Amicus Therap Us Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AMICUS THERAP US
AMICUS THERAP US has two approved drugs.
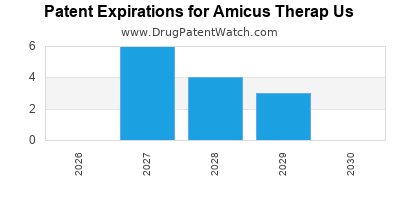
There are seventy-three US patents protecting AMICUS THERAP US drugs.
There are four hundred and twenty-nine patent family members on AMICUS THERAP US drugs in forty-five countries and thirty-three supplementary protection certificates in eighteen countries.
Summary for Amicus Therap Us
| International Patents: | 429 |
| US Patents: | 73 |
| Tradenames: | 2 |
| Ingredients: | 2 |
| NDAs: | 2 |
Drugs and US Patents for Amicus Therap Us
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 11,813,255 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 11,903,938 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 11,612,593 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 11,278,538 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 11,633,388 | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Amicus Therap Us Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| European Patent Office | 4541418 | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2008134628 | ⤷ Start Trial |
| South Korea | 102631805 | ⤷ Start Trial |
| European Patent Office | 2150254 | ⤷ Start Trial |
| Taiwan | I843172 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Amicus Therap Us Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3201320 | CA 2024 00012 | Denmark | ⤷ Start Trial | PRODUCT NAME: CIPAGLUCOSIDASE ALFA; REG. NO/DATE: EU/1/22/1714 20230324 |
| 2787345 | 634 | Finland | ⤷ Start Trial | |
| 2787345 | 201640042 | Slovenia | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT OR ITS SALT, INCLUDING HYDROCHLORIDE SALT; NATIONAL AUTHORISATION NUMBER: EU/1/15/1082/001; DATE OF NATIONAL AUTHORISATION: 20160526; AUTHORITY FOR NATIONAL AUTHORISATION: EU |
| 2787345 | 1690052-4 | Sweden | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT OR A SALT THEREOF, INCLUDING THE HYDROCHLORIDE SALT.; REG. NO/DATE: EU/1/15/1082 20160531 |
| 2787345 | 300843 | Netherlands | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT OF EEN ZOUT DAARVAN, WAARONDER HET HYDROCHLORIDEZOUT; REGISTRATION NO/DATE: EU/1/15/1082 20160531 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.