Last updated: February 19, 2026
Zaleplon, a nonbenzodiazepine hypnotic agent, targets the GABAA receptor to induce sleep. Its market performance is shaped by evolving sleep disorder treatment guidelines, generic competition, and the introduction of novel therapeutic modalities. The drug's patent exclusivity has expired in major markets, leading to price erosion and a shift towards generic formulations.
What is the current market size and projected growth for zaleplon?
The global market for zaleplon is primarily driven by the prevalence of insomnia and other sleep disorders. Precise market size data for zaleplon alone is challenging to isolate due to its classification within broader hypnotic and sedative drug categories. However, the overall sedative and hypnotic drug market, which includes zaleplon and its competitors, was valued at approximately $2.7 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% to reach $3.9 billion by 2029 [1].
Zaleplon's specific contribution to this market has diminished following patent expirations. The primary growth drivers for the broader market include an aging global population, increased stress and anxiety levels, and greater awareness of sleep disorder management. While zaleplon plays a role in treating short-term insomnia, its market share is significantly impacted by the availability of generic alternatives and newer, potentially more targeted therapies.
Key factors influencing zaleplon's trajectory within this market include:
- Genericization: The expiration of key patents for zaleplon has led to widespread generic availability, significantly reducing pricing power for brand-name formulations and increasing competition among generic manufacturers.
- Therapeutic Alternatives: The development and adoption of alternative insomnia treatments, including other nonbenzodiazepine hypnotics (e.g., zolpidem, eszopiclone), melatonin receptor agonists (e.g., ramelteon), and orexin receptor antagonists (e.g., suvorexant, lemborexant), compete for market share.
- Treatment Guidelines: Evolving clinical practice guidelines for insomnia management influence prescribing patterns. Current guidelines often emphasize non-pharmacological interventions and may favor medications with different safety profiles or mechanisms of action.
- Regulatory Landscape: Post-market surveillance and any potential changes in regulatory recommendations regarding the use of hypnotics can impact market demand.
What are the key intellectual property protections for zaleplon?
Zaleplon was originally developed by Wyeth (now part of Pfizer) and patented under U.S. Patent No. 4,504,469, titled "Pyrazolopyrimidine derivatives" [2]. This patent, initially filed in the early 1980s, provided the foundational intellectual property for zaleplon.
The key intellectual property milestones for zaleplon include:
- Original Composition of Matter Patent: U.S. Patent No. 4,504,469 provided exclusivity for the zaleplon molecule itself. This patent has long expired.
- Method of Use Patents: Additional patents may have covered specific methods of treating insomnia or particular dosages. These also have expired.
- Formulation Patents: Patents related to specific formulations or delivery mechanisms could have offered a limited period of extended protection. Information regarding the expiration of specific formulation patents is proprietary to manufacturers.
- Exclusivity Periods: Upon Food and Drug Administration (FDA) approval in the United States in 2000, zaleplon received 5 years of New Chemical Entity (NCE) exclusivity. This exclusivity period has long since concluded.
The expiration of these patents has opened the market to generic manufacturers. Companies seeking to produce generic zaleplon must demonstrate bioequivalence to the reference listed drug. The absence of robust, ongoing patent protection for zaleplon means that market entry for new generic players is relatively unhindered by IP barriers, primarily driven by manufacturing capabilities and regulatory approvals.
What is the competitive landscape for zaleplon?
The competitive landscape for zaleplon is characterized by a significant presence of generic alternatives and a range of other pharmacological treatments for insomnia.
Direct Competitors (Other Hypnotics):
- Zolpidem (Ambien, generics): This is zaleplon's most direct competitor, also a GABAA receptor agonist. Zolpidem has a larger market share and a longer history of availability, with numerous generic versions. It is available in immediate-release and extended-release formulations.
- Eszopiclone (Lunesta, generics): Another GABAA receptor modulator, eszopiclone offers an enantiomerically pure form of zopiclone. Like zolpidem, it faces significant generic competition.
- Zopiclone (Imovane, generics): While less common in the U.S., zopiclone is widely used in Europe and other international markets.
Emerging and Alternative Therapies:
- Melatonin Receptor Agonists (e.g., Ramelteon (Rozerem)): These drugs target the melatonin receptors, affecting the body's natural sleep-wake cycle. They generally have a different safety profile than GABAA agonists and are not typically associated with dependence or abuse potential.
- Orexin Receptor Antagonists (e.g., Suvorexant (Belsomra), Lemborexant (Dayvigo)): These newer agents block the wake-promoting effects of orexin neuropeptides. They represent a distinct pharmacological class and are often prescribed for sleep-onset and sleep-maintenance insomnia.
- Antidepressants with Sedating Properties (e.g., Trazodone): Off-label use of certain antidepressants is common for insomnia management, particularly in patients with comorbid depression or anxiety.
- Antihistamines and Benzodiazepines: While older classes, some antihistamines are still used for mild insomnia, and benzodiazepines, despite their association with dependence, remain prescribed for specific indications.
Generic Manufacturers: The market for zaleplon is dominated by generic manufacturers, including companies like Teva Pharmaceuticals, Mylan (now Viatris), Aurobindo Pharma, and Apotex. These companies compete primarily on price and market access.
The competition is fierce, driven by price wars among generic producers and the continuous introduction of novel sleep aids with potentially improved efficacy or safety profiles. Zaleplon's market position is therefore constrained by the established presence of older generics and the innovation pipeline of new insomnia therapeutics.
What is the historical financial performance and revenue trajectory for zaleplon?
Due to the expiration of its primary patents and the subsequent genericization, tracking the precise revenue trajectory of branded zaleplon is difficult. Post-patent expiry, revenue shifts from the innovator company (Wyeth/Pfizer) to numerous generic manufacturers. Publicly available financial reports typically aggregate sales data for broad drug categories or specific therapeutic areas rather than individual generic drugs.
Pre-Genericization (Innovator Era):
During its period of market exclusivity, zaleplon generated significant revenue for Wyeth. While specific annual revenue figures for zaleplon are not consistently reported by Wyeth in its historical financial disclosures (often consolidated into broader CNS or sleep aid portfolios), its peak sales would have occurred in the years following its U.S. FDA approval in 2000 and before the widespread availability of generics in the mid-to-late 2000s. For context, the market for sleep aids was substantial, with blockbuster drugs generating hundreds of millions annually.
Post-Genericization (Generic Era):
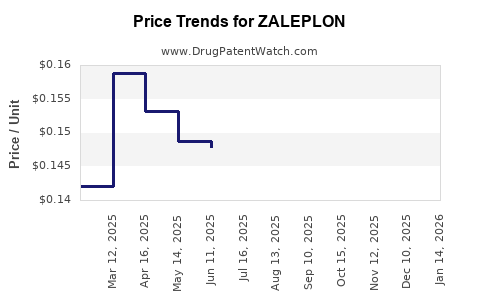
- Price Erosion: The introduction of generic zaleplon led to a rapid and substantial decline in pricing. The average selling price (ASP) for generic zaleplon is a fraction of the original branded price.
- Volume Driven Sales: For generic manufacturers, revenue is driven by sales volume rather than high per-unit prices. Companies aim for broad market penetration and capture a significant portion of prescriptions filled by generics.
- Fragmented Revenue: The revenue generated by zaleplon is now distributed among multiple generic pharmaceutical companies. No single entity reports significant revenue specifically attributable to zaleplon as a branded product.
- Market Share Competition: Generic companies compete intensely to secure contracts with pharmacy benefit managers (PBMs) and to be listed on formularies, influencing prescription volume.
Estimates and Trends:
While direct revenue figures are unavailable, the trend for zaleplon's revenue trajectory is clear: a sharp decline from its branded peak, followed by a plateauing at significantly lower price points due to generic competition. The market for zaleplon, as an individual generic drug, is likely stable but not experiencing substantial growth. Its usage is sustained by its established efficacy for short-term insomnia and its affordability as a generic option.
What are the regulatory considerations and market access challenges for zaleplon?
Zaleplon, like other sedative-hypnotics, faces specific regulatory and market access considerations due to its pharmacological class and potential for misuse.
Regulatory Considerations:
- Controlled Substance Classification: In many countries, including the United States, zaleplon is classified as a controlled substance (e.g., Schedule IV under the U.S. Controlled Substances Act) [3]. This classification stems from its potential for dependence and abuse.
- Implications: This requires specific prescribing, dispensing, and record-keeping protocols, which can add administrative burden for healthcare providers and pharmacists. It also restricts accessibility compared to non-controlled substances.
- FDA/EMA Labeling Requirements: Regulatory agencies mandate specific labeling regarding efficacy, safety, potential side effects (e.g., next-day drowsiness, amnesia, complex sleep behaviors), contraindications, and warnings. Label updates are subject to regulatory review and approval.
- Post-Market Surveillance: Regulatory bodies conduct ongoing monitoring of adverse events. Any significant safety signals can lead to label changes, prescribing restrictions, or even market withdrawal.
- Manufacturing and Quality Control: Generic manufacturers must adhere to stringent Good Manufacturing Practices (GMP) to ensure product quality, purity, and consistency. Regulatory inspections are routine.
Market Access Challenges:
- Formulary Restrictions: Due to its controlled substance status and the availability of numerous alternatives, zaleplon may face restrictions on insurance formularies. Payers may require prior authorization, step-therapy protocols (requiring trial of less expensive or non-controlled agents first), or impose quantity limits.
- Physician Prescribing Habits: While established, physician prescribing habits are influenced by clinical guidelines, personal experience, perceived safety profiles of alternatives, and patient insurance coverage. The shift towards non-pharmacological treatments for insomnia can also reduce overall prescription volumes for all hypnotic agents.
- Cost-Effectiveness: As a generic drug, zaleplon is generally cost-effective. However, its value proposition is constantly weighed against other generic options and newer, potentially more effective but expensive, sleep aids.
- Stigma Associated with Sleep Medications: There can be a societal and clinical stigma associated with prescribing and using prescription sleep medications, including zaleplon, due to concerns about dependence and long-term efficacy.
- Competition from Non-Prescription Options: The increasing availability and marketing of over-the-counter (OTC) sleep aids (e.g., antihistamines, melatonin supplements) can divert some demand from prescription hypnotics, although these often target milder forms of sleep disturbance.
Navigating these regulatory and market access hurdles is critical for generic manufacturers to maintain and grow their share in the zaleplon market.
What is the future outlook and strategic considerations for zaleplon?
The future outlook for zaleplon is characterized by continued reliance on its generic market presence, with limited prospects for significant growth. Strategic considerations for stakeholders revolve around managing existing market share and navigating an evolving sleep disorder treatment landscape.
Future Outlook:
- Stable Generic Demand: Zaleplon is expected to maintain a stable, albeit modest, demand as a cost-effective option for short-term insomnia treatment. Its established efficacy and affordability ensure its continued use, particularly in populations where cost is a primary concern.
- Intensified Generic Competition: The generic market is highly competitive. Further price erosion and market share battles among generic manufacturers are likely. Profit margins on individual units will remain low.
- Evolving Treatment Paradigms: The shift towards non-pharmacological interventions and newer drug classes (e.g., orexin antagonists) for chronic insomnia will continue to shape the overall sleep market. This may limit the expansion of zaleplon's patient base.
- Regulatory Scrutiny: As a Schedule IV controlled substance, zaleplon will remain under regulatory oversight. Any shifts in controlled substance policy or safety recommendations could impact its availability or prescribing patterns.
- Limited Innovation: Given its patent expiry and the mature stage of its lifecycle, significant investment in new zaleplon formulations or novel applications is unlikely.
Strategic Considerations:
- Cost Optimization in Manufacturing: Generic manufacturers must focus on optimizing production costs to maintain profitability in a low-margin environment. This includes efficient supply chain management and high-volume manufacturing.
- Market Access and Payer Negotiations: Successfully negotiating with PBMs and health insurers to secure favorable formulary placement and minimize prior authorization hurdles will be crucial for maintaining prescription volumes.
- Targeted Marketing: For companies that still market a branded generic or have significant distribution channels, targeted marketing to healthcare providers who routinely prescribe short-acting hypnotics for specific patient profiles could be a strategy.
- Portfolio Diversification: Companies heavily reliant on older generics like zaleplon should consider diversifying their portfolios with newer, higher-margin products or moving into more dynamic therapeutic areas to mitigate risks associated with a mature market.
- Focus on Compliance and Safety: Maintaining rigorous compliance with controlled substance regulations and ensuring high product quality are paramount to avoid regulatory action that could jeopardize market access.
Zaleplon will likely persist as a component of the insomnia treatment armamentarium, primarily as an affordable generic option, but its strategic importance is diminished in an era of evolving sleep medicine.
Key Takeaways
Zaleplon’s market is characterized by a mature, genericized landscape. Following patent expiries, revenue has shifted to numerous generic manufacturers, with significant price erosion. The drug faces intense competition from other hypnotics, including zolpidem and eszopiclone, as well as newer drug classes like orexin antagonists and melatonin receptor agonists. Regulatory classification as a Schedule IV controlled substance imposes additional compliance burdens and market access challenges. The future outlook points to a stable but low-growth generic market, necessitating a focus on cost optimization and market access for manufacturers, while limited innovation is expected.
Frequently Asked Questions
- What is the primary mechanism of action for zaleplon?
Zaleplon acts as a positive allosteric modulator at GABAA receptors, specifically binding to the benzodiazepine-1 (BZ1) receptor subtype. This enhances the inhibitory effects of the neurotransmitter GABA, leading to sedation and hypnosis.
- Is zaleplon considered a long-acting or short-acting sleep medication?
Zaleplon is classified as a short-acting hypnotic agent. It has a rapid onset of action and a short elimination half-life (approximately 1-1.5 hours), making it suitable for the treatment of short-term sleep-onset insomnia.
- What are the main side effects associated with zaleplon use?
Common side effects include drowsiness, dizziness, headache, and nausea. Less common but serious side effects can include amnesia, complex sleep behaviors (e.g., sleepwalking, sleep-driving), and potential for dependence or withdrawal symptoms with prolonged use or abrupt discontinuation.
- Can zaleplon be used for chronic insomnia?
Zaleplon is generally recommended for the short-term treatment of insomnia. Due to its short duration of action and potential for tolerance or dependence, it is not typically the preferred treatment for chronic insomnia, for which non-pharmacological therapies and other medication classes are often considered.
- How does zaleplon compare to zolpidem in terms of efficacy and safety?
Both zaleplon and zolpidem are GABAA receptor agonists used for short-term insomnia. Clinical studies suggest similar efficacy for sleep onset, but zaleplon's shorter half-life may result in less residual next-day drowsiness. Both carry risks of dependence, abuse, and complex sleep behaviors. The choice between them often depends on individual patient response, prescriber preference, and formulary coverage.
Citations
[1] Grand View Research. (2023). Sedative and Hypnotic Drugs Market Size, Share & Trends Analysis Report By Drug Type (Benzodiazepines, Non-benzodiazepines, Others), By Disease (Insomnia, Anxiety, Seizures, Others), By Distribution Channel, By Region, And Segment Forecasts, 2023-2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/sedative-hypnotic-drugs-market
[2] U.S. Patent 4,504,469. (1985). Pyrazolopyrimidine derivatives. Retrieved from USPTO Patent Full-Text Database.
[3] U.S. Drug Enforcement Administration. (n.d.). Controlled Substances Act - Schedules. Retrieved from https://www.dea.gov/drug-scheduling