VITEKTA Drug Patent Profile
✉ Email this page to a colleague
When do Vitekta patents expire, and when can generic versions of Vitekta launch?
Vitekta is a drug marketed by Gilead Sciences Inc and is included in one NDA. There are three patents protecting this drug.
This drug has ninety-two patent family members in thirty-six countries.
The generic ingredient in VITEKTA is elvitegravir. There are six drug master file entries for this compound. Additional details are available on the elvitegravir profile page.
DrugPatentWatch® Generic Entry Outlook for Vitekta
Vitekta was eligible for patent challenges on August 27, 2016.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 26, 2027. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for VITEKTA
| International Patents: | 92 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 82 |
| Patent Applications: | 1,458 |
| Formulation / Manufacturing: | see details |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VITEKTA |
| DailyMed Link: | VITEKTA at DailyMed |
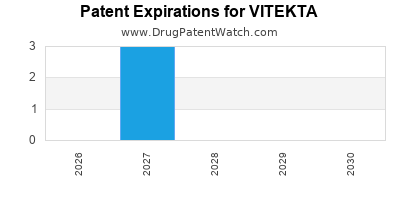
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VITEKTA
Generic Entry Date for VITEKTA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Anatomical Therapeutic Chemical (ATC) Classes for VITEKTA
US Patents and Regulatory Information for VITEKTA
VITEKTA is protected by three US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VITEKTA is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting VITEKTA
4-oxoquinoline compound and use thereof as pharmaceutical agent
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Stable crystal of 4-oxoquinoline compound
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Stable crystal of 4-oxoquinoline compound
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-001 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-002 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-001 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Gilead Sciences Inc | VITEKTA | elvitegravir | TABLET;ORAL | 203093-001 | Sep 24, 2014 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for VITEKTA
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences International Ltd | Vitekta | elvitegravir | EMEA/H/C/002577 Vitekta co-administered with a ritonavir-boosted protease inhibitor and with other antiretroviral agents, is indicated for the treatment of human-immunodeficiency-virus-1 (HIV-1) infection in adults who are infected with HIV-1 without known mutations associated with resistance to elvitegravir. |
Withdrawn | no | no | no | 2013-11-13 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for VITEKTA
When does loss-of-exclusivity occur for VITEKTA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 9280
Estimated Expiration: ⤷ Try a Trial
Patent: 6100
Estimated Expiration: ⤷ Try a Trial
Australia
Patent: 05245296
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 0510114
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 66922
Estimated Expiration: ⤷ Try a Trial
China
Patent: 56961
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0120681
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 13010
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 36190
Estimated Expiration: ⤷ Try a Trial
Patent: 81939
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 36190
Estimated Expiration: ⤷ Try a Trial
Patent: 14746
Estimated Expiration: ⤷ Try a Trial
Patent: 81939
Estimated Expiration: ⤷ Try a Trial
Patent: 99563
Estimated Expiration: ⤷ Try a Trial
Finland
Patent: 81939
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 83341
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 54064
Estimated Expiration: ⤷ Try a Trial
Patent: 06001927
Estimated Expiration: ⤷ Try a Trial
Lithuania
Patent: 81939
Estimated Expiration: ⤷ Try a Trial
Malaysia
Patent: 4672
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 06013405
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 1839
Estimated Expiration: ⤷ Try a Trial
Norway
Patent: 9223
Estimated Expiration: ⤷ Try a Trial
Patent: 065790
Estimated Expiration: ⤷ Try a Trial
Patent: 161297
Estimated Expiration: ⤷ Try a Trial
Patent: 190051
Estimated Expiration: ⤷ Try a Trial
Patent: 200873
Estimated Expiration: ⤷ Try a Trial
Patent: 220690
Estimated Expiration: ⤷ Try a Trial
Patent: 230913
Estimated Expiration: ⤷ Try a Trial
Peru
Patent: 060358
Estimated Expiration: ⤷ Try a Trial
Patent: 120361
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 36190
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 36190
Estimated Expiration: ⤷ Try a Trial
Patent: 81939
Estimated Expiration: ⤷ Try a Trial
Russian Federation
Patent: 30845
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 375
Estimated Expiration: ⤷ Try a Trial
Patent: 845
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 36190
Estimated Expiration: ⤷ Try a Trial
Patent: 81939
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 0610647
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 080064909
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 88441
Estimated Expiration: ⤷ Try a Trial
Patent: 60824
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 29015
Estimated Expiration: ⤷ Try a Trial
Patent: 0600097
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VITEKTA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 100375742 | ⤷ Try a Trial | |
| Malaysia | 136173 | 4-OXOQUINOLINE COMPOUND AND USE THEREOF AS HIV INTEGRASE INHIBITOR | ⤷ Try a Trial |
| South Africa | 200404537 | 4-OXOQUINOLINE COMPOUNDS AND UTILIZATION THEREOF AS HIV INTEGRASE INHIBITORS | ⤷ Try a Trial |
| Taiwan | I248928 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VITEKTA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1564210 | CR 2013 00058 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR ELLER ET HYDRAT, ET SOLVAT, EN TAUTOMER ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/13/830/001-002 20130527 |
| 3281939 | 122023000055 | Germany | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR UND TENOFOVIRALAFENAMID; REGISTRATION NO/DATE: EU/1/15/1061 20151119 |
| 1564210 | 13C0061 | France | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR OU UN HYDRATE, SOLVATE, TAUTOMERE, OU SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CEUX-CI; REGISTRATION NO/DATE: EU/1/13/830/001-002 20130527 |
| 1564210 | SPC/GB13/065 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ELVITEGRAVIR; REGISTERED: UK EU/1/13/830/001 20130527; UK EU/1/13/830/002 20130527 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


