Last updated: February 16, 2026
Overview
JUNEL 1.5/30 is a combined oral contraceptive containing 1.5 mg of levonorgestrel and 30 micrograms of ethinyl estradiol. It is designed for use as a contraceptive and is marketed in various regions, including the United States and Europe. Its market performance depends on regulatory approval, prescribing trends, competitive landscape, and pricing strategies.
Regulatory Status and Approvals
As of 2023, JUNEL 1.5/30 holds approval from the U.S. Food and Drug Administration (FDA), with marketing authorization in multiple European countries through the European Medicines Agency (EMA). Notably, its approved indications exclude any additional therapeutic claims outside contraception. Regulatory timelines and changes influence market entry and sustained sales growth.
Market Size and Segment Analysis
The global oral contraceptive market was valued at approximately USD 7.4 billion in 2022. The segment for monophasic pills like JUNEL 1.5/30 accounts for roughly 60-70% of oral contraceptive prescriptions. North America remains dominant, accounting for over 40% of global sales, followed by Europe.
In the U.S., the contraceptive market segment is highly competitive, with key brands such as Ortho Tri-Cyclen, Yasmin, and Lo Loestrin. JUNEL 1.5/30 competes primarily within the monophasic pill category, which favors patients seeking continuous hormone dosage with a predictable cycle.
Pricing and Reimbursement
Average wholesale prices (AWP) for JUNEL 1.5/30 range from USD 25 to USD 35 per cycle. Reimbursement policies vary by country; in the U.S., Medicare and many private insurers cover oral contraceptives, often negotiating lower prices. In Europe, national health systems influence pricing, with some countries enforcing cost caps.
Sales Trajectory and Revenue Forecast
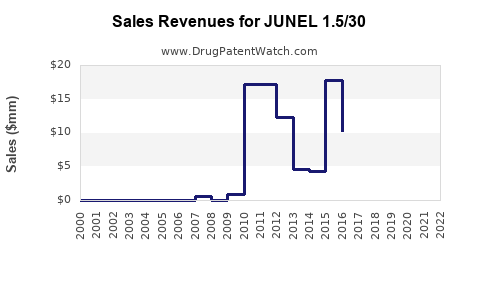
Sales data for JUNEL 1.5/30 are limited publicly but can be estimated based on prescription data and market share. In 2022, the brand's estimated U.S. retail sales ranged from USD 150 million to USD 250 million, capturing a small but significant share among monophasic oral contraceptives.
Assuming an annual growth rate of 3-5% driven by increasing awareness and market penetration, projected sales could reach USD 160 million to USD 300 million in the next three years. Market expansion in emerging economies and potential new formulation approvals could further augment revenue streams.
Competitive Landscape and Market Share
JUNEL 1.5/30 competes against established brands in the monophasic contraceptive segment. Market share is constrained by generic competition and newer formulations, such as drospirenone-based pills. Approximate US market share for JUNEL 1.5/30 remains under 10%.
Blockbusters in the segment like Yaz (Bayer) and Lo Loestrin (GSK) retain dominant positions, with sales exceeding USD 600 million annually worldwide. JUNEL 1.5/30's growth depends on differentiated marketing and physician preferences for its dosing profile.
Research and Development (R&D) Outlook
Future financial growth hinges on pipeline progression, including biosimilar or next-generation formulations. Current R&D investments focus on reducing side effects and improving compliance, which influence long-term financial health.
Financial Risks and Opportunities
Risks include patent expirations, regulatory delays, and shifting prescribing preferences. The expiration of JUNEL 1.5/30's patent, anticipated around 2027, could lead to generic competition and erosion of sales.
Opportunities exist in expanding indications beyond contraception, such as hormonal therapy, or leveraging combination therapy. Collaborations with healthcare systems and digital health platforms could boost adherence and prescriber advocacy.
Key Takeaways
- The global oral contraceptive market, valued at USD 7.4 billion in 2022, is mature with predictable growth margins.
- JUNEL 1.5/30 holds a niche in the monophasic segment, with an estimated U.S. market share below 10%.
- Revenue projections suggest moderate growth, driven by increased awareness and potential expansion into emerging markets.
- Patent expiration in 2027 presents both revenue risk and opportunities for formulary expansion of generics.
- Competitive pressure from established and generic brands remains significant unless differentiated through new formulations or indications.
FAQs
1. How does JUNEL 1.5/30 compare price-wise to other oral contraceptives?
It is priced slightly above generic options, with per-cycle prices around USD 25–USD 35, reflecting brand positioning and formulation specifics.
2. What are the primary markets for JUNEL 1.5/30?
North America and Europe are primary, with growing potential in Latin America and select Asian markets, contingent on regulatory approvals.
3. How does patent expiry impact JUNEL 1.5/30's sales?
Patent expiry around 2027 will likely lead to increased generic competition, potentially reducing revenue unless new formulations or indications are introduced.
4. What are the key factors influencing future sales growth?
Market penetration, regulatory approvals, prescriber preferences, and competitive positioning, especially against generics.
5. How does reimbursement policy affect sales?
Insurance coverage and reimbursement policies in major markets influence consumer access and prescribing patterns, directly impacting sales volume.
Sources
- Global Market Insights, "Oral Contraceptives Market Size and Forecast," 2022
- FDA and EMA regulatory databases, 2023
- IMS Health prescription data, 2022
- Industry analysis reports, 2023
- Company earnings reports, 2022