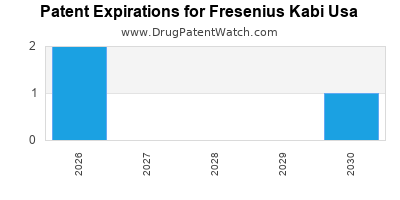
There are twenty-three US patents protecting FRESENIUS KABI USA drugs. There are two tentative approvals on FRESENIUS KABI USA drugs.
There are one hundred and twenty-nine patent family members on FRESENIUS KABI USA drugs in twenty-five countries and four hundred and sixty-six supplementary protection certificates in seventeen countries.
Summary for Fresenius Kabi Usa
| International Patents: | 129 |
| US Patents: | 23 |
| Tradenames: | 253 |
| Ingredients: | 187 |
| NDAs: | 303 |
| Patent Litigation for Fresenius Kabi Usa: | See patent lawsuits for Fresenius Kabi Usa |
| PTAB Cases with Fresenius Kabi Usa as petitioner: | See PTAB cases with Fresenius Kabi Usa as petitioner |
| PTAB Cases with Fresenius Kabi Usa as patent owner: | See PTAB cases with Fresenius Kabi Usa as patent owner |
Drugs and US Patents for Fresenius Kabi Usa
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fresenius Kabi Usa | MORPHINE SULFATE | morphine sulfate | SOLUTION;INTRAMUSCULAR, INTRAVENOUS | 204223-005 | Oct 30, 2013 | RX | Yes | Yes | 9,072,781 | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Fresenius Kabi Usa | HEPARIN SODIUM 25,000 UNITS IN SODIUM CHLORIDE 0.45% | heparin sodium | INJECTABLE;INJECTION | 017029-025 | Aug 24, 2017 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | |||||
| Fresenius Kabi Usa | CEFOTETAN | cefotetan disodium | INJECTABLE;INJECTION | 065375-001 | Aug 9, 2007 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Fresenius Kabi Usa
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Fresenius Kabi Usa | NAROPIN | ropivacaine hydrochloride | SOLUTION;INJECTION | 020533-005 | Sep 24, 1996 | 5,670,524 | ⤷ Sign Up |
| Fresenius Kabi Usa | NAROPIN | ropivacaine hydrochloride | SOLUTION;INJECTION | 020533-005 | Sep 24, 1996 | 5,834,489 | ⤷ Sign Up |
| Fresenius Kabi Usa | DIPRIVAN | propofol | INJECTABLE;INJECTION | 019627-002 | Jun 11, 1996 | 5,731,356*PED | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for FRESENIUS KABI USA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Injection | 1 mg/mL, 50 mL vials | ➤ Subscribe | 2011-12-16 |
| ➤ Subscribe | Injection | 2 mg/mL | ➤ Subscribe | 2011-06-22 |
| ➤ Subscribe | Injection | 10 mg/mL | ➤ Subscribe | 2011-11-04 |
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
| ➤ Subscribe | Injection | 2 mg/mL, 100 mL | ➤ Subscribe | 2015-01-30 |
| ➤ Subscribe | Injection | 100 mg/mL, 2.5 mL vials | ➤ Subscribe | 2007-09-24 |
| ➤ Subscribe | Oral Solution | 5 mg/5mL | ➤ Subscribe | 2011-02-25 |
| ➤ Subscribe | Tablets | 2 mg, 4 mg, and 8 mg | ➤ Subscribe | 2013-08-05 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
| ➤ Subscribe | Injection | 2 mg/mL, 5 mg/mL and 10 mg/mL, 20 mL, 30 mL and 20 mL vials | ➤ Subscribe | 2006-11-13 |
| ➤ Subscribe | Injection | 2 mg/mL, 200 mL | ➤ Subscribe | 2015-09-03 |
International Patents for Fresenius Kabi Usa Drugs
Supplementary Protection Certificates for Fresenius Kabi Usa Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0984957 | C300483 | Netherlands | ⤷ Sign Up | PRODUCT NAME: NAXOPREN EN ESOMEPRAZOL ALS MAGNESIUMTRIHYFRAAT; NAT. REGISTRATION NO/DATE: RVG 106235 20101118; FIRST REGISTRATION: PL 17091/0263 - 0001 20110524 |
| 0748228 | 11C0002 | France | ⤷ Sign Up | PRODUCT NAME: MANNITOL; NAT. REGISTRATION NO/DATE: NL 34557 20080819; FIRST REGISTRATION: SE - 22708 20061020 |
| 3141251 | SPC/GB20/075 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: A MEDICINAL PRODUCT CONSISTING OF A COMBINATION OF A FIRST DOSE PHARMACEUTICAL COMPOSITION AND A SECOND DOSE PHARMACEUTICAL COMPOSITION, THE FIRST DOSE PHARMACEUTICAL COMPOSITION CONSISTING OF THE ACTIVE INGREDIENTS: POLYETHYLENE GLYCOL 3350, SODIUM SULPH; REGISTERED: IS IS/1/17/063/01 20171016; UK PL 20011/0040 20171016 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.