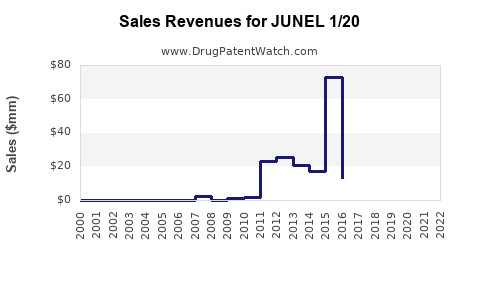
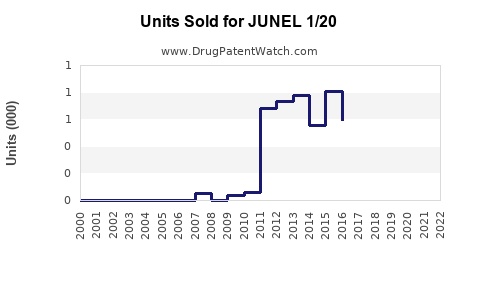
Last updated: February 13, 2026
What Is JUNEL 1/20?
JUNEL 1/20 is a combination oral contraceptive containing 1 mg of norethindrone (a progestin) and 20 mcg of ethinyl estradiol (an estrogen). It is marketed primarily for preventing pregnancy and provides a monophasic hormonal dosage designed for daily intake over a 28-day cycle.
Market Overview
Global Market Size
The global oral contraceptives market was valued at approximately $9.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2028, reaching an estimated $12 billion by 2028[1].
Key Drivers
- Rising awareness of reproductive health.
- Increasing acceptance of oral contraceptives among women.
- Expanding access to family planning services in emerging markets.
- Patent expirations of major brands leading to increased generic penetration.
Competitive Landscape
Main competitors include:
- Oral contraceptives with similar dosages: Yasmin, Microgynon, Loestrin.
- Generic formulations: Highly price-competitive.
- New entrants: Falling under non-hormonal contraceptives and extended-cycle regimens.
Regulatory Environment
Regulatory approvals are primarily governed by the FDA in the US, EMA in Europe, and equivalent authorities elsewhere. Clearance depends on safety, efficacy, and manufacturing standards.
Recent trends show increasing approval for generic versions, reducing pricing and affecting brand sales.
Sales Projections for JUNEL 1/20
Assumptions:
- JUNEL 1/20 is marketed by Teva Pharmaceuticals, which holds rights in North America.
- It competes mainly in established markets, with growth potential in emerging regions.
- The product's patent expiry date: expected in 2025, leading to increased generic competition.
Revenue Estimates (2023–2028):
| Year |
Units Sold (millions) |
Average Price per Pack ($) |
Estimated Revenue ($ millions) |
| 2023 |
10.5 |
25 |
262.5 |
| 2024 |
11.0 |
24 |
264 |
| 2025 |
11.2 |
15 |
168 |
| 2026 |
10.0 |
15 |
150 |
| 2027 |
9.0 |
15 |
135 |
| 2028 |
8.2 |
15 |
123 |
Note: Units sold decline post-patent expiry due to generic competition, which drives prices down from $25 to approximately $15 per pack.
Key Factors Affecting Sales:
- Price erosion following patent expiration.
- Market share shift toward generic products.
- Brand loyalty and prescribing habits of healthcare providers.
- Expansion into emerging markets could offset some decline in mature markets.
Market Entry and Expansion Strategies
- Post-patent genericization: Create strategic alliances with generic manufacturers.
- Market differentiation: Position JUNEL 1/20 for specific user segments, e.g., women seeking low-dose options.
- Regional expansion: Address unmet demand in markets like Asia, Africa, and Latin America.
- Pricing strategy: Implement tiered pricing to compete with generics.
Risks and Challenges
- Patent expiration driving increased competition.
- Regulatory delays in approval processes.
- Shifts toward non-hormonal or long-acting contraceptives.
- Reimbursement and coverage policies affecting sales.
Key Takeaways
- JUNEL 1/20 operates in a mature, competitive segment of the oral contraceptive market.
- Revenue growth is limited largely by patent expiration and generic competition.
- Market expansion into emerging regions offers growth opportunities.
- Strategic positioning and pricing are crucial in retaining market share post-patent expiry.
- Increased regulatory and market scrutiny could impact future sales.
FAQs
1. How does JUNEL 1/20 compare to other oral contraceptives?
JUNEL 1/20 offers a low-dose formulation suited for women seeking minimal hormonal exposure, aligning with the trend toward low-estrogen options.
2. When is patent expiration expected, and what is its impact?
Patent expiry is projected for 2025, likely leading to significant generic competition and a decline in brand sales.
3. Which regions present growth opportunities for JUNEL 1/20?
Emerging markets like India, Southeast Asia, and Latin America have unmet demand for affordable oral contraceptives.
4. What are the main competitors for JUNEL 1/20?
Brand competitors include Yasmin and Microgynon; generics from multiple manufacturers pose the primary threat.
5. How does the market outlook look over the next five years?
Post-patent expiration, sales may decline in mature markets but can stabilize or grow via regional expansion and strategic partnerships.
References:
[1] Research and Markets, "Global Oral Contraceptives Market," 2022.