European Patent Office: These 62 Drugs Face Patent Expirations and Generic Entry From 2024 - 2025
DrugPatentWatch® Estimated Loss of Exclusivity Dates in European Patent Office
The content of this page is licensed under a Creative Commons Attribution 4.0 International License.

Generic Entry Dates in Other Countries
Friedman, Yali, "European Patent Office: These 62 Drugs Face Patent Expirations and Generic Entry From 2024 - 2025" DrugPatentWatch.com thinkBiotech, 2024 www.drugpatentwatch.com/p/expiring-drug-patents-generic-entry/.
Media collateral
These estimated drug patent expiration dates and generic entry opportunity dates are calculated from analysis of known patents covering drugs. Many factors can influence early or late generic entry. This information is provided as a rough estimate of generic entry potential and should not be used as an independent source. The methodology is described in this blog post.
When can VIZIMPRO (dacomitinib) generic drug versions launch?
Generic name: dacomitinib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 06, 2024
Generic Entry Controlled by: European Patent Office Patent 1,746,999
Patent Title: 4-PHENYLAMINO-QUINAZOLIN-6-YL-AMIDES (4-PHENYLAMINO-QUINAZOLIN-6-YL-AMIDES)
VIZIMPRO is a drug marketed by Pfizer. There are four patents protecting this drug.
This drug has ninety-three patent family members in forty-eight countries.
See drug price trends for VIZIMPRO.
The generic ingredient in VIZIMPRO is dacomitinib. Two suppliers are listed for this generic product. Additional details are available on the dacomitinib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: European Patent Office Patent 1,756,079
Patent Title: SYNTHESE DE CETO-EPOXYDES D'ACIDES AMINES (SYNTHESIS OF AMINO ACID KETO-EPOXIDES)
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: European Patent Office Patent 1,758,921
Patent Title: COMPOSES D'INHIBITION ENZYMATIQUE PROTEASOME (COMPOUNDS FOR PROTEASOME ENZYME INHIBITION)
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: European Patent Office Patent 1,819,353
Patent Title: COMPOSITION DESTINEE A INHIBER LE PROTEASOME (COMPOSITION FOR PROTEASOME INHIBITION)
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: European Patent Office Patent 2,030,981
Patent Title: Composés pour l'inhibition d'une enzyme de proteasome (Compounds for proteasome enzyme inhibition)
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: European Patent Office Patent 2,260,835
Patent Title: Composition destinée a inhiber le proteasome (Composition for proteasome inhibition)
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: European Patent Office Patent 2,261,236
Patent Title: Composition destinée à inhiber le protéasome (Composition for proteasome inhibition)
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can KYPROLIS (carfilzomib) generic drug versions launch?
Generic name: carfilzomib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 10, 2024
Generic Entry Controlled by: European Patent Office Patent 2,564,834
Patent Title: Composés pour l'inhibition d'une enzyme du protéasome (Compounds for Proteasome Enzyme Inhibition)
KYPROLIS is a drug marketed by Onyx Pharms Amgen. There are eleven patents protecting this drug and three Paragraph IV challenges. Four tentatively approved generics are ready to enter the market.
This drug has two hundred and twenty patent family members in forty-two countries. There has been litigation on patents covering KYPROLIS
See drug price trends for KYPROLIS.
The generic ingredient in KYPROLIS is carfilzomib. There are ten drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the carfilzomib profile page.
When can ARCAPTA NEOHALER (indacaterol maleate) generic drug versions launch?
Generic name: indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: European Patent Office Patent 1,747,036
Patent Title: Dispositif d'inhalation (Inhaler device)

This drug has eighty-six patent family members in thirty-nine countries.
See drug price trends for ARCAPTA NEOHALER.
The generic ingredient in ARCAPTA NEOHALER is indacaterol maleate. Additional details are available on the indacaterol maleate profile page.
When can UTIBRON (glycopyrrolate ; indacaterol maleate) generic drug versions launch?
Generic name: glycopyrrolate ; indacaterol maleate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 13, 2024
Generic Entry Controlled by: European Patent Office Patent 1,747,036
Patent Title: Dispositif d'inhalation (Inhaler device)
UTIBRON is a drug marketed by Novartis. There are three patents protecting this drug.
This drug has one hundred and forty-eight patent family members in forty countries. There has been litigation on patents covering UTIBRON
See drug price trends for UTIBRON.
The generic ingredient in UTIBRON is glycopyrrolate ; indacaterol maleate. There are seventeen drug master file entries for this API. Additional details are available on the glycopyrrolate ; indacaterol maleate profile page.
When can VITEKTA (elvitegravir) generic drug versions launch?
Generic name: elvitegravir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 20, 2024
Generic Entry Controlled by: European Patent Office Patent 1,636,190
Patent Title: CRISTAL STABLE DE COMPOSE 4-OXOQUINOLINE (STABLE CRYSTAL OF 4-OXOQUINOLINE COMPOUND)
VITEKTA is a drug marketed by Gilead Sciences Inc. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries. There has been litigation on patents covering VITEKTA
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this API. Additional details are available on the elvitegravir profile page.
When can VITEKTA (elvitegravir) generic drug versions launch?
Generic name: elvitegravir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 20, 2024
Generic Entry Controlled by: European Patent Office Patent 2,514,746
Patent Title: Cristal stable de composé 4-oxoquinoline (Stable crystal of 4-oxoquinoline compound)
VITEKTA is a drug marketed by Gilead Sciences Inc. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries. There has been litigation on patents covering VITEKTA
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this API. Additional details are available on the elvitegravir profile page.
When can VITEKTA (elvitegravir) generic drug versions launch?
Generic name: elvitegravir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 20, 2024
Generic Entry Controlled by: European Patent Office Patent 3,281,939
Patent Title: MÉTHODE POUR PRÉPARATION D'UN CRISTAL STABLE DE COMPOSÉ 4-OXOQUINOLINE (METHOD OF PRODUCING STABLE CRYSTAL OF 4-OXOQUINOLINE COMPOUND)
VITEKTA is a drug marketed by Gilead Sciences Inc. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries. There has been litigation on patents covering VITEKTA
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this API. Additional details are available on the elvitegravir profile page.
When can VITEKTA (elvitegravir) generic drug versions launch?
Generic name: elvitegravir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 20, 2024
Generic Entry Controlled by: European Patent Office Patent 4,299,563
Patent Title: COMBINAISON COMPRENANT UN CRISTAL STABLE D'UN COMPOSÉ 4-OXOQUINOLINE (COMBINATION COMPRISING A STABLE CRYSTAL OF A 4-OXOQUINOLINE COMPOUND)
VITEKTA is a drug marketed by Gilead Sciences Inc. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries. There has been litigation on patents covering VITEKTA
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this API. Additional details are available on the elvitegravir profile page.
When can CAPRELSA (vandetanib) generic drug versions launch?
Generic name: vandetanib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 21, 2024
Generic Entry Controlled by: European Patent Office Patent 1,753,431
Patent Title: COMPOSITIONS PHARMACEUTIQUES COMPORTANT ZD6474 (PHARMACEUTICAL COMPOSITIONS COMPRISING ZD6474)
CAPRELSA is a drug marketed by Genzyme Corp. There is one patent protecting this drug.
This drug has forty patent family members in thirty-three countries.
See drug price trends for CAPRELSA.
The generic ingredient in CAPRELSA is vandetanib. One supplier is listed for this generic product. Additional details are available on the vandetanib profile page.
When can QNASL (beclomethasone dipropionate) generic drug versions launch?
Generic name: beclomethasone dipropionate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 21, 2024
Generic Entry Controlled by: European Patent Office Patent 1,758,631
Patent Title: APPAREIL DISTRIBUTEUR A COMPTEUR DE DOSES (DISPENSING APPARATUS WITH COUNTER)
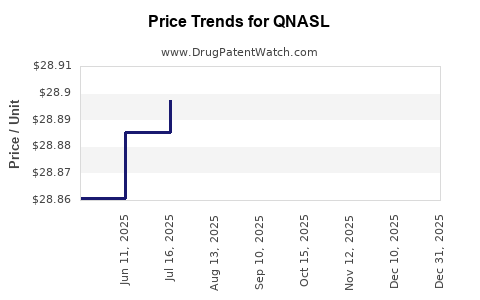
This drug has fifty-two patent family members in thirty-two countries.
See drug price trends for QNASL.
The generic ingredient in QNASL is beclomethasone dipropionate. There are twenty drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the beclomethasone dipropionate profile page.
When can CHILDREN'S ADVIL ALLERGY SINUS (chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride) generic drug versions launch?
Generic name: chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 25, 2024
Generic Entry Controlled by: European Patent Office Patent 1,748,765
Patent Title: SUSPENSION PHARMACEUTIQUE (PHARMACEUTICAL SUSPENSION COMPOSITION)
CHILDREN'S ADVIL ALLERGY SINUS is a drug marketed by Haleon Us Holdings. There is one patent protecting this drug.
This drug has nine patent family members in eight countries.
The generic ingredient in CHILDREN'S ADVIL ALLERGY SINUS is chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride. There are twenty-nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride profile page.
When can CHILDREN'S ADVIL ALLERGY SINUS (chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride) generic drug versions launch?
Generic name: chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 25, 2024
Generic Entry Controlled by: European Patent Office Patent 2,465,497
Patent Title: Suspension pharmaceutique (Pharmaceutical suspension composition)
CHILDREN'S ADVIL ALLERGY SINUS is a drug marketed by Haleon Us Holdings. There is one patent protecting this drug.
This drug has nine patent family members in eight countries.
The generic ingredient in CHILDREN'S ADVIL ALLERGY SINUS is chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride. There are twenty-nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the chlorpheniramine maleate; ibuprofen; pseudoephedrine hydrochloride profile page.
When can KOMBIGLYZE XR (metformin hydrochloride; saxagliptin hydrochloride) generic drug versions launch?
Generic name: metformin hydrochloride; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: European Patent Office Patent 1,753,406
Patent Title: FORMULATION DE COMPRIME REVETU ET PROCEDE CORRESPONDANT (COATED TABLET FORMULATION AND METHOD)

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering KOMBIGLYZE XR
See drug price trends for KOMBIGLYZE XR.
The generic ingredient in KOMBIGLYZE XR is metformin hydrochloride; saxagliptin hydrochloride. There are forty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the metformin hydrochloride; saxagliptin hydrochloride profile page.
When can KOMBIGLYZE XR (metformin hydrochloride; saxagliptin hydrochloride) generic drug versions launch?
Generic name: metformin hydrochloride; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: European Patent Office Patent 2,298,288
Patent Title: Formulation de comprime revêtu et procède correspondant (Coated tablet formulation and method)

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering KOMBIGLYZE XR
See drug price trends for KOMBIGLYZE XR.
The generic ingredient in KOMBIGLYZE XR is metformin hydrochloride; saxagliptin hydrochloride. There are forty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the metformin hydrochloride; saxagliptin hydrochloride profile page.
When can KOMBIGLYZE XR (metformin hydrochloride; saxagliptin hydrochloride) generic drug versions launch?
Generic name: metformin hydrochloride; saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: European Patent Office Patent 3,078,369
Patent Title: FORMULATION DE COMPRIMÉ REVÊTU ET PROCÉDÉ CORRESPONDANT (COATED TABLET FORMULATION AND METHOD)

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering KOMBIGLYZE XR
See drug price trends for KOMBIGLYZE XR.
The generic ingredient in KOMBIGLYZE XR is metformin hydrochloride; saxagliptin hydrochloride. There are forty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the metformin hydrochloride; saxagliptin hydrochloride profile page.
When can ONGLYZA (saxagliptin hydrochloride) generic drug versions launch?
Generic name: saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: European Patent Office Patent 1,753,406
Patent Title: FORMULATION DE COMPRIME REVETU ET PROCEDE CORRESPONDANT (COATED TABLET FORMULATION AND METHOD)

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering ONGLYZA
See drug price trends for ONGLYZA.
The generic ingredient in ONGLYZA is saxagliptin hydrochloride. There are fifteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the saxagliptin hydrochloride profile page.
When can ONGLYZA (saxagliptin hydrochloride) generic drug versions launch?
Generic name: saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: European Patent Office Patent 2,298,288
Patent Title: Formulation de comprime revêtu et procède correspondant (Coated tablet formulation and method)

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering ONGLYZA
See drug price trends for ONGLYZA.
The generic ingredient in ONGLYZA is saxagliptin hydrochloride. There are fifteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the saxagliptin hydrochloride profile page.
When can ONGLYZA (saxagliptin hydrochloride) generic drug versions launch?
Generic name: saxagliptin hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 28, 2024
Generic Entry Controlled by: European Patent Office Patent 3,078,369
Patent Title: FORMULATION DE COMPRIMÉ REVÊTU ET PROCÉDÉ CORRESPONDANT (COATED TABLET FORMULATION AND METHOD)

This drug has fifty-seven patent family members in thirty-two countries. There has been litigation on patents covering ONGLYZA
See drug price trends for ONGLYZA.
The generic ingredient in ONGLYZA is saxagliptin hydrochloride. There are fifteen drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the saxagliptin hydrochloride profile page.
When can FOLOTYN (pralatrexate) generic drug versions launch?
Generic name: pralatrexate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: May 30, 2024
Generic Entry Controlled by: European Patent Office Patent 1,750,716
Patent Title: TRAITEMENT DU LYMPHOME T UTILISANT 10-PROPARGYL-10-DEAZAAMINOPTERINE (TREATMENT OF T-CELL LYMPHOMA USING 10-PROPARGYL-10-DEAZAAMINOPTERIN)
FOLOTYN is a drug marketed by Acrotech Biopharma. There are two patents protecting this drug and one Paragraph IV challenge. One tentatively approved generic is ready to enter the market.
This drug has thirty-one patent family members in twenty-three countries. There has been litigation on patents covering FOLOTYN
See drug price trends for FOLOTYN.
The generic ingredient in FOLOTYN is pralatrexate. There are two drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the pralatrexate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: European Patent Office Patent 1,771,431
Patent Title: INTERMEDIAIRES POUR LA PREPARATION D'HALICHONDRINE B (INTERMEDIATES FOR THE PREPARATION OF HALICHONDRIN B)
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: European Patent Office Patent 2,522,663
Patent Title: Intermédiaires pour la préparation d'halichondrine b (Intermediates for the preparation of halichondrin B)
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: European Patent Office Patent 2,949,652
Patent Title: Intermédiaires pour la préparation d'halichondrine B (Intermediates for the preparation of halichondrin B)
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can HALAVEN (eribulin mesylate) generic drug versions launch?
Generic name: eribulin mesylate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 03, 2024
Generic Entry Controlled by: European Patent Office Patent 3,587,408
Patent Title: INTERMÉDIAIRES POUR LA PRÉPARATION D'HALICHONDRINE B (INTERMEDIATES FOR THE PREPARATION OF HALICHONDRIN B)
HALAVEN is a drug marketed by Eisai Inc. There are two patents protecting this drug.
This drug has seventy-eight patent family members in twenty-six countries. There has been litigation on patents covering HALAVEN
See drug price trends for HALAVEN.
The generic ingredient in HALAVEN is eribulin mesylate. Two suppliers are listed for this generic product. Additional details are available on the eribulin mesylate profile page.
When can BYVALSON (nebivolol hydrochloride; valsartan) generic drug versions launch?
Generic name: nebivolol hydrochloride; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 04, 2024
Generic Entry Controlled by: European Patent Office Patent 1,753,418
Patent Title: COMPOSITIONS COMPRENANT DU NÉBIVOLOL (COMPOSITIONS COMPRISING NEBIVOLOL)

This drug has thirty-seven patent family members in fifteen countries. There has been litigation on patents covering BYVALSON
See drug price trends for BYVALSON.
The generic ingredient in BYVALSON is nebivolol hydrochloride; valsartan. There are fourteen drug master file entries for this API. Additional details are available on the nebivolol hydrochloride; valsartan profile page.
When can BYVALSON (nebivolol hydrochloride; valsartan) generic drug versions launch?
Generic name: nebivolol hydrochloride; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 04, 2024
Generic Entry Controlled by: European Patent Office Patent 1,890,691
Patent Title: COMPOSITIONS A BASE DE NEBIVOLOL (COMPOSITIONS COMRISING NEBIVOLOL)

This drug has thirty-seven patent family members in fifteen countries. There has been litigation on patents covering BYVALSON
See drug price trends for BYVALSON.
The generic ingredient in BYVALSON is nebivolol hydrochloride; valsartan. There are fourteen drug master file entries for this API. Additional details are available on the nebivolol hydrochloride; valsartan profile page.
When can BYVALSON (nebivolol hydrochloride; valsartan) generic drug versions launch?
Generic name: nebivolol hydrochloride; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 04, 2024
Generic Entry Controlled by: European Patent Office Patent 1,982,711
Patent Title: Compositions comportant du nebivolol (Compositions comprsing nebivolol)

This drug has thirty-seven patent family members in fifteen countries. There has been litigation on patents covering BYVALSON
See drug price trends for BYVALSON.
The generic ingredient in BYVALSON is nebivolol hydrochloride; valsartan. There are fourteen drug master file entries for this API. Additional details are available on the nebivolol hydrochloride; valsartan profile page.
When can BYVALSON (nebivolol hydrochloride; valsartan) generic drug versions launch?
Generic name: nebivolol hydrochloride; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 04, 2024
Generic Entry Controlled by: European Patent Office Patent 2,174,658
Patent Title: Compositions comportant du nebivolol (Compositions comprising nebivolol)

This drug has thirty-seven patent family members in fifteen countries. There has been litigation on patents covering BYVALSON
See drug price trends for BYVALSON.
The generic ingredient in BYVALSON is nebivolol hydrochloride; valsartan. There are fourteen drug master file entries for this API. Additional details are available on the nebivolol hydrochloride; valsartan profile page.
When can BYVALSON (nebivolol hydrochloride; valsartan) generic drug versions launch?
Generic name: nebivolol hydrochloride; valsartan
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 04, 2024
Generic Entry Controlled by: European Patent Office Patent 2,808,015
Patent Title: Compositions comportant du nebivolol (Compositions comprising nebivolol)

This drug has thirty-seven patent family members in fifteen countries. There has been litigation on patents covering BYVALSON
See drug price trends for BYVALSON.
The generic ingredient in BYVALSON is nebivolol hydrochloride; valsartan. There are fourteen drug master file entries for this API. Additional details are available on the nebivolol hydrochloride; valsartan profile page.
When can SUPPRELIN LA (histrelin acetate) generic drug versions launch?
Generic name: histrelin acetate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 17, 2024
Generic Entry Controlled by: European Patent Office Patent 1,773,293
Patent Title: COMPOSITIONS ET METHODES DE TRAITEMENT DE LA PUBERTE CENTRALE PRECOCE (COMPOSITIONS AND METHODS FOR TREATING CENTRAL PRECOCIOUS PUBERTY)
SUPPRELIN LA is a drug marketed by Endo Pharm. There is one patent protecting this drug.
This drug has three patent family members in three countries.
See drug price trends for SUPPRELIN LA.
The generic ingredient in SUPPRELIN LA is histrelin acetate. There are two drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the histrelin acetate profile page.
When can AEMCOLO (rifamycin) generic drug versions launch?
Generic name: rifamycin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 25, 2024
Generic Entry Controlled by: European Patent Office Patent 1,763,339
Patent Title: COMPOSITIONS PHARMACEUTIQUES ANTIMICROBIENNES ORALES (ORAL ANTIMICROBIAL PHARMCEUTICAL COMPOSITIONS)
AEMCOLO is a drug marketed by Redhill. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries.
See drug price trends for AEMCOLO.
The generic ingredient in AEMCOLO is rifamycin. There are eleven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the rifamycin profile page.
When can AEMCOLO (rifamycin) generic drug versions launch?
Generic name: rifamycin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 25, 2024
Generic Entry Controlled by: European Patent Office Patent 1,894,560
Patent Title: Compositions pharmaceutiques antimicrobiennes orales comprenant du métronidazole (Oral antimicrobial pharmaceutical compositions comprising metronidazole)
AEMCOLO is a drug marketed by Redhill. There are four patents protecting this drug.
This drug has twenty-eight patent family members in twenty-one countries.
See drug price trends for AEMCOLO.
The generic ingredient in AEMCOLO is rifamycin. There are eleven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the rifamycin profile page.
When can NUCYNTA (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 1,612,203
Patent Title: Formes cristallines de chlorhydrate de (-)-(1R,2R)-3-(3-diméthylamino-1-ethyl-2-méthylpropyl)-phénol (Crystalline forms of (-)-(1R,2R)-3-(3-dimethylamino-1-ethyl-2-methylpropyl)-phenol hydrochloride)

This drug has forty-five patent family members in twenty-six countries. There has been litigation on patents covering NUCYNTA
See drug price trends for NUCYNTA.
The generic ingredient in NUCYNTA is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can NUCYNTA (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 1,799,633
Patent Title: FORMES CRISTALLINES DE CHLORHYDRATE DE (-)-(1R,2R)-3-(3-DIMETHYLAMINO-1-ETHYL-2-METHYLPROPYL)-PHENOL POUR UTILISATION COMME AGENT ACTIF DANS DES COMPOSITIONS PHARMACEUTIQUES (CRYSTALLINE FORMS OF (-)-(1R,2R)-3-(3-DIMETHYLAMINO-1-ETHYL-2-METHYLPROPYL)-PHENOL HYDROCHLORIDE FOR USE AS ACTIVE INGREDIENT IN PHARMACEUTICAL COMPOSITIONS)

This drug has forty-five patent family members in twenty-six countries. There has been litigation on patents covering NUCYNTA
See drug price trends for NUCYNTA.
The generic ingredient in NUCYNTA is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can NUCYNTA ER (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 1,612,203
Patent Title: Formes cristallines de chlorhydrate de (-)-(1R,2R)-3-(3-diméthylamino-1-ethyl-2-méthylpropyl)-phénol (Crystalline forms of (-)-(1R,2R)-3-(3-dimethylamino-1-ethyl-2-methylpropyl)-phenol hydrochloride)

This drug has two hundred and seventy-two patent family members in thirty-one countries. There has been litigation on patents covering NUCYNTA ER
See drug price trends for NUCYNTA ER.
The generic ingredient in NUCYNTA ER is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can NUCYNTA ER (tapentadol hydrochloride) generic drug versions launch?
Generic name: tapentadol hydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 1,799,633
Patent Title: FORMES CRISTALLINES DE CHLORHYDRATE DE (-)-(1R,2R)-3-(3-DIMETHYLAMINO-1-ETHYL-2-METHYLPROPYL)-PHENOL POUR UTILISATION COMME AGENT ACTIF DANS DES COMPOSITIONS PHARMACEUTIQUES (CRYSTALLINE FORMS OF (-)-(1R,2R)-3-(3-DIMETHYLAMINO-1-ETHYL-2-METHYLPROPYL)-PHENOL HYDROCHLORIDE FOR USE AS ACTIVE INGREDIENT IN PHARMACEUTICAL COMPOSITIONS)

This drug has two hundred and seventy-two patent family members in thirty-one countries. There has been litigation on patents covering NUCYNTA ER
See drug price trends for NUCYNTA ER.
The generic ingredient in NUCYNTA ER is tapentadol hydrochloride. There are five drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the tapentadol hydrochloride profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 1,766,010
Patent Title: OLIGONUCLÉOTIDES ANTISENS PERMETTANT D'INDUIRE UN SAUT D'EXON ET LEURS PROCÉDÉS D'UTILISATION (ANTISENSE OLIGONUCLEOTIDES FOR INDUCING EXON SKIPPING AND METHODS OF USE THEREOF)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 2,206,781
Patent Title: Oligonucléotides antisens permettant d'induire un saut d'exon et leurs procédés d'utilisation (Antisense oligonucleotides for inducing exon skipping and methods of use thereof)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 2,305,200
Patent Title: Parure de transfert d'occupant (Occupant transfer topper)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 2,500,430
Patent Title: Oligonucléotides antisens permettant d'induire un saut d'exon et leurs procédés d'utilisation (Antisense oligonucleotides for inducing exon skipping and methods of use thereof)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 2,933,332
Patent Title: OLIGONUCLÉOTIDES ANTISENS PERMETTANT D'INDUIRE UN SAUT D'EXON ET LEURS PROCÉDÉS D'UTILISATION (ANTISENSE OLIGONUCLEOTIDES FOR INDUCING EXON SKIPPING AND METHODS OF USE THEREOF)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 3,029,142
Patent Title: OLIGONUCLÉOTIDES ANTISENS PERMETTANT D'INDUIRE UN SAUT D'EXON ET LEURS PROCÉDÉS D'UTILISATION (ANTISENSE OLIGONUCLEOTIDES FOR INDUCING EXON SKIPPING AND METHODS OF USE THEREOF)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 3,228,711
Patent Title: OLIGONUCLÉOTIDES ANTISENS PERMETTANT D'INDUIRE UN SAUT D'EXON ET LEURS PROCÉDÉS D'UTILISATION (ANTISENSE OLIGONUCLEOTIDES FOR INDUCING EXON SKIPPING AND METHODS OF USE THEREOF)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 3,808,845
Patent Title: OLIGONUCLÉOTIDES ANTISENS POUR INDUIRE L'OMISSION DE L'EXON ET LEURS PROCÉDÉS D'UTILISATION (ANTISENSE OLIGONUCLEOTIDES FOR INDUCING EXON SKIPPING AND METHODS OF USE THEREOF)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 4,047,096
Patent Title: OLIGONUCLÉOTIDES ANTISENS POUR INDUIRE L'OMISSION DE L'EXON ET LEURS PROCÉDÉS D'UTILISATION (ANTISENSE OLIGONUCLEOTIDES FOR INDUCING EXON SKIPPING AND METHODS OF USE THEREOF)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can VYONDYS 53 (golodirsen) generic drug versions launch?
Generic name: golodirsen
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: June 28, 2024
Generic Entry Controlled by: European Patent Office Patent 4,272,748
Patent Title: OLIGONUCLÉOTIDES ANTISENS POUR INDUIRE L'OMISSION DE L'EXON ET LEURS PROCÉDÉS D'UTILISATION (ANTISENSE OLIGONUCLEOTIDES FOR INDUCING EXON SKIPPING AND METHODS OF USE THEREOF)
VYONDYS 53 is a drug marketed by Sarepta Theraps Inc. There are eight patents protecting this drug.
This drug has thirty patent family members in thirteen countries. There has been litigation on patents covering VYONDYS 53
The generic ingredient in VYONDYS 53 is golodirsen. One supplier is listed for this generic product. Additional details are available on the golodirsen profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: European Patent Office Patent 1,764,361
Patent Title: CRISTAL D'UN COMPOSÉ DE DIHYDROPYRIDINE ET PROCÉDÉ POUR LA FABRICATION DE CELUI-CI (CRYSTAL OF 1,2-DIHYDROPYRIDINE COMPOUND AND METHOD FOR PRODUCING SAME)
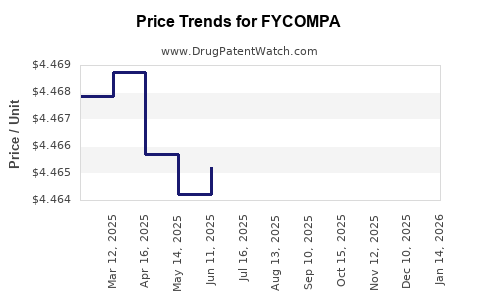
This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: European Patent Office Patent 1,772,450
Patent Title: PROCÉDÉ POUR LA FABRICATION D"UN COMPOSÉ DE 1,2-DIHYDROPYRIDINE-2-ONE (METHOD FOR PRODUCING 1,2-DIHYDROPYRIDINE-2-ONE COMPOUND)

This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can FYCOMPA (perampanel) generic drug versions launch?
Generic name: perampanel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: July 06, 2024
Generic Entry Controlled by: European Patent Office Patent 2,586,771
Patent Title: Cristal d'un composé de 1,2-dihydropyridine et son procédé de production (Crystal of 1,2-dihydropyridine compound and method for producing same)

This drug has one hundred and one patent family members in thirty-three countries. There has been litigation on patents covering FYCOMPA
See drug price trends for FYCOMPA.
The generic ingredient in FYCOMPA is perampanel. There are five drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the perampanel profile page.
When can ARNUITY ELLIPTA (fluticasone furoate) generic drug versions launch?
Generic name: fluticasone furoate
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: August 16, 2024
Generic Entry Controlled by: European Patent Office Patent 1,786,498
Patent Title: Distributeur de médicament (Medicament dispenser)
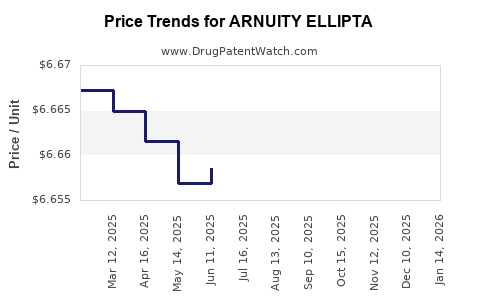
This drug has one hundred and seventeen patent family members in twenty-seven countries. There has been litigation on patents covering ARNUITY ELLIPTA
See drug price trends for ARNUITY ELLIPTA.
The generic ingredient in ARNUITY ELLIPTA is fluticasone furoate. There are twenty-nine drug master file entries for this API. Three suppliers are listed for this generic product. Additional details are available on the fluticasone furoate profile page.
When can ERIVEDGE (vismodegib) generic drug versions launch?
Generic name: vismodegib
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 02, 2024
Generic Entry Controlled by: European Patent Office Patent 1,789,390
Patent Title: INHIBITEURS PYRIDYLES DE LA SIGNALISATION HEDGEHOG (PYRIDYL INHIBITORS OF HEDGEHOG SIGNALLING)
ERIVEDGE is a drug marketed by Genentech. There are three patents protecting this drug.
This drug has fifty-four patent family members in twenty-four countries.
See drug price trends for ERIVEDGE.
The generic ingredient in ERIVEDGE is vismodegib. One supplier is listed for this generic product. Additional details are available on the vismodegib profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: European Patent Office Patent 1,796,629
Patent Title: VEHICULE D'ADMINISTRATION SEMI-SOLIDE ET COMPOSITIONS PHARMACEUTIQUES (SEMI-SOLID DELIVERY VEHICLE AND PHARMACEUTICAL COMPOSITIONS)
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: European Patent Office Patent 2,902,012
Patent Title: Véhicule d'administration semi-solide et compositions pharmaceutiques (Semi-solid delivery vehicle and pharmaceutical compositions)
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: European Patent Office Patent 3,424,492
Patent Title: VÉHICULE D'ADMINISTRATION SEMI-SOLIDE ET COMPOSITIONS PHARMACEUTIQUES (SEMI-SOLID DELIVERY VECHICLE AND PHARMACEUTICAL COMPOSITIONS)
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can SUSTOL (granisetron) generic drug versions launch?
Generic name: granisetron
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: September 28, 2024
Generic Entry Controlled by: European Patent Office Patent 3,834,817
Patent Title: VÉHICULE D'ADMINISTRATION SEMI-SOLIDE ET COMPOSITIONS PHARMACEUTIQUES (SEMI-SOLID DELIVERY VEHICLE AND PHARMACEUTICAL COMPOSITIONS)
SUSTOL is a drug marketed by Heron Theraps Inc. There are five patents protecting this drug.
This drug has eighteen patent family members in ten countries.
See drug price trends for SUSTOL.
The generic ingredient in SUSTOL is granisetron. There are twenty-six drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the granisetron profile page.
When can QUARTETTE (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 07, 2024
Generic Entry Controlled by: European Patent Office Patent 1,814,556
Patent Title: PROCEDES DE TRAITEMENT HORMONAL UTILISANT DES SCHEMAS POSOLOGIQUES A CYCLE ETENDU A AUGMENTATION DE DOSAGE (METHODS OF HORMONAL TREATMENT UTILIZING ASCENDING-DOSE EXTENDED CYCLE REGIMENS)
QUARTETTE is a drug marketed by Teva Branded Pharm. There are two patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has twenty-three patent family members in thirteen countries. There has been litigation on patents covering QUARTETTE
See drug price trends for QUARTETTE.
The generic ingredient in QUARTETTE is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can QUARTETTE (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 07, 2024
Generic Entry Controlled by: European Patent Office Patent 2,079,461
Patent Title: PROCÉDÉS DE TRAITEMENT HORMONAL UTILISANT DES SCHÉMAS POSOLOGIQUES CYCLIQUES PROLONGÉS À DOSES CROISSANTES (METHODS OF HORMONAL TREATMENT UTILIZING ASCENDING-DOSE EXTENDED CYCLE REGIMENS)
QUARTETTE is a drug marketed by Teva Branded Pharm. There are two patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has twenty-three patent family members in thirteen countries. There has been litigation on patents covering QUARTETTE
See drug price trends for QUARTETTE.
The generic ingredient in QUARTETTE is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can QUARTETTE (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 07, 2024
Generic Entry Controlled by: European Patent Office Patent 2,392,332
Patent Title: Procédé de traitement hormonal utilisant des régimes de cycle étendu à dose croissante (Methods of hormonal treatment utilizing ascending-dose extended cycle regimens)
QUARTETTE is a drug marketed by Teva Branded Pharm. There are two patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has twenty-three patent family members in thirteen countries. There has been litigation on patents covering QUARTETTE
See drug price trends for QUARTETTE.
The generic ingredient in QUARTETTE is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can QUARTETTE (ethinyl estradiol; levonorgestrel) generic drug versions launch?
Generic name: ethinyl estradiol; levonorgestrel
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 07, 2024
Generic Entry Controlled by: European Patent Office Patent 2,392,333
Patent Title: Procédé de traitement hormonal utilisant des régimes de cycle étendu à dose croissante (Methods of hormonal treatment utilizing ascending-dose extended cycle regimens)
QUARTETTE is a drug marketed by Teva Branded Pharm. There are two patents protecting this drug and one Paragraph IV challenge. Two tentatively approved generics are ready to enter the market.
This drug has twenty-three patent family members in thirteen countries. There has been litigation on patents covering QUARTETTE
See drug price trends for QUARTETTE.
The generic ingredient in QUARTETTE is ethinyl estradiol; levonorgestrel. There are twenty-six drug master file entries for this API. Twenty-six suppliers are listed for this generic product. Additional details are available on the ethinyl estradiol; levonorgestrel profile page.
When can BAXDELA (delafloxacin meglumine) generic drug versions launch?
Generic name: delafloxacin meglumine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 08, 2024
Generic Entry Controlled by: European Patent Office Patent 1,802,607
Patent Title: SEL ET SES FORMES CRISTALLINES D'UN MEDICAMENT (SALT AND CRYSTALLINE FORMS THEREOF OF A DRUG)

This drug has one hundred and ten patent family members in twenty-eight countries. There has been litigation on patents covering BAXDELA
See drug price trends for BAXDELA.
The generic ingredient in BAXDELA is delafloxacin meglumine. One supplier is listed for this generic product. Additional details are available on the delafloxacin meglumine profile page.
When can BAXDELA (delafloxacin meglumine) generic drug versions launch?
Generic name: delafloxacin meglumine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 08, 2024
Generic Entry Controlled by: European Patent Office Patent 3,056,492
Patent Title: SEL DE MEGLUMINE ET FORMES CRISTALLINES CORRESPONDANTES D'UN MÉDICAMENT (DELAFLOXACIN) (MEGUMINE SALT AND CRYSTALLINE FORMS THEREOF OF A DRUG (DELAFLOXACIN))

This drug has one hundred and ten patent family members in twenty-eight countries. There has been litigation on patents covering BAXDELA
See drug price trends for BAXDELA.
The generic ingredient in BAXDELA is delafloxacin meglumine. One supplier is listed for this generic product. Additional details are available on the delafloxacin meglumine profile page.
When can BAXDELA (delafloxacin meglumine) generic drug versions launch?
Generic name: delafloxacin meglumine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 08, 2024
Generic Entry Controlled by: European Patent Office Patent 3,957,632
Patent Title: SEL DE MÉGLUMINE ET FORMES CRISTALLINES CORRESPONDANTES D'UN MÉDICAMENT (DELAFLOXACIN) (MEGLUMINE SALT AND CRYSTALLINE FORMS THEREOF OF A DRUG (DELAFLOXACIN))

This drug has one hundred and ten patent family members in twenty-eight countries. There has been litigation on patents covering BAXDELA
See drug price trends for BAXDELA.
The generic ingredient in BAXDELA is delafloxacin meglumine. One supplier is listed for this generic product. Additional details are available on the delafloxacin meglumine profile page.
When can INCIVEK (telaprevir) generic drug versions launch?
Generic name: telaprevir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 29, 2024
Generic Entry Controlled by: European Patent Office Patent 1,819,336
Patent Title: FORMES DE DOSAGE COMPRENANT DE VX-950 ET LEUR REGIME DE DOSAGE (DOSE FORMS COMPRISING VX-950 AND THEIR DOSAGE REGIMEN)
INCIVEK is a drug marketed by Vertex Pharms. There are two patents protecting this drug.
This drug has one hundred and twenty-four patent family members in thirty-eight countries.
The generic ingredient in INCIVEK is telaprevir. There are three drug master file entries for this API. Additional details are available on the telaprevir profile page.
When can INCIVEK (telaprevir) generic drug versions launch?
Generic name: telaprevir
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 29, 2024
Generic Entry Controlled by: European Patent Office Patent 2,500,021
Patent Title: Utilisation thérapeutique du VX-950 (Therapeutic uses of VX-950)
INCIVEK is a drug marketed by Vertex Pharms. There are two patents protecting this drug.
This drug has one hundred and twenty-four patent family members in thirty-eight countries.
The generic ingredient in INCIVEK is telaprevir. There are three drug master file entries for this API. Additional details are available on the telaprevir profile page.
When can YONDELIS (trabectedin) generic drug versions launch?
Generic name: trabectedin
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: October 29, 2024
Generic Entry Controlled by: European Patent Office Patent 1,658,848
Patent Title: Compositions contenant d'ecteinascidin et undisaccharide (Formulations comprising ecteinascidin and a disaccharide)
YONDELIS is a drug marketed by Janssen Prods. There is one patent protecting this drug. One tentatively approved generic is ready to enter the market.
This drug has forty-four patent family members in thirty-six countries. There has been litigation on patents covering YONDELIS
See drug price trends for YONDELIS.
The generic ingredient in YONDELIS is trabectedin. There are four drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the trabectedin profile page.
When can DEFENCATH (heparin sodium; taurolidine) generic drug versions launch?
Generic name: heparin sodium; taurolidine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 02, 2024
Generic Entry Controlled by: European Patent Office Patent 1,814,562
Patent Title: SOLUTIONS DE BLOCAGE ANTI-MICROBIEN COMPRENANT DES DERIVES DE TAURINAMIDE AINSI QUE DES SELS ET DES ACIDES BIOLOGIQUEMENT ACCEPTABLES, AVEC ADJONCTION D'HEPARINE EN FAIBLES CONCENTRATIONS (ANTIMICROBIAL LOCKING SOLUTIONS COMPRISING TAURINAMIDE DERIVATIVES AND BIOLOGICALLY ACCEPTABLE SALTS AND ACIDS, WITH THE ADDITION OF SMALL CONCENTRATIONS OF HEPARIN)
DEFENCATH is a drug marketed by Cormedix. There are four patents protecting this drug.
This drug has fourteen patent family members in eight countries.
The generic ingredient in DEFENCATH is heparin sodium; taurolidine. There are seventy-seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the heparin sodium; taurolidine profile page.
When can DEFENCATH (heparin sodium; taurolidine) generic drug versions launch?
Generic name: heparin sodium; taurolidine
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 02, 2024
Generic Entry Controlled by: European Patent Office Patent 2,742,945
Patent Title: Solutions de blocage anti-microbien comprenant des dérivés de taurinamide ainsi que des sels et des acides biologiquement acceptables, avec adjonction d'heparine en faibles concentrations (Antimicrobial Locking Solutions Comprising Taurinamide Derivatives and Biologically Acceptable Salts and Acids, with the addition of Small Concentrations of Heparin)
DEFENCATH is a drug marketed by Cormedix. There are four patents protecting this drug.
This drug has fourteen patent family members in eight countries.
The generic ingredient in DEFENCATH is heparin sodium; taurolidine. There are seventy-seven drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the heparin sodium; taurolidine profile page.
When can HORIZANT (gabapentin enacarbil) generic drug versions launch?
Generic name: gabapentin enacarbil
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 04, 2024
Generic Entry Controlled by: European Patent Office Patent 1,811,986
Patent Title: FORMES POSOLOGIQUES ORALES A LIBERATION PROLONGEE D'UN PROMEDICAMENT DE LA GABAPENTINE (GABAPENTIN PRODRUG SUSTAINED RELEASE ORAL DOSAGE FORMS)
HORIZANT is a drug marketed by Azurity. There are five patents protecting this drug.
This drug has one hundred and forty-six patent family members in twenty-six countries.
See drug price trends for HORIZANT.
The generic ingredient in HORIZANT is gabapentin enacarbil. There are twenty-nine drug master file entries for this API. One supplier is listed for this generic product. Additional details are available on the gabapentin enacarbil profile page.
When can KUVAN (sapropterin dihydrochloride) generic drug versions launch?
Generic name: sapropterin dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 17, 2024
Generic Entry Controlled by: European Patent Office Patent 1,845,952
Patent Title: FORMULE STABLE DE COMPRIMES DE TETRAHYDROBIOPTERINE (STABLE TABLET FORMULATION OF TETRAHYDROBIOPTERIN)
KUVAN is a drug marketed by Biomarin Pharm. There are ten patents protecting this drug and three Paragraph IV challenges.
This drug has one hundred and seventeen patent family members in twenty-six countries. There has been litigation on patents covering KUVAN
See drug price trends for KUVAN.
The generic ingredient in KUVAN is sapropterin dihydrochloride. There are three drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the sapropterin dihydrochloride profile page.
When can KUVAN (sapropterin dihydrochloride) generic drug versions launch?
Generic name: sapropterin dihydrochloride
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: November 17, 2024
Generic Entry Controlled by: European Patent Office Patent 2,436,379
Patent Title: Formule de comprimé stable (Stable tablet formulation)
KUVAN is a drug marketed by Biomarin Pharm. There are ten patents protecting this drug and three Paragraph IV challenges.
This drug has one hundred and seventeen patent family members in twenty-six countries. There has been litigation on patents covering KUVAN
See drug price trends for KUVAN.
The generic ingredient in KUVAN is sapropterin dihydrochloride. There are three drug master file entries for this API. Six suppliers are listed for this generic product. Additional details are available on the sapropterin dihydrochloride profile page.
When can NEVANAC (nepafenac) generic drug versions launch?
Generic name: nepafenac
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 02, 2024
Generic Entry Controlled by: European Patent Office Patent 1,819,362
Patent Title: PREPARATIONS TOPIQUES A BASE DE NEPAFENAC (TOPICAL NEPAFENAC FORMULATIONS)
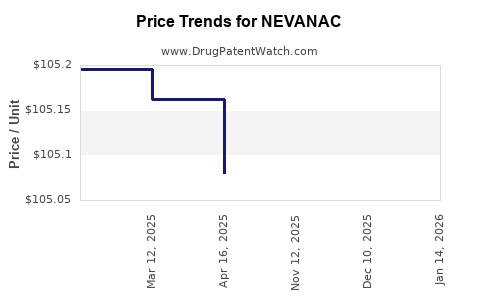
This drug has twenty-seven patent family members in twenty-three countries.
See drug price trends for NEVANAC.
The generic ingredient in NEVANAC is nepafenac. There are eight drug master file entries for this API. Two suppliers are listed for this generic product. Additional details are available on the nepafenac profile page.
When can DUTREBIS (lamivudine; raltegravir potassium) generic drug versions launch?
Generic name: lamivudine; raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: European Patent Office Patent 1,819,683
Patent Title: SELS DE POTASSIUM D'UN INHIBITEUR D'INTEGRASE HIV (POTASSIUM SALT OF AN HIV INTEGRASE INHIBITOR)
DUTREBIS is a drug marketed by Merck Sharp Dohme. There are two patents protecting this drug.
This drug has ninety-five patent family members in forty-three countries.
The generic ingredient in DUTREBIS is lamivudine; raltegravir potassium. There are twenty-nine drug master file entries for this API. Additional details are available on the lamivudine; raltegravir potassium profile page.
When can DUTREBIS (lamivudine; raltegravir potassium) generic drug versions launch?
Generic name: lamivudine; raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: European Patent Office Patent 1,819,700
Patent Title: SEL DE POTASSIUM D'UN INHIBITEUR DE L'INTEGRASE DU VIH (POTASSIUM SALT OF AN HIV INTEGRASE INHIBITOR)
DUTREBIS is a drug marketed by Merck Sharp Dohme. There are two patents protecting this drug.
This drug has ninety-five patent family members in forty-three countries.
The generic ingredient in DUTREBIS is lamivudine; raltegravir potassium. There are twenty-nine drug master file entries for this API. Additional details are available on the lamivudine; raltegravir potassium profile page.
When can ISENTRESS (raltegravir potassium) generic drug versions launch?
Generic name: raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: European Patent Office Patent 1,819,683
Patent Title: SELS DE POTASSIUM D'UN INHIBITEUR D'INTEGRASE HIV (POTASSIUM SALT OF AN HIV INTEGRASE INHIBITOR)
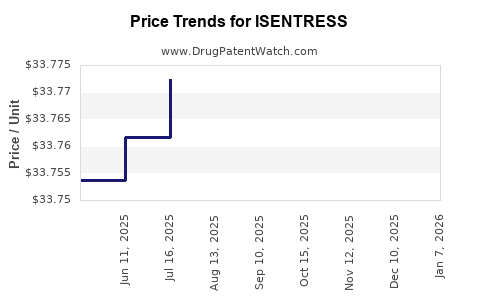
This drug has one hundred and twenty-five patent family members in forty-five countries.
See drug price trends for ISENTRESS.
The generic ingredient in ISENTRESS is raltegravir potassium. There are five drug master file entries for this API. Nine suppliers are listed for this generic product. Additional details are available on the raltegravir potassium profile page.
When can ISENTRESS (raltegravir potassium) generic drug versions launch?
Generic name: raltegravir potassium
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 03, 2024
Generic Entry Controlled by: European Patent Office Patent 1,819,700
Patent Title: SEL DE POTASSIUM D'UN INHIBITEUR DE L'INTEGRASE DU VIH (POTASSIUM SALT OF AN HIV INTEGRASE INHIBITOR)

This drug has one hundred and twenty-five patent family members in forty-five countries.
See drug price trends for ISENTRESS.
The generic ingredient in ISENTRESS is raltegravir potassium. There are five drug master file entries for this API. Nine suppliers are listed for this generic product. Additional details are available on the raltegravir potassium profile page.
When can NEURACEQ (florbetaben f-18) generic drug versions launch?
Generic name: florbetaben f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 17, 2024
Generic Entry Controlled by: European Patent Office Patent 1,838,298
Patent Title: Dérivés du stilbène et leur utilisation pour l'imagerie des plaques amyloïdes et pour se lier à elles (STILBENE DERIVATIVES AND THEIR USE FOR BINDING AND IMAGING AMYLOID PLAQUES)
NEURACEQ is a drug marketed by Life Molecular. There is one patent protecting this drug.
This drug has thirty-five patent family members in twenty-seven countries.
The generic ingredient in NEURACEQ is florbetaben f-18. One supplier is listed for this generic product. Additional details are available on the florbetaben f-18 profile page.
When can NEURACEQ (florbetaben f-18) generic drug versions launch?
Generic name: florbetaben f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 17, 2024
Generic Entry Controlled by: European Patent Office Patent 1,841,465
Patent Title: DERIVES DE STILBENE ET LEUR UTILISATION POUR LA LIAISON ET L'IMAGERIE DE PLAQUES AMYLOIDES (STILBENE DERIVATIVES AND THEIR USE FOR BINDING AND IMAGING AMYLOID PLAQUES)
NEURACEQ is a drug marketed by Life Molecular. There is one patent protecting this drug.
This drug has thirty-five patent family members in twenty-seven countries.
The generic ingredient in NEURACEQ is florbetaben f-18. One supplier is listed for this generic product. Additional details are available on the florbetaben f-18 profile page.
When can NEURACEQ (florbetaben f-18) generic drug versions launch?
Generic name: florbetaben f-18
DrugPatentWatch® Estimated Key Patent Expiration / Generic Entry Date: December 17, 2024
Generic Entry Controlled by: European Patent Office Patent 2,213,652
Patent Title: Dérivés de stilbène et leur utilisation pour l'imagerie des plaques amyloïdes et leur liaison (Stilbene derivatives and their use for binding and imaging amyloid plaques)
NEURACEQ is a drug marketed by Life Molecular. There is one patent protecting this drug.
This drug has thirty-five patent family members in twenty-seven countries.
The generic ingredient in NEURACEQ is florbetaben f-18. One supplier is listed for this generic product. Additional details are available on the florbetaben f-18 profile page.