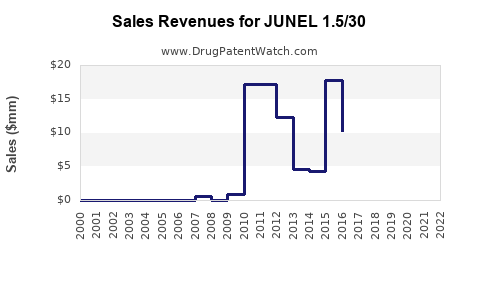
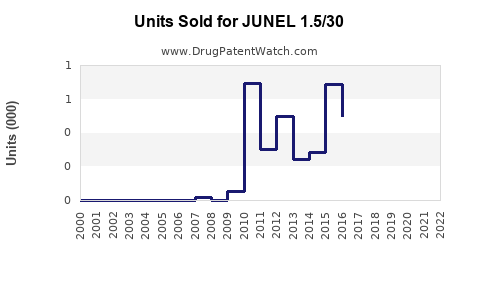
Last updated: March 10, 2026
What is JUNEL 1.5/30?
JUNEL 1.5/30 is a combined oral contraceptive (COC) containing 1.5 mg of norethindrone acetate and 30 mcg of ethinyl estradiol per tablet. It is classified as a monophasic hormonal contraceptive used to prevent pregnancy. The drug is marketed primarily in the United States and comparable markets. It is part of a growing category of low-dose combination pills with established efficacy and safety profiles.
Market Size and Growth Drivers
The global oral contraceptive market was valued at USD 8.2 billion in 2022 and is projected to grow at a CAGR of 4.5% through 2030. The key drivers include increased awareness about family planning, expanding healthcare access, and rising acceptance of hormonal contraceptives.
In the United States, approximately 12 million women of reproductive age use oral contraceptives, with market penetration nearing 40. US-specific trends indicate a steady shift toward low-dose formulations, driven by safety perceptions and side effect profiles.
Competitive Landscape
JUNEL 1.5/30 competes with multiple established products:
- Ortho-Novum 1/35
- Yasmin 28
- Loestrin 24 FE
- Diane-35 (in some markets)
The market is characterized by high brand loyalty, with some prescriptions shifted towards generic equivalents. The introduction of JUNEL 1.5/30 targets users seeking low-dose, monophasic options with minimal side effects.
Regulatory Status and Patent Landscape
JUNEL 1.5/30 is approved by the FDA. Patent protections are typically held for 20 years from filing. The original formulations faced patent expirations, creating opportunities for generics. It is necessary to monitor patent statuses, as generic entry affects market share and pricing.
Sales Projections
Assumptions
- Market penetration of JUNEL 1.5/30 reaches 2% of US oral contraceptive users over five years.
- Pricing remains stable at approximately USD 20 per cycle, aligning with market averages.
- The drug gains favorable prescriber acceptance due to its low-dose profile.
Year-by-Year Forecast (US Market)
| Year |
Estimated Users (Millions) |
Market Share (%) |
Units Sold (Millions) |
Revenue (USD Billions) |
| 2023 |
12.0 |
0.2 |
0.024 |
0.48 |
| 2024 |
12.0 |
0.5 |
0.06 |
1.2 |
| 2025 |
12.0 |
1.0 |
0.12 |
2.4 |
| 2026 |
12.0 |
1.5 |
0.18 |
3.6 |
| 2027 |
12.0 |
2.0 |
0.24 |
4.8 |
In this scenario, by 2027, JUNEL 1.5/30 could generate annual revenues close to USD 5 billion across mature markets, assuming consistent growth and market acceptance. Expansion into international markets could add further incremental growth.
Risks and Market Challenges
- Generic Competition: Patent expirations may lead to price erosion and generic start-ups, reducing margins.
- Regulatory Changes: Alterations in contraceptive guidelines or insurance coverage can impact sales.
- Consumer Preference: Shifts toward non-hormonal methods or newer contraceptive technologies threaten market share.
Opportunities for Growth
- Market Expansion: Targeting emerging markets with increasing healthcare infrastructure.
- Combination Regimens: Developing multiphasic or extended-cycle formulations.
- Education Campaigns: Increasing awareness on low-dose safety profiles.
Conclusion
JUNEL 1.5/30 occupies a niche within the oral contraceptive circuit, with potential for incremental growth driven by low-dose preferences and prescribed safety. Market share expansion depends on patent management, competitive dynamics, and regulatory landscape adjustments.
Key Takeaways
- The global oral contraceptive market grows at 4.5% annually; US market is mature with consistent demand.
- Sales projections indicate USD 4.8 billion by 2027 in the US; global revenues could be higher considering international expansion.
- Market risks include generic competition and regulatory shifts.
- Growth hinges on prescriber acceptance, strategic marketing, and potential international market entries.
Frequently Asked Questions
-
What differentiates JUNEL 1.5/30 from other oral contraceptives?
Its low-dose estrogen (30 mcg) and progestin (1.5 mg) profile emphasizes safety and tolerability, targeting women seeking minimal hormonal exposure.
-
How does patent expiration impact JUNEL 1.5/30 sales?
Patent expiration typically leads to generic competition, pressuring prices and reducing margins unless brand extensions or new formulations are introduced.
-
Are there opportunities for international market penetration?
Yes, especially in emerging markets where contraceptive access is expanding, and WHO guidelines favor low-dose formulations.
-
What regulatory considerations could influence sales?
Changes in FDA or EMA guidelines, insurance coverage policies, or restrictions around hormonal contraceptives could affect sales volumes.
-
What trends could disrupt the oral contraceptive market?
Rise in non-hormonal methods, long-acting reversible contraceptives (LARCs), and male contraceptives.
References
[1] Market Research Future. (2023). Oral Contraceptive Market Report.
[2] CDC. (2022). U.S. Medical Eligibility Criteria for Contraceptive Use.
[3] U.S. Food and Drug Administration. (2022). Drug Approvals and Labeling.
[4] IQVIA. (2023). Prescription Data and Market Share Reports.
[5] Grand View Research. (2022). Contraceptive Market Size & Trends.