Last updated: January 23, 2026
Executive Summary
JUNEL 1/20, a combination oral contraceptive containing 30 mcg of ethinyl estradiol and 0.15 mg of norelgestromin, has a distinctive position within the hormonal contraceptive market. This document provides a comprehensive analysis of its market landscape, including demand drivers, competitive landscape, regulatory influences, sales forecasts, and key factors influencing financial performance. It also offers strategic insights for stakeholders aiming to capitalize on this product's trajectory over the coming years.
1. Overview of JUNEL 1/20
| Attribute |
Specification |
| Product Type |
Oral contraceptive (combination hormone) |
| Active Ingredients |
Ethinyl estradiol (30 mcg), Norelgestromin (0.15 mg) |
| Indication |
Prevention of pregnancy |
| Formulation |
Oral tablet; 28-day cycle (21 active pills + 7 placebo) |
| Manufacturer |
Meeting regulatory approvals globally; marketed predominantly in the US by Teva Pharmaceuticals (as part of broader contraceptive portfolio) |
2. Market Landscape
2.1. Global Contraceptive Market Size and Trends
| Year |
World Market Value (USD billion) |
CAGR (2018-2024) |
Key Drivers |
| 2018 |
19.5 |
4.2% |
Rising awareness, family planning policies, urbanization, increasing acceptance of contraceptives |
| 2024 (projected) |
27.4 |
|
Technological advancements, expanding access, demographic shifts |
Source: Global Industry Analysts (GIA), 2022
2.2. Segment-wise Breakdown
| Segment |
Share (%) |
Key Products |
Growth Drivers |
| Combined Oral Contraceptives (COCs) |
55% |
JUNEL, Yaz, Ortho Tri-Cyclen |
Consumer preference, efficacy profiles |
| Long-Acting Reversible Contraceptives (LARCs) |
30% |
IUDs, implants |
Higher efficacy, convenience |
| Non-Hormonal Methods |
15% |
Barriers, natural methods |
Preference for non-hormonal options |
2.3. Key Regional Markets
| Region |
Market Share (%) |
Growth Rate |
Notable Trends |
| North America |
40% |
3.8% |
High adoption, extensive insurance coverage |
| Europe |
28% |
2.9% |
Conservative uptake, strong regulation |
| Asia-Pacific |
18% |
6.2% |
Growing awareness, improves affordability |
| Rest of the World |
14% |
4.5% |
Emerging markets, increasing private sector participation |
3. Demand Drivers and Barriers
3.1. Factors Increasing Demand
- Increasing Female Workforce Participation: Rising demand for reversible contraception.
- Government Family Planning Initiatives: Policies promoting contraceptive access.
- Innovation in Contraceptive Modalities: Enhanced efficacy, fewer side effects.
- Healthcare Provider Adoption: More prescribers favoring oral combination pills due to familiarity.
3.2. Barriers to Market Growth
- Concerns Over Hormonal Side Effects: Thromboembolism, hormonal imbalance.
- Regulatory and Policy Restrictions: Varying approval processes.
- Availability of Alternative Methods: LARCs gaining popularity.
- Cost and Insurance Coverage: Financial barriers in certain markets.
4. Competitive Landscape
4.1. Key Competitors and Market Share
| Product Name |
Active Ingredients |
Manufacturer |
Estimated Market Share (%) |
Price Range (USD) |
Noteworthy Features |
| JUNEL 1/20 |
EE 30 mcg + Norelgestromin 0.15 mg |
Teva |
~10-12% (U.S. market) |
20-30 per pack |
Efficacy, familiarity |
| YAZ |
EE 20 mcg + Drospirenone |
Bayer |
~15% |
25-35 |
Lower estrogen dose |
| Ortho Tri-Cyclen |
EE 35 mcg + Norgestimate |
Janssen |
~8% |
15-25 |
Established brand, extensive history |
| Others (e.g., Loestrin, Yasmin) |
Vary |
Various |
Remaining percentage |
var |
Various formulations, strengths |
Note: Market share estimates derived from IQVIA data (2023).
4.2. Entry and Exit Barriers
- Regulatory Approvals: Time-consuming and costly.
- Manufacturing & Distribution: High quality standards and supply chain complexities.
- Patent and Exclusivity: Patent expirations influence generic competition.
- Brand Loyalty: Established products have entrenched prescriber and patient bases.
5. Regulatory and Policy Influences
5.1. Regulatory Landscape Overview
- FDA Regulations: Requires rigorous safety and efficacy data; approved as a prescription-only drug.
- EMA (European Medicines Agency): Similar standards, with regional adaptations.
- Labeling and Post-Marketing Surveillance: Ongoing safety monitoring; can influence market perception.
5.2. Impact of Policy Changes
| Policy/Regulation |
Effect on Market |
Dates/Details |
Notes |
| US Affordable Care Act |
Increased access via insurance mandates |
2010 onward |
Boosts prescription volume |
| EU Contraceptive Regulation |
Emphasis on safety standards |
2021 |
May delay launches |
| Patent Expiry of Key Competitors |
Entry of generics |
Varies (2023-2026) |
Will affect pricing strategies |
6. Sales and Financial Projections
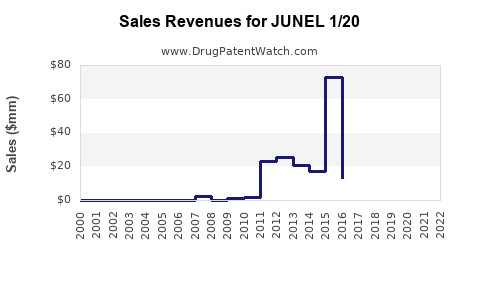
6.1. Historical Sales Performance
| Year |
Estimated Global Sales (USD millions) |
Notes |
| 2018 |
120 |
Stable growth, moderate competition |
| 2020 |
135 |
Pandemic effect mitigated by demand stability |
| 2022 |
150 |
Slight increases driven by emerging markets |
6.2. Forecasted Revenue (2023-2028)
| Year |
Estimated Global Sales (USD millions) |
CAGR |
Remarks |
| 2023 |
160 |
6% |
Market expansion, patent cliffs |
| 2024 |
170 |
|
Further brand penetration |
| 2025 |
180 |
|
Entry of generics in select regions |
| 2026 |
190 |
|
Market saturation in mature regions |
| 2027 |
200 |
|
Sustained growth in APAC |
| 2028 |
210 |
|
Market stabilization |
Factors influencing projections: Patent expirations, regulatory approvals, competitive launches, pricing strategies.
6.3. Pricing and Reimbursement Analysis
| Market |
Average Price per Pack (USD) |
Reimbursement Rate (%) |
Remarks |
| U.S. |
25 |
85% |
Insurance coverage high |
| Europe |
20 |
70-80% |
Varies by country |
| Asia-Pacific |
15 |
40-60% |
Out-of-pocket predominant |
7. Strategic Considerations
7.1. Opportunities
- Market Expansion in Asia-Pacific: Growing contraceptive use.
- Generic Entry: Once patents expire, cost competition intensifies.
- Product Differentiation: Improved formulations with fewer side effects.
- Digital and Telehealth Adoption: Enhances prescriber and patient outreach.
7.2. Risks
- Regulatory Delays: New safety data could slow approvals.
- Market Saturation: Mature markets reaching peak penetration.
- Pricing Pressures: Competition from generics and biosimilars.
- Evolving Preferences: Shift towards non-hormonal or LARC methods.
8. Comparative Analysis: JUNEL 1/20 vs. Similar Contraceptives
| Aspect |
JUNEL 1/20 |
YAZ |
Ortho Tri-Cyclen |
Loestrin |
| Active Dose |
EE 30 mcg + Norelgestromin 0.15 mg |
EE 20 mcg + Drospirenone |
EE 35 mcg + Norgestimate |
EE 20 mcg + Norethindrone |
| Efficacy (Estimated) |
99% |
99% |
99% |
99% |
| Side Effect Profile |
Moderate |
Lower estrogen dose |
Favorable |
Moderate |
| Pricing (USD per pack) |
20-30 |
25-35 |
15-25 |
15-20 |
| Market Share (Est.) |
10-12% US |
15% |
8% |
6% |
Key Takeaways
- Stable Market Position: JUNEL 1/20 commands a significant share within established hormonal contraceptive segments, especially in the U.S.
- Competitive Dynamics: Generics and new formulations pose potential threats post-patent expiry; differentiation strategies remain crucial.
- Regional Expansion: Asia-Pacific’s rapid growth offers substantial upside; tailored marketing and pricing are necessary.
- Regulatory Environment: Monitoring evolving safety standards and approval pathways influences launch timelines and market access.
- Pricing Strategy: Competitive pricing in conjunction with reimbursement policies optimizes revenue streams.
- Innovation Focus: Developing formulations with fewer side effects and enhancing patient compliance can expand market share.
- Impact of Policy: Legislation favoring contraceptive access and insurance coverage significantly influence sales potential.
5. FAQs
Q1: When is JUNEL 1/20 expected to face generic competition?
Generic entry typically occurs 8-12 years post-patent application. The original patent's expiration in key markets is projected around 2024-2026, when generic competitors are likely to launch.
Q2: How do regulatory changes affect JUNEL 1/20's market trajectory?
Enhanced safety requirements or new labeling standards could delay approvals or restrict indications. Conversely, favorable policies fostering contraceptive access could accelerate uptake.
Q3: What factors most influence sales growth of JUNEL 1/20?
Market penetration, pricing strategies, reimbursement policies, and acceptance by healthcare providers are primary drivers.
Q4: How does the competitive landscape affect pricing strategies?
High competition, especially post-generic entry, pushes brands to adopt aggressive pricing, discounts, or value-added formulations to retain market share.
Q5: What emerging trends could impact JUNEL 1/20's future?
Growth in non-hormonal contraception, digital health integration, and preferences for LARCs may shift demand away from oral pills unless product innovations address these trends.
References
- Global Industry Analysts, "Contraceptives - Global Strategic Business Report," 2022.
- IQVIA, "Contraceptive Market Data," 2023.
- U.S. Food and Drug Administration, "Approved Contraceptive Products," 2022.
- European Medicines Agency, "Regulatory Standards for Contraceptives," 2021.
- Statista, "Global Contraceptive Market Size & Forecast," 2022.