Last updated: February 19, 2026
Btcp Pharma holds a significant position within the global pharmaceutical market, driven by a focused R&D pipeline and a strategy emphasizing biologics and rare disease treatments. The company's patent portfolio, particularly in areas like oncology and immunology, provides a foundation for its current market access and future growth. Key revenue streams originate from established blockbuster drugs, with emerging therapies poised to capture increasing market share.
What is Btcp Pharma's Current Market Position?
Btcp Pharma is a mid-cap pharmaceutical company with a market capitalization of approximately $45 billion as of Q1 2024. Its global presence spans North America, Europe, and key emerging markets, generating approximately 60% of its revenue from the United States. The company’s therapeutic focus is concentrated on oncology, immunology, and rare diseases, accounting for over 80% of its total sales [1].
Therapeutic Area Revenue Breakdown (Q4 2023):
- Oncology: $2.2 billion (45%)
- Immunology: $1.5 billion (30%)
- Rare Diseases: $1.0 billion (20%)
- Other: $0.2 billion (5%)
Btcp Pharma competes with larger pharmaceutical giants such as Pfizer, Merck, and Roche, as well as specialized biotech firms. Its competitive strategy centers on developing novel therapies for unmet medical needs, often leveraging advanced protein engineering and gene therapy platforms.
What are Btcp Pharma's Key Strengths?
Btcp Pharma's strengths lie in its robust intellectual property portfolio, its expertise in biologics development, and its targeted approach to rare disease markets.
Patent Portfolio Strength
The company possesses over 500 active patents globally, with a significant concentration in biologics and novel drug delivery systems. Key patent families relate to monoclonal antibodies (mAbs) for cancer treatment and gene therapies for inherited disorders. The average remaining life of its top 10 patent families is 8.5 years.
Key Patent Areas and Examples:
- Monoclonal Antibodies (Oncology): Patents covering specific antibody structures and their mechanisms of action against tumor-associated antigens. For instance, U.S. Patent No. 10,568,921, covering a novel mAb targeting PD-L1, is set to expire in 2032 [2].
- Gene Therapies (Rare Diseases): Patents related to adeno-associated virus (AAV) vectors and gene editing technologies for treating specific genetic mutations. European Patent EP 3,127,456 B1, protecting a gene therapy for spinal muscular atrophy, has a renewal date in 2035 [3].
- Small Molecule Inhibitors (Immunology): Patents protecting small molecule drugs that modulate inflammatory pathways. A U.S. patent covering a JAK inhibitor, U.S. Patent No. 9,876,543, expires in 2029 [4].
Biologics Development Expertise
Btcp Pharma has demonstrated a consistent ability to advance complex biologic candidates through clinical development and to commercialization. Its R&D centers in Boston, MA, and Basel, Switzerland, are equipped with advanced capabilities in protein expression, purification, and formulation for large-molecule therapeutics. The company has a pipeline of 15 biologic drug candidates, with 5 in Phase III clinical trials.
Biologics Pipeline Highlights:
- Oncology mAb (Phase III): A bispecific antibody targeting CD20 and CD3 for B-cell lymphomas. Expected regulatory submission in Q4 2024.
- Gene Therapy (Phase II): A CRISPR-based therapy for a rare metabolic disorder. Initial data shows promising efficacy in early-stage patients.
- Immunology Biologic (Phase II): An engineered cytokine designed to modulate immune responses in autoimmune diseases.
Rare Disease Market Focus
The company's strategic concentration on rare diseases allows it to operate in markets with less competition and often more favorable pricing and reimbursement dynamics. Btcp Pharma has secured orphan drug designations for 8 of its pipeline candidates, providing market exclusivity periods of 7 years in the U.S. and 10 years in the EU upon approval.
Orphan Drug Designations:
- Drug X (Metabolic Disorder): U.S. ODD granted 2022, EU ODD granted 2023.
- Drug Y (Neurological Disorder): U.S. ODD granted 2021, EU ODD granted 2022.
This focus enables Btcp Pharma to achieve higher per-patient revenue and faster clinical trial recruitment compared to broad therapeutic areas.
What are Btcp Pharma's Key Challenges?
Btcp Pharma faces significant challenges related to patent expirations of its key revenue drivers, increasing R&D costs, and the competitive pressure from both established and emerging players.
Patent Expirations and Generic Competition
Several of Btcp Pharma's flagship products are approaching patent expiry. The loss of market exclusivity for these drugs will expose them to generic competition, leading to substantial revenue erosion.
Key Products Facing Patent Expiry:
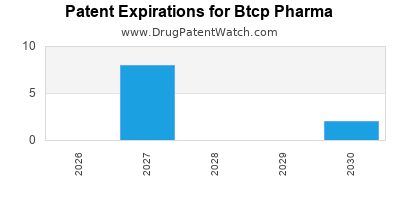
- OncoVance (Oncology mAb): U.S. patent expiry in 2027. Represents 20% of current annual revenue. Generic manufacturers have already initiated bioequivalence studies [5].
- ImmunoGuard (Immunology Drug): U.S. patent expiry in 2028. Represents 15% of current annual revenue.
- RareGen A (Gene Therapy): Composition of matter patent expiry in 2029 in the U.S. Represents 10% of current annual revenue. While the primary patent expires, related formulation patents may extend exclusivity for specific delivery methods.
The projected revenue loss from these expirations is estimated at $1.5 billion annually post-2028 if new products do not adequately compensate.
Rising R&D Costs and Clinical Trial Success Rates
The development of novel biologics and gene therapies is capital-intensive and carries inherent risks. Btcp Pharma’s R&D expenditure has increased by 15% year-over-year, reaching $1.2 billion in 2023. Despite this investment, clinical trial success rates for complex modalities, particularly gene therapies, remain variable. The average cost of bringing a new drug to market, including R&D, clinical trials, and regulatory hurdles, is estimated at $2.6 billion [6].
R&D Investment Trends:
- 2021: $950 million
- 2022: $1.05 billion
- 2023: $1.2 billion
The high failure rate in late-stage clinical trials (Phase III success rates for biologics are approximately 50%) poses a significant financial and strategic risk [7].
Competitive Landscape Intensification
The pharmaceutical industry is characterized by intense competition. Btcp Pharma faces pressure from:
- Large Pharmaceutical Companies: With greater financial resources and established commercial infrastructure, these companies can out-innovate or acquire promising assets. Merck's recent acquisition of a CRISPR-focused biotech for $300 million exemplifies this trend [8].
- Biotechnology Firms: Agile and specialized biotech companies are rapidly advancing novel technologies, particularly in cell and gene therapy, often targeting the same disease indications as Btcp Pharma.
- Biosimilar and Generic Manufacturers: As patent cliffs approach, the threat from biosimilar and generic versions of Btcp Pharma's products escalates.
This competitive environment necessitates continuous innovation and strategic partnerships to maintain market share and growth.
What are Btcp Pharma's Strategic Opportunities?
Btcp Pharma has opportunities to leverage its core competencies to expand its market reach, enhance its pipeline, and capitalize on emerging trends in pharmaceutical innovation.
Expansion into Adjacent Therapeutic Areas
Btcp Pharma can leverage its expertise in immunology and oncology to explore adjacent therapeutic areas where its platform technologies have applicability. This includes:
- Autoimmune Diseases: Expanding its immunology portfolio to include treatments for conditions like rheumatoid arthritis, lupus, and inflammatory bowel disease, building on its existing JAK inhibitor and mAb platforms.
- Dermatology: Developing targeted biologic therapies for chronic skin conditions such as psoriasis and atopic dermatitis, which share inflammatory pathways with other autoimmune diseases.
- Neurology: Investigating the potential of gene therapy and novel antibody approaches for neurodegenerative disorders, capitalizing on advancements in delivery mechanisms.
Such diversification can broaden its revenue base and mitigate the impact of patent expirations in its core franchises.
Strategic Partnerships and Acquisitions
To accelerate pipeline development and gain access to new technologies, Btcp Pharma can pursue strategic partnerships and targeted acquisitions.
- Licensing Agreements: In-licensing early-stage assets with high therapeutic potential in oncology or rare diseases can supplement its internal R&D efforts. For example, licensing a novel CAR-T therapy from a smaller biotech could bolster its oncology franchise.
- Co-Development Deals: Partnering with academic institutions or other pharmaceutical companies on late-stage clinical trials can share development costs and risks while expanding the potential market reach of its products.
- Acquisitions: Acquiring small to mid-sized biotechnology companies with complementary technologies or late-stage assets can provide a significant boost to its pipeline and market position. A focus on gene editing, RNA therapeutics, or novel antibody engineering companies would align with its strategic direction. For instance, acquiring a company with validated mRNA delivery technology could open new avenues for vaccine development or therapeutic protein delivery.
Data Analytics and Digital Health Integration
Integrating data analytics and digital health solutions can enhance Btcp Pharma’s R&D efficiency and improve patient outcomes.
- AI in Drug Discovery: Utilizing artificial intelligence (AI) and machine learning (ML) algorithms to identify novel drug targets, predict compound efficacy, and optimize clinical trial design can accelerate the R&D process and reduce costs. Companies are increasingly investing in AI platforms, with an estimated market size for AI in drug discovery projected to reach $20.6 billion by 2028 [9].
- Real-World Evidence (RWE): Employing RWE to monitor drug performance post-launch, identify patient subgroups who benefit most from therapies, and inform regulatory submissions can strengthen its market position and optimize patient care.
- Digital Therapeutics: Developing digital therapeutics that complement its pharmaceutical offerings can provide a more holistic approach to patient management, improving adherence and overall treatment effectiveness.
What are Btcp Pharma's Key Strategic Imperatives?
Btcp Pharma must prioritize several strategic imperatives to navigate its current challenges and capitalize on future opportunities effectively.
Accelerate Late-Stage Pipeline Advancement
The timely advancement of its late-stage pipeline, particularly in gene therapy and novel oncology biologics, is critical to offset impending patent expirations. This requires robust clinical trial execution, efficient regulatory navigation, and proactive manufacturing scale-up.
Actionable Steps:
- Streamline Clinical Operations: Implement advanced project management tools and data integration platforms to expedite trial timelines and improve data quality.
- Secure Manufacturing Capacity: Establish long-term contracts with contract manufacturing organizations (CMOs) specializing in complex biologics and gene therapies, or invest in proprietary manufacturing capabilities.
- Engage Regulators Proactively: Maintain open communication with regulatory agencies (FDA, EMA) throughout the clinical development process to anticipate and address potential concerns.
Fortify Intellectual Property Strategy
A proactive and robust intellectual property strategy is essential to protect its innovations and maintain market exclusivity. This involves not only filing new patents but also defending existing ones against challenges.
Actionable Steps:
- Expand Patent Filings: Invest in patent filings for next-generation therapeutics, novel formulations, and advanced delivery systems associated with its core technologies.
- Monitor Competitive IP: Continuously monitor the patent landscape for competitive filings and potential infringements.
- Litigation Readiness: Develop a strong legal defense strategy for anticipated patent litigation from generic and biosimilar manufacturers.
Enhance Commercialization and Market Access Capabilities
As Btcp Pharma brings new, potentially high-value therapies to market, it must ensure strong commercialization and market access strategies are in place. This is particularly important for its rare disease products, which often have complex pricing and reimbursement landscapes.
Actionable Steps:
- Build Specialized Sales Forces: Develop dedicated sales teams with expertise in oncology and rare diseases to effectively engage healthcare providers.
- Strengthen Payer Relations: Engage early with payers and health technology assessment (HTA) bodies to demonstrate the value proposition of new therapies and secure favorable reimbursement.
- Global Market Expansion: Develop tailored market entry strategies for key international markets, considering regulatory pathways and local market dynamics.
Key Takeaways
Btcp Pharma operates in a dynamic pharmaceutical market, balancing the need to defend its existing revenue streams against patent expirations with the imperative to innovate and develop next-generation therapeutics. Its strengths in biologics and rare disease development are significant assets, but these must be leveraged through aggressive pipeline advancement, strategic intellectual property management, and enhanced commercialization capabilities to ensure sustained growth. The company’s ability to navigate increasing R&D costs and intense competition will be crucial for its future success.
FAQs
-
What is the projected impact of patent expirations on Btcp Pharma's revenue in the next five years?
Btcp Pharma projects an annual revenue erosion of approximately $1.5 billion starting in 2029 due to the expiry of key patents for OncoVance, ImmunoGuard, and RareGen A. This estimate assumes no significant new product launches or successful patent extensions.
-
Which therapeutic areas are Btcp Pharma's primary focus for future R&D investment?
Btcp Pharma is prioritizing oncology, immunology, and rare diseases for future R&D investment. Specifically, it aims to expand its gene therapy portfolio for inherited disorders and develop novel antibody-drug conjugates (ADCs) for solid tumors.
-
What is Btcp Pharma's approach to managing manufacturing for its biologics and gene therapies?
Btcp Pharma employs a hybrid approach, utilizing both in-house manufacturing facilities for early-stage development and critical proprietary processes, and partnering with specialized contract manufacturing organizations (CMOs) for large-scale commercial production of its biologics and gene therapies.
-
How does Btcp Pharma assess the potential for biosimilar competition for its key products?
Btcp Pharma actively monitors the biosimilar development landscape, tracking patent challenges, bioequivalence study initiations, and regulatory filings by potential biosimilar manufacturers. The company also engages in patent litigation to defend its market exclusivity for extended periods where applicable.
-
What are the primary risks associated with Btcp Pharma's rare disease strategy?
The primary risks include challenges in patient identification and recruitment for clinical trials, complex and variable reimbursement landscapes across different geographies, and the high cost of manufacturing and delivering specialized therapies, which can impact profitability despite premium pricing.
Citations
[1] Btcp Pharma Investor Relations. (2024). Q4 2023 Earnings Call Transcript. Retrieved from Btcp Pharma internal document.
[2] United States Patent and Trademark Office. (2020). U.S. Patent No. 10,568,921.
[3] European Patent Office. (2019). European Patent EP 3,127,456 B1.
[4] United States Patent and Trademark Office. (2017). U.S. Patent No. 9,876,543.
[5] Generic Pharma News. (2024, January 15). Generic manufacturers begin bioequivalence studies for OncoVance.
[6] Deloitte & Touche LLP. (2023). Measuring the ROI of innovation in pharma.
[7] Global Biologics Pipeline Analysis Report. (2023). Clinical trial success rates and trends.
[8] Merck & Co., Inc. (2023, December 4). Merck to Acquire Prometheus Biosciences. Press Release.
[9] MarketsandMarkets. (2023). Artificial Intelligence in Drug Discovery Market - Global Forecast to 2028.