Japan: These 55 Drugs Face Patent Expirations and Generic Entry From 2024 - 2025
DrugPatentWatch® Estimated Loss of Exclusivity Dates in Japan
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "Japan: These 55 Drugs Face Patent Expirations and Generic Entry From 2024 - 2025" DrugPatentWatch.com thinkBiotech, 2024 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can VIZIMPRO (dacomitinib) generic drug versions launch?
Generic name: dacomitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 06, 2024
Generic Entry Controlled by: Japan Patent 2,007,536,368
VIZIMPRO is a drug marketed by Pfizer. There are four patents protecting this drug.
This drug has ninety-three patent family members in forty-eight countries.
See drug price trends for VIZIMPRO.
The generic ingredient in VIZIMPRO is dacomitinib. Two suppliers are listed for this generic product. Additional details are available on the dacomitinib profile page.
When can VIZIMPRO (dacomitinib) generic drug versions launch?
Generic name: dacomitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 06, 2024
Generic Entry Controlled by: Japan Patent 2,009,007,363
Patent Title: 4-PHENYLAMINO-QUINAZOLIN-6-YL-AMIDE
VIZIMPRO is a drug marketed by Pfizer. There are four patents protecting this drug.
This drug has ninety-three patent family members in forty-eight countries.
See drug price trends for VIZIMPRO.
The generic ingredient in VIZIMPRO is dacomitinib. Two suppliers are listed for this generic product. Additional details are available on the dacomitinib profile page.
When can VIZIMPRO (dacomitinib) generic drug versions launch?
Generic name: dacomitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 06, 2024
Generic Entry Controlled by: Japan Patent 4,205,757
VIZIMPRO is a drug marketed by Pfizer. There are four patents protecting this drug.
This drug has ninety-three patent family members in forty-eight countries.
See drug price trends for VIZIMPRO.
The generic ingredient in VIZIMPRO is dacomitinib. Two suppliers are listed for this generic product. Additional details are available on the dacomitinib profile page.
When can VIZIMPRO (dacomitinib) generic drug versions launch?
Generic name: dacomitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 06, 2024
Generic Entry Controlled by: Japan Patent 4,966,923
VIZIMPRO is a drug marketed by Pfizer. There are four patents protecting this drug.
This drug has ninety-three patent family members in forty-eight countries.
See drug price trends for VIZIMPRO.
The generic ingredient in VIZIMPRO is dacomitinib. Two suppliers are listed for this generic product. Additional details are available on the dacomitinib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Japan Patent 2,007,537,265
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Japan Patent 2,007,538,081
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Japan Patent 2,008,522,981
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Japan Patent 2,012,121,895
Patent Title: COMPOUND FOR ENZYME INHIBITION
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Japan Patent 2,014,141,456
Patent Title: COMPOUND FOR ENZYME INHIBITION
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Japan Patent 4,990,155
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: Japan Patent 5,108,509
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can ARCAPTA NEOHALER (indacaterol maleate) generic drug versions launch?
Generic name: indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: Japan Patent 2,007,536,962

This drug has eighty-six patent family members in thirty-nine countries.
See drug price trends for ARCAPTA NEOHALER.
The generic ingredient in ARCAPTA NEOHALER is indacaterol maleate. Additional details are available on the indacaterol maleate profile page.
When can ARCAPTA NEOHALER (indacaterol maleate) generic drug versions launch?
Generic name: indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: Japan Patent 2,012,071,146
Patent Title: INHALER

This drug has eighty-six patent family members in thirty-nine countries.
See drug price trends for ARCAPTA NEOHALER.
The generic ingredient in ARCAPTA NEOHALER is indacaterol maleate. Additional details are available on the indacaterol maleate profile page.
When can ARCAPTA NEOHALER (indacaterol maleate) generic drug versions launch?
Generic name: indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: Japan Patent 4,914,345

This drug has eighty-six patent family members in thirty-nine countries.
See drug price trends for ARCAPTA NEOHALER.
The generic ingredient in ARCAPTA NEOHALER is indacaterol maleate. Additional details are available on the indacaterol maleate profile page.
When can UTIBRON (glycopyrrolate ; indacaterol maleate) generic drug versions launch?
Generic name: glycopyrrolate ; indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: Japan Patent 2,007,536,962
UTIBRON is a drug marketed by Novartis. There are three patents protecting this drug.
This drug has one hundred and forty-eight patent family members in forty countries. There has been litigation on patents covering UTIBRON
See drug price trends for UTIBRON.
The generic ingredient in UTIBRON is glycopyrrolate ; indacaterol maleate. There are seventeen drug master file entries for this API. Additional details are available on the glycopyrrolate ; indacaterol maleate profile page.
When can UTIBRON (glycopyrrolate ; indacaterol maleate) generic drug versions launch?
Generic name: glycopyrrolate ; indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: Japan Patent 2,012,071,146
Patent Title: INHALER
UTIBRON is a drug marketed by Novartis. There are three patents protecting this drug.
This drug has one hundred and forty-eight patent family members in forty countries. There has been litigation on patents covering UTIBRON
See drug price trends for UTIBRON.
The generic ingredient in UTIBRON is glycopyrrolate ; indacaterol maleate. There are seventeen drug master file entries for this API. Additional details are available on the glycopyrrolate ; indacaterol maleate profile page.
When can UTIBRON (glycopyrrolate ; indacaterol maleate) generic drug versions launch?
Generic name: glycopyrrolate ; indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: Japan Patent 4,914,345
UTIBRON is a drug marketed by Novartis. There are three patents protecting this drug.
This drug has one hundred and forty-eight patent family members in forty countries. There has been litigation on patents covering UTIBRON
See drug price trends for UTIBRON.
The generic ingredient in UTIBRON is glycopyrrolate ; indacaterol maleate. There are seventeen drug master file entries for this API. Additional details are available on the glycopyrrolate ; indacaterol maleate profile page.
When can VITEKTA (elvitegravir) generic drug versions launch?
Generic name: elvitegravir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 20, 2024
Generic Entry Controlled by: Japan Patent 2,006,001,927
Patent Title: STABLE CRYSTAL FORM OF 4-OXOQUINOLINE COMPOUND
VITEKTA is a drug marketed by Gilead Sciences Inc. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries. There has been litigation on patents covering VITEKTA
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this API. Additional details are available on the elvitegravir profile page.
When can VITEKTA (elvitegravir) generic drug versions launch?
Generic name: elvitegravir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 20, 2024
Generic Entry Controlled by: Japan Patent 3,754,064
VITEKTA is a drug marketed by Gilead Sciences Inc. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries. There has been litigation on patents covering VITEKTA
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this API. Additional details are available on the elvitegravir profile page.
When can CAPRELSA (vandetanib) generic drug versions launch?
Generic name: vandetanib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 21, 2024
Generic Entry Controlled by: Japan Patent 2,007,538,060
CAPRELSA is a drug marketed by Genzyme Corp. There is one patent protecting this drug.
This drug has forty patent family members in thirty-three countries.
See drug price trends for CAPRELSA.
The generic ingredient in CAPRELSA is vandetanib. One supplier is listed for this generic product. Additional details are available on the vandetanib profile page.
When can CAPRELSA (vandetanib) generic drug versions launch?
Generic name: vandetanib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 21, 2024
Generic Entry Controlled by: Japan Patent 4,740,947
CAPRELSA is a drug marketed by Genzyme Corp. There is one patent protecting this drug.
This drug has forty patent family members in thirty-three countries.
See drug price trends for CAPRELSA.
The generic ingredient in CAPRELSA is vandetanib. One supplier is listed for this generic product. Additional details are available on the vandetanib profile page.
When can KOMBIGLYZE XR (metformin hydrochloride; saxagliptin hydrochloride) generic drug versions launch?
Generic name: metformin hydrochloride; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Japan Patent 2,008,501,025

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering KOMBIGLYZE XR
See drug price trends for KOMBIGLYZE XR.
The generic ingredient in KOMBIGLYZE XR is metformin hydrochloride; saxagliptin hydrochloride. There are forty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the metformin hydrochloride; saxagliptin hydrochloride profile page.
When can KOMBIGLYZE XR (metformin hydrochloride; saxagliptin hydrochloride) generic drug versions launch?
Generic name: metformin hydrochloride; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Japan Patent 4,901,727

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering KOMBIGLYZE XR
See drug price trends for KOMBIGLYZE XR.
The generic ingredient in KOMBIGLYZE XR is metformin hydrochloride; saxagliptin hydrochloride. There are forty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the metformin hydrochloride; saxagliptin hydrochloride profile page.
When can ONGLYZA (saxagliptin hydrochloride) generic drug versions launch?
Generic name: saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Japan Patent 2,008,501,025

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering ONGLYZA
See drug price trends for ONGLYZA.
The generic ingredient in ONGLYZA is saxagliptin hydrochloride. There are fifteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the saxagliptin hydrochloride profile page.
When can ONGLYZA (saxagliptin hydrochloride) generic drug versions launch?
Generic name: saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: Japan Patent 4,901,727

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering ONGLYZA
See drug price trends for ONGLYZA.
The generic ingredient in ONGLYZA is saxagliptin hydrochloride. There are fifteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the saxagliptin hydrochloride profile page.
When can FOLOTYN (pralatrexate) generic drug versions launch?
Generic name: pralatrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 30, 2024
Generic Entry Controlled by: Japan Patent 2,008,501,038
FOLOTYN is a drug marketed by Acrotech Biopharma. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-one patent family members in twenty-three countries. There has been litigation on patents covering FOLOTYN
See drug price trends for FOLOTYN.
The generic ingredient in FOLOTYN is pralatrexate. There are two drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pralatrexate profile page.
When can FOLOTYN (pralatrexate) generic drug versions launch?
Generic name: pralatrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 30, 2024
Generic Entry Controlled by: Japan Patent 5,005,532
FOLOTYN is a drug marketed by Acrotech Biopharma. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-one patent family members in twenty-three countries. There has been litigation on patents covering FOLOTYN
See drug price trends for FOLOTYN.
The generic ingredient in FOLOTYN is pralatrexate. There are two drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pralatrexate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 2,008,501,715
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 2,013,056,923
Patent Title: INTERMEDIATES FOR PREPARATION OF ANALOGS OF HALICHONDRIN B
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 2,015,061,845
Patent Title: ハリコンドリンB類似体の調製用中間体 (INTERMEDIATES FOR PREPARATION OF ANALOGS OF HALICHONDRIN B)
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 2,016,183,171
Patent Title: ハリコンドリンB類似体の調製用中間体 (INTERMEDIATES FOR PREPARATION OF HALICHONDRIN B ANALOGS)
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 2,017,141,241
Patent Title: ハリコンドリンB類似体の調製用中間体 (INTERMEDIATES FOR PREPARATION OF HALICHONDRIN B ANALOGS)
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 5,226,304
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 5,705,814
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 5,964,926
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 6,138,316
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: Japan Patent 6,306,235
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can BYVALSON (nebivolol hydrochloride; valsartan) generic drug versions launch?
Generic name: nebivolol hydrochloride; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 04, 2024
Generic Entry Controlled by: Japan Patent 2,008,545,742

This drug has thirty-seven patent family members in fifteen countries. There has been litigation on patents covering BYVALSON
See drug price trends for BYVALSON.
The generic ingredient in BYVALSON is nebivolol hydrochloride; valsartan. There are fourteen drug master file entries for this API. Additional details are available on the nebivolol hydrochloride; valsartan profile page.
When can AEMCOLO (rifamycin) generic drug versions launch?
Generic name: rifamycin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 25, 2024
Generic Entry Controlled by: Japan Patent 2,008,163,038
Patent Title: ORAL ANTIMICROBIAL PHARMACEUTICAL COMPOSITION
AEMCOLO is a drug marketed by Redhill. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries.
See drug price trends for AEMCOLO.
The generic ingredient in AEMCOLO is rifamycin. There are eleven drug master file entries for this API. Additional details are available on the rifamycin profile page.
When can AEMCOLO (rifamycin) generic drug versions launch?
Generic name: rifamycin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 25, 2024
Generic Entry Controlled by: Japan Patent 2,008,503,540
AEMCOLO is a drug marketed by Redhill. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries.
See drug price trends for AEMCOLO.
The generic ingredient in AEMCOLO is rifamycin. There are eleven drug master file entries for this API. Additional details are available on the rifamycin profile page.
When can AEMCOLO (rifamycin) generic drug versions launch?
Generic name: rifamycin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 25, 2024
Generic Entry Controlled by: Japan Patent 5,026,262
AEMCOLO is a drug marketed by Redhill. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries.
See drug price trends for AEMCOLO.
The generic ingredient in AEMCOLO is rifamycin. There are eleven drug master file entries for this API. Additional details are available on the rifamycin profile page.
When can NUCYNTA (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Japan Patent 2,008,504,326

This drug has forty-five patent family members in twenty-six countries. There has been litigation on patents covering NUCYNTA
See drug price trends for NUCYNTA.
The generic ingredient in NUCYNTA is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can NUCYNTA (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Japan Patent 4,990,764

This drug has forty-five patent family members in twenty-six countries. There has been litigation on patents covering NUCYNTA
See drug price trends for NUCYNTA.
The generic ingredient in NUCYNTA is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can NUCYNTA ER (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Japan Patent 2,008,504,326

This drug has two hundred and seventy-two patent family members in thirty-one countries. There has been litigation on patents covering NUCYNTA ER
See drug price trends for NUCYNTA ER.
The generic ingredient in NUCYNTA ER is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can NUCYNTA ER (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: Japan Patent 4,990,764

This drug has two hundred and seventy-two patent family members in thirty-one countries. There has been litigation on patents covering NUCYNTA ER
See drug price trends for NUCYNTA ER.
The generic ingredient in NUCYNTA ER is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: Japan Patent 2,007,099,781
Patent Title: METHOD FOR PRODUCING CRYSTAL OF 1,2-DIHYDROPYRIDINE COMPOUND
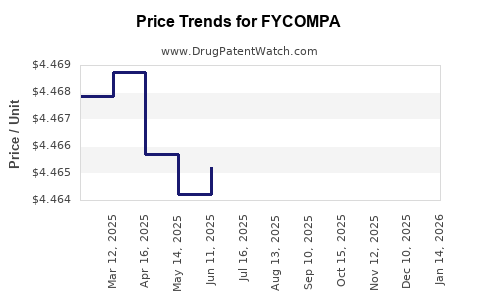
This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: Japan Patent 2,012,021,029
Patent Title: CRYSTAL OF 1,2-DIHYDROPYRIDINE COMPOUND

This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: Japan Patent 5,030,206

This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: Japan Patent 5,419,944

This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: Japan Patent WO2006004100
Patent Title: 1,2−ジヒドロピリジン−2−オン化合物の製造方法

This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: Japan Patent WO2006004107
Patent Title: 1,2−ジヒドロピリジン化合物の結晶およびその製造方法

This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can ARNUITY ELLIPTA (fluticasone furoate) generic drug versions launch?
Generic name: fluticasone furoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 16, 2024
Generic Entry Controlled by: Japan Patent 2,008,509,766
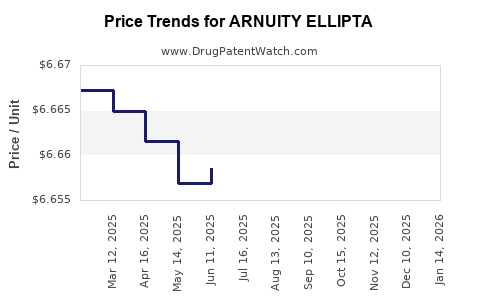
This drug has one hundred and seventeen patent family members in twenty-seven countries. There has been litigation on patents covering ARNUITY ELLIPTA
See drug price trends for ARNUITY ELLIPTA.
The generic ingredient in ARNUITY ELLIPTA is fluticasone furoate. There are twenty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the fluticasone furoate profile page.
When can ARNUITY ELLIPTA (fluticasone furoate) generic drug versions launch?
Generic name: fluticasone furoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 16, 2024
Generic Entry Controlled by: Japan Patent 4,960,868

This drug has one hundred and seventeen patent family members in twenty-seven countries. There has been litigation on patents covering ARNUITY ELLIPTA
See drug price trends for ARNUITY ELLIPTA.
The generic ingredient in ARNUITY ELLIPTA is fluticasone furoate. There are twenty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the fluticasone furoate profile page.
When can ERIVEDGE (vismodegib) generic drug versions launch?
Generic name: vismodegib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 02, 2024
Generic Entry Controlled by: Japan Patent 2,008,511,675
ERIVEDGE is a drug marketed by Genentech. There are three patents protecting this drug.
This drug has fifty-four patent family members in twenty-four countries.
See drug price trends for ERIVEDGE.
The generic ingredient in ERIVEDGE is vismodegib. One supplier is listed for this generic product. Additional details are available on the vismodegib profile page.
When can ERIVEDGE (vismodegib) generic drug versions launch?
Generic name: vismodegib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 02, 2024
Generic Entry Controlled by: Japan Patent 2,012,184,255
Patent Title: HEDGEHOG SIGNAL TRANSDUCTION PYRIDYL INHIBITOR
ERIVEDGE is a drug marketed by Genentech. There are three patents protecting this drug.
This drug has fifty-four patent family members in twenty-four countries.
See drug price trends for ERIVEDGE.
The generic ingredient in ERIVEDGE is vismodegib. One supplier is listed for this generic product. Additional details are available on the vismodegib profile page.
When can ERIVEDGE (vismodegib) generic drug versions launch?
Generic name: vismodegib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 02, 2024
Generic Entry Controlled by: Japan Patent 2,014,148,537
Patent Title: PYRIDYL INHIBITORS OF HEDGEHOG SIGNALLING
ERIVEDGE is a drug marketed by Genentech. There are three patents protecting this drug.
This drug has fifty-four patent family members in twenty-four countries.
See drug price trends for ERIVEDGE.
The generic ingredient in ERIVEDGE is vismodegib. One supplier is listed for this generic product. Additional details are available on the vismodegib profile page.
When can ERIVEDGE (vismodegib) generic drug versions launch?
Generic name: vismodegib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 02, 2024
Generic Entry Controlled by: Japan Patent 5,143,558
ERIVEDGE is a drug marketed by Genentech. There are three patents protecting this drug.
This drug has fifty-four patent family members in twenty-four countries.
See drug price trends for ERIVEDGE.
The generic ingredient in ERIVEDGE is vismodegib. One supplier is listed for this generic product. Additional details are available on the vismodegib profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: Japan Patent 2,008,514,646
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: Japan Patent 2,012,107,064
Patent Title: SEMI-SOLID DELIVERY VEHICLE AND PHARMACEUTICAL COMPOSITION
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: Japan Patent 2,015,034,182
Patent Title: 半固体送達賦形剤および薬学的組成物 (SEMI-SOLID DELIVERY VEHICLE AND PHARMACEUTICAL COMPOSITIONS)
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: Japan Patent 2,017,008,113
Patent Title: 半固体送達賦形剤および薬学的組成物 (SEMI-SOLID DELIVERY VEHICLE AND PHARMACEUTICAL COMPOSITIONS)
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: Japan Patent 6,057,972
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can QUARTETTE (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 07, 2024
Generic Entry Controlled by: Japan Patent 2,008,515,909
QUARTETTE is a drug marketed by Teva Branded Pharm. There are two patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has twenty-three patent family members in thirteen countries. There has been litigation on patents covering QUARTETTE
See drug price trends for QUARTETTE.
The generic ingredient in QUARTETTE is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can QUARTETTE (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 07, 2024
Generic Entry Controlled by: Japan Patent 2,010,508,275
QUARTETTE is a drug marketed by Teva Branded Pharm. There are two patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has twenty-three patent family members in thirteen countries. There has been litigation on patents covering QUARTETTE
See drug price trends for QUARTETTE.
The generic ingredient in QUARTETTE is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can BAXDELA (delafloxacin meglumine) generic drug versions launch?
Generic name: delafloxacin meglumine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 08, 2024
Generic Entry Controlled by: Japan Patent 2,008,515,910

This drug has one hundred and ten patent family members in twenty-eight countries. There has been litigation on patents covering BAXDELA
See drug price trends for BAXDELA.
The generic ingredient in BAXDELA is delafloxacin meglumine. One supplier is listed for this generic product. Additional details are available on the delafloxacin meglumine profile page.
When can BAXDELA (delafloxacin meglumine) generic drug versions launch?
Generic name: delafloxacin meglumine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 08, 2024
Generic Entry Controlled by: Japan Patent 5,294,633

This drug has one hundred and ten patent family members in twenty-eight countries. There has been litigation on patents covering BAXDELA
See drug price trends for BAXDELA.
The generic ingredient in BAXDELA is delafloxacin meglumine. One supplier is listed for this generic product. Additional details are available on the delafloxacin meglumine profile page.
When can INCIVEK (telaprevir) generic drug versions launch?
Generic name: telaprevir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 29, 2024
Generic Entry Controlled by: Japan Patent 2,008,518,943
INCIVEK is a drug marketed by Vertex Pharms. There are two patents protecting this drug.
This drug has one hundred and twenty-four patent family members in thirty-eight countries.
The generic ingredient in INCIVEK is telaprevir. There are three drug master file entries for this API. Additional details are available on the telaprevir profile page.
When can INCIVEK (telaprevir) generic drug versions launch?
Generic name: telaprevir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 29, 2024
Generic Entry Controlled by: Japan Patent 2,013,060,474
Patent Title: DOSE FORMS COMPRISING VX-950 AND THEIR DOSAGE REGIMEN
INCIVEK is a drug marketed by Vertex Pharms. There are two patents protecting this drug.
This drug has one hundred and twenty-four patent family members in thirty-eight countries.
The generic ingredient in INCIVEK is telaprevir. There are three drug master file entries for this API. Additional details are available on the telaprevir profile page.
When can YONDELIS (trabectedin) generic drug versions launch?
Generic name: trabectedin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 29, 2024
Generic Entry Controlled by: Japan Patent 2,006,124,393
Patent Title: FORMULATION
YONDELIS is a drug marketed by Janssen Prods. There is one patent protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has forty-four patent family members in thirty-six countries. There has been litigation on patents covering YONDELIS
See drug price trends for YONDELIS.
The generic ingredient in YONDELIS is trabectedin. There are four drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the trabectedin profile page.
When can YONDELIS (trabectedin) generic drug versions launch?
Generic name: trabectedin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 29, 2024
Generic Entry Controlled by: Japan Patent 4,268,163
YONDELIS is a drug marketed by Janssen Prods. There is one patent protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has forty-four patent family members in thirty-six countries. There has been litigation on patents covering YONDELIS
See drug price trends for YONDELIS.
The generic ingredient in YONDELIS is trabectedin. There are four drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the trabectedin profile page.
When can DEFENCATH (heparin sodium; taurolidine) generic drug versions launch?
Generic name: heparin sodium; taurolidine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 02, 2024
Generic Entry Controlled by: Japan Patent 2,008,518,739
DEFENCATH is a drug marketed by Cormedix. There are four patents protecting this drug.
This drug has fourteen patent family members in eight countries.
The generic ingredient in DEFENCATH is heparin sodium; taurolidine. There are seventy-seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the heparin sodium; taurolidine profile page.
When can DEFENCATH (heparin sodium; taurolidine) generic drug versions launch?
Generic name: heparin sodium; taurolidine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 02, 2024
Generic Entry Controlled by: Japan Patent 2,013,090,933
Patent Title: ANTIMICROBIAL LOCKING SOLUTION COMPRISING TAURINAMIDE DERIVATIVE AND BIOLOGICALLY ACCEPTABLE SALT AND ACID, WITH ADDITION OF SMALL CONCENTRATION OF HEPARIN
DEFENCATH is a drug marketed by Cormedix. There are four patents protecting this drug.
This drug has fourteen patent family members in eight countries.
The generic ingredient in DEFENCATH is heparin sodium; taurolidine. There are seventy-seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the heparin sodium; taurolidine profile page.
When can DEFENCATH (heparin sodium; taurolidine) generic drug versions launch?
Generic name: heparin sodium; taurolidine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 02, 2024
Generic Entry Controlled by: Japan Patent 5,805,617
DEFENCATH is a drug marketed by Cormedix. There are four patents protecting this drug.
This drug has fourteen patent family members in eight countries.
The generic ingredient in DEFENCATH is heparin sodium; taurolidine. There are seventy-seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the heparin sodium; taurolidine profile page.
When can HORIZANT (gabapentin enacarbil) generic drug versions launch?
Generic name: gabapentin enacarbil
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 04, 2024
Generic Entry Controlled by: Japan Patent 2,008,518,971
HORIZANT is a drug marketed by Azurity. There are five patents protecting this drug.
This drug has one hundred and forty-six patent family members in twenty-six countries.
See drug price trends for HORIZANT.
The generic ingredient in HORIZANT is gabapentin enacarbil. There are twenty-nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the gabapentin enacarbil profile page.
When can HORIZANT (gabapentin enacarbil) generic drug versions launch?
Generic name: gabapentin enacarbil
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 04, 2024
Generic Entry Controlled by: Japan Patent 2,013,107,900
Patent Title: GABAPENTIN PRODRUG SUSTAINED RELEASE ORAL DRUG
HORIZANT is a drug marketed by Azurity. There are five patents protecting this drug.
This drug has one hundred and forty-six patent family members in twenty-six countries.
See drug price trends for HORIZANT.
The generic ingredient in HORIZANT is gabapentin enacarbil. There are twenty-nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the gabapentin enacarbil profile page.
When can HORIZANT (gabapentin enacarbil) generic drug versions launch?
Generic name: gabapentin enacarbil
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 04, 2024
Generic Entry Controlled by: Japan Patent 5,856,095
HORIZANT is a drug marketed by Azurity. There are five patents protecting this drug.
This drug has one hundred and forty-six patent family members in twenty-six countries.
See drug price trends for HORIZANT.
The generic ingredient in HORIZANT is gabapentin enacarbil. There are twenty-nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the gabapentin enacarbil profile page.
When can KUVAN (sapropterin dihydrochloride) generic drug versions launch?
Generic name: sapropterin dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 17, 2024
Generic Entry Controlled by: Japan Patent 2,008,520,574
KUVAN is a drug marketed by Biomarin Pharm. There are ten patents protecting this drug and three Paragraph IV challenges.
This drug has one hundred and seventeen patent family members in twenty-six countries. There has been litigation on patents covering KUVAN
See drug price trends for KUVAN.
The generic ingredient in KUVAN is sapropterin dihydrochloride. There are three drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the sapropterin dihydrochloride profile page.
When can KUVAN (sapropterin dihydrochloride) generic drug versions launch?
Generic name: sapropterin dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 17, 2024
Generic Entry Controlled by: Japan Patent 2,014,012,740
Patent Title: STABLE TABLET FORMULATION OF TETRAHYDROBIOPTERIN
KUVAN is a drug marketed by Biomarin Pharm. There are ten patents protecting this drug and three Paragraph IV challenges.
This drug has one hundred and seventeen patent family members in twenty-six countries. There has been litigation on patents covering KUVAN
See drug price trends for KUVAN.
The generic ingredient in KUVAN is sapropterin dihydrochloride. There are three drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the sapropterin dihydrochloride profile page.
When can NEVANAC (nepafenac) generic drug versions launch?
Generic name: nepafenac
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 02, 2024
Generic Entry Controlled by: Japan Patent 2,008,521,926
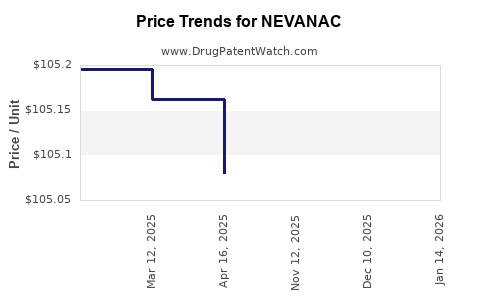
This drug has twenty-seven patent family members in twenty-three countries.
See drug price trends for NEVANAC.
The generic ingredient in NEVANAC is nepafenac. There are eight drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the nepafenac profile page.
When can NEVANAC (nepafenac) generic drug versions launch?
Generic name: nepafenac
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 02, 2024
Generic Entry Controlled by: Japan Patent 2,012,041,368
Patent Title: TOPICAL NEPAFENAC FORMULATION

This drug has twenty-seven patent family members in twenty-three countries.
See drug price trends for NEVANAC.
The generic ingredient in NEVANAC is nepafenac. There are eight drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the nepafenac profile page.
When can NEVANAC (nepafenac) generic drug versions launch?
Generic name: nepafenac
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 02, 2024
Generic Entry Controlled by: Japan Patent 4,968,844

This drug has twenty-seven patent family members in twenty-three countries.
See drug price trends for NEVANAC.
The generic ingredient in NEVANAC is nepafenac. There are eight drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the nepafenac profile page.
When can DUTREBIS (lamivudine; raltegravir potassium) generic drug versions launch?
Generic name: lamivudine; raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: Japan Patent 2,008,521,933
DUTREBIS is a drug marketed by Merck Sharp Dohme. There are two patents protecting this drug.
This drug has ninety-five patent family members in forty-three countries.
The generic ingredient in DUTREBIS is lamivudine; raltegravir potassium. There are twenty-nine drug master file entries for this API. Additional details are available on the lamivudine; raltegravir potassium profile page.
When can DUTREBIS (lamivudine; raltegravir potassium) generic drug versions launch?
Generic name: lamivudine; raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: Japan Patent 4,705,956
DUTREBIS is a drug marketed by Merck Sharp Dohme. There are two patents protecting this drug.
This drug has ninety-five patent family members in forty-three countries.
The generic ingredient in DUTREBIS is lamivudine; raltegravir potassium. There are twenty-nine drug master file entries for this API. Additional details are available on the lamivudine; raltegravir potassium profile page.