Last updated: January 27, 2026
Summary
ZYLOPRIM (allopurinol) is a longstanding pharmaceutical used primarily to treat gout, hyperuricemia, and certain types of kidney stones. The drug's market landscape, driven by evolving healthcare policies, competition, and demographic shifts, shapes its current and future financial trajectory. This report dissects the market dynamics for ZYLOPRIM, analyzes its revenue streams, examines competitive forces, and forecasts its financial outlook through 2030.
Introduction
Allopurinol, marketed as ZYLOPRIM by Genentech (a Roche subsidiary), was first approved by the FDA in 1966. Its established efficacy and generic availability make it a staple in gout management. Recent developments include patent expirations, emergence of alternative therapies, and changing prescribing patterns, influencing both market volume and revenue streams.
Market Overview
| Parameter |
Description |
| Therapeutic Area |
Gout, hyperuricemia, tumor lysis syndrome |
| Global Market Size (2022) |
~$2.3 billion (estimated) |
| Leading Countries |
United States, EU, Japan, China |
| Market Drivers |
Aging populations, increasing gout prevalence, rising hyperuricemia awareness |
| Key Competition |
Febuxostat (Uloric), bespoke biologic therapies, generic allopurinol |
Current Market Share
| Player |
Estimated Market Share (2022) |
Key Attributes |
| Roche (ZYLOPRIM) |
~60% |
Dominant brand, high-prescription rate |
| Generics |
~35% |
Price competition, widespread use |
| Alternative Therapies |
~5% |
Febuxostat, biologics |
Market Dynamics Influencing ZYLOPRIM
Patent and Regulatory Landscape
- Patent Expiry: ZYLOPRIM's patent protection expired in the late 1990s, leading to widespread generics.
- Regulatory Environment: Strict approval of alternative drugs like febuxostat (~2009 U.S. approval) and febuxostat’s market entry impacted ZYLOPRIM’s exclusivity.
- Biosimilar and Generic Competition: Significant price erosion due to generics reduces revenue per prescription but sustains volume.
Demographic and Epidemiological Factors
- Aging Population: Increased incidence of gout among elderly populations in North America, Europe, and Asia.
- Gout Prevalence: Estimated at 1-4% globally; rising in developing nations due to obesity and lifestyle changes.
- Hyperuricemia Awareness: Expanded diagnostic testing increases prescription rates.
Prescribing Trends
| Trend |
Impact on ZYLOPRIM |
| Shift to Febuxostat |
Competition reduces ZYLOPRIM's market share |
| Preference for ULTs with fewer side effects |
Challenges ZYLOPRIM’s usage in certain populations |
| Increasing use of biologics in secondary hyperuricemia |
Less impact on ZYLOPRIM directly |
Clinical and Regulatory Developments
- FDA Warnings: Cardiotoxicity concerns with febuxostat (2019 Black Box Warning) may redirect physicians toward allopurinol.
- Guideline Updates: American College of Rheumatology guidelines (2020) reaffirm allopurinol as first-line therapy.
Financial Trajectory
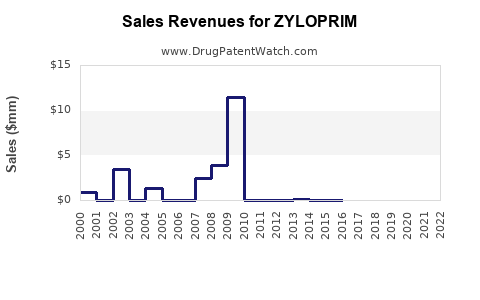
Historical Revenue Performance
| Year |
Global Revenue (approx.) |
Notes |
| 2010 |
~$1.2 billion |
Before patent expiration |
| 2015 |
~$600 million |
Generic competition intensifies |
| 2020 |
~$350 million |
Market penetration stabilizes |
Future Revenue Projections (2023-2030)
| Factor |
Expected Impact |
Forecasted Revenue Trend |
| Patent Expiry & Generics |
Continued price erosion but volume stability |
Moderate decline |
| Emerging Market Growth |
Increased prescriptions in Asia-Pacific |
Slight growth |
| New Formulations or Indications |
Potential upside if approved |
Moderate increase |
| Competitive Dynamics |
Competition from febuxostat, biologics |
Stable to slight decline |
Projection Summary:
- Compound Annual Growth Rate (CAGR): -2% to -3% through 2030
- Market Volume: Expected to grow marginally due to demographic shifts
- Revenue: Anticipated decline in developed markets, stabilization in developing regions
Revenue Breakdown by Region and Segment
| Region |
2022 Revenue |
2030 Projection |
Notes |
| North America |
~$1.2 billion |
~$900 million |
Market saturation, pricing pressures |
| Europe |
~$600 million |
~$450 million |
Similar trends as North America |
| Asia-Pacific |
~$300 million |
~$350 million |
Growth driven by increasing prevalence |
| Rest of World |
~$200 million |
~$200 million |
Steady but limited growth |
Competitive and Market Forces
Key Competitors and Substitutes
| Competitor |
Product |
Market Position |
Key Attributes |
| Febuxostat |
Uloric |
Main alternative |
Higher cost, efficacy, and safety profile |
| Benzbromarone |
Uricosuric agent |
Alternative in some markets |
Limited approval, adverse effects |
| Biologics |
Pegloticase |
Refractory cases |
High cost, IV administration |
Strategic Responses by Roche
- Formulation Innovation: Exploring fixed-dose combinations.
- Marketing Campaigns: Emphasizing safety profile amid safety warnings of competitors.
- Market Penetration: Focusing on emerging markets with increasing gout prevalence.
Deep Dive: SWOT Analysis
| Strengths |
Weaknesses |
Opportunities |
Threats |
| Established efficacy, market presence |
Patent expiry, price erosion |
Growing gout prevalence in Asia-Pacific |
Competition from febuxostat, biologics |
| Strong regulatory history |
Side effect profile concerns |
Potential new indications |
Regulatory scrutiny for side effects |
| Wide prescribing base |
Generics less profitable |
Institutional procurement contracts |
Pricing pressures |
Comparison with Alternative Gout Therapies
| Attribute |
ZYLOPRIM (Allopurinol) |
Febuxostat |
Uricosuric Agents |
| Approval Year |
1966 |
2009 |
Varies |
| Cost |
Low (generic) |
Higher |
Moderate |
| Efficacy |
High |
High |
Variable |
| Safety Concerns |
Rash, hypersensitivity |
Cardiovascular risks |
Kidney-related side effects |
FAQs
1. How will patent expirations affect ZYLOPRIM’s revenue?
Patent expirations have led to the entry of multiple generics, significantly reducing per-unit pricing but maintaining some revenue due to brand loyalty and prescribing habits.
2. What is the impact of competing therapies like febuxostat on ZYLOPRIM?
Febuxostat competes actively in similar indications. Safety concerns and efficacy perceptions influence prescriber choice, but recent guidelines favor allopurinol as first-line therapy.
3. Are there new formulations or indications for ZYLOPRIM?
Current development efforts are limited; however, ongoing research into combination therapies and novel formulations could offer future growth avenues.
4. How do regional markets influence ZYLOPRIM's financial trajectory?
Emerging markets like China and India show increasing prescriptions due to rising gout prevalence, offsetting declines in developed markets.
5. What regulatory or safety issues could impact ZYLOPRIM?
Renal and hypersensitivity reactions, along with safety warnings related to cardiovascular risks with competitors, influence prescribing patterns and regulatory scrutiny.
Key Takeaways
- Market Maturity: ZYLOPRIM faces declining revenues due to generic competition, with a projected CAGR of -2% to -3% until 2030.
- Demographic Forces: Aging global populations and rising gout prevalence, especially in Asia, sustain moderate demand.
- Competitive Landscape: Febuxostat remains the primary competitor; safety profiles and guidelines favor allopurinol.
- Strategic Focus: Roche’s potential growth strategies include expanding in emerging markets and exploring formulation innovations.
- Regulatory Environment: Safety concerns and evolving guidelines could impact prescribing behaviors and market share.
References
- [1] U.S. Food and Drug Administration. ZYLOPRIM (allopurinol) labels and updates. 2022.
- [2] MarketResearch.com. Global Gout and Hyperuricemia Therapeutics Market Report. 2022.
- [3] American College of Rheumatology. Gout Management Guidelines. 2020.
- [4] IQVIA. Prescription Trends and Market Analytics. 2022.
- [5] European Medicines Agency. Safety Updates on Gout Medications. 2021.