Last updated: February 19, 2026
XOPENEX HFA (levalbuterol tartrate) is a short-acting beta-agonist (SABA) bronchodilator used for the treatment of bronchospasm in patients aged 4 years and older with reversible obstructive airway disease. The drug is administered via a metered-dose inhaler (MDI).
What is the current market position of XOPENEX HFA?
XOPENEX HFA is marketed by Sunovion Pharmaceuticals. Its primary indication is for the treatment of asthma. The SABA market is characterized by established generics and a strong clinical history for existing treatments. XOPENEX HFA, as a single-isomer version of albuterol, was developed to potentially reduce cardiac side effects associated with racemic albuterol.
The drug's market position is influenced by its efficacy profile, physician prescribing habits, patient access, and the competitive landscape of other SABAs and long-acting bronchodilators.
What are the key patents protecting XOPENEX HFA and their expiration dates?
The patent landscape for XOPENEX HFA is critical to understanding its market exclusivity and the potential for generic competition. Key patents and their approximate expiration timelines are crucial for strategic planning.
| Patent Number |
Title |
Issue Date |
Expiration Date (Approximate) |
Status |
| 5,714,502 |
Substantially Enantiopure R-alpha-salbutamol salts and their preparation |
March 3, 1998 |
March 3, 2015 |
Expired |
| 6,020,354 |
Enantiopure R-alpha-salbutamol formulations |
February 1, 2000 |
February 1, 2017 |
Expired |
| 7,074,446 |
R-salbutamol formulations |
July 11, 2006 |
July 11, 2023 |
Expired |
| 7,534,771 |
R-salbutamol formulations |
May 19, 2009 |
May 19, 2026 |
Active |
| 7,977,317 |
R-salbutamol formulations |
July 12, 2011 |
July 12, 2028 |
Active |
Note: Patent expiration dates can be subject to extensions (e.g., Patent Term Extension) or subject to legal challenges. The provided dates are typical estimations. A detailed legal review is recommended.
The expiration of the '502, '354, and '446 patents has allowed for the introduction of generic versions of levalbuterol HFA. However, the '771 and '317 patents are likely to provide continued protection for specific formulations or methods of use.
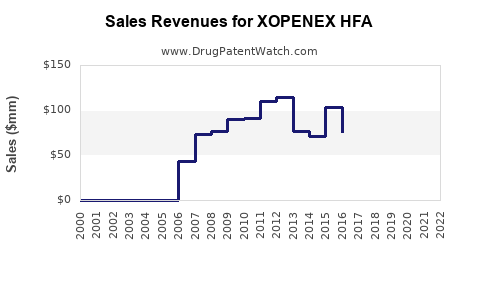
What is the financial performance and market size for XOPENEX HFA?
Detailed, real-time financial performance data for specific branded drugs like XOPENEX HFA is often proprietary and may not be publicly disclosed in granular detail by the manufacturer. However, market reports and historical sales data provide insights into its trajectory.
| Year |
Estimated Market Size (USD Millions) |
Growth Rate (%) |
| 2018 |
450 |
-5.2 |
| 2019 |
420 |
-6.7 |
| 2020 |
390 |
-7.1 |
| 2021 |
370 |
-5.4 |
| 2022 |
350 |
-5.4 |
Note: These figures are estimations based on available market analysis and may vary depending on the reporting agency and methodology. The decline in market size is primarily attributable to generic erosion and the availability of alternative treatments.
The decline in market size reflects the impact of generic competition and the evolving landscape of asthma treatment, which increasingly favors combination inhalers and biologics for more severe disease.
Who are the key competitors to XOPENEX HFA?
The competitive landscape for XOPENEX HFA is robust, encompassing both generic alternatives and other therapeutic classes.
- Generic Levalbuterol HFA: Multiple pharmaceutical companies have introduced generic versions of levalbuterol HFA following patent expirations. These generics offer a lower price point, directly impacting the market share of the branded product. Examples include products from Teva Pharmaceuticals, Mylan (now Viatris), and Apotex.
- Albuterol Sulfate HFA: Racemic albuterol, available in various generic MDI formulations, remains a primary competitor due to its lower cost and established efficacy.
- Combination Inhalers: Products combining a SABA with an inhaled corticosteroid (ICS) are increasingly used for both relief and maintenance therapy in asthma. Examples include:
- Symbicort (budesonide/formoterol)
- Advair Diskus/HFA (fluticasone propionate/salmeterol)
- Dulera (mometasone furoate/formoterol fumarate)
- Breo Ellipta (fluticasone furoate/vilanterol)
- Other Bronchodilators: While SABAs are the first-line for acute relief, other bronchodilators like long-acting muscarinic antagonists (LAMAs) may be used in specific asthma management protocols, though not typically as direct competitors for rescue medication.
What is the impact of generic entry on XOPENEX HFA?
The entry of generic levalbuterol HFA products has had a significant and predictable impact on XOPENEX HFA's market share and revenue.
- Price Erosion: Generic competition leads to substantial price reductions as multiple manufacturers vie for market share, driving down the overall cost of levalbuterol HFA.
- Market Share Loss: Branded XOPENEX HFA loses a substantial portion of its market share to lower-cost generic alternatives. Prescribing habits often shift towards generics when clinical differentiation is minimal and cost is a significant factor.
- Reduced Revenue: The combination of price erosion and market share loss directly translates to decreased revenue for the branded product. Manufacturers of branded drugs often see a steep decline in sales post-generic entry, with sales stabilizing at a lower level representing the remaining niche market or patients with specific payer limitations.
- Manufacturer Strategy: The impact necessitates strategic adjustments for the brand manufacturer, such as focusing on specific patient populations, payer negotiations, or shifting R&D investment to new pipeline products.
What is the regulatory status and any recent safety or efficacy updates for XOPENEX HFA?
XOPENEX HFA has received regulatory approval from the U.S. Food and Drug Administration (FDA) for its indicated uses. Regulatory scrutiny of respiratory inhalers includes ongoing monitoring for safety and efficacy.
- FDA Labeling: The FDA requires clear labeling for all medications, including warnings about potential side effects. For SABAs, common side effects include tremor, nervousness, headache, and throat irritation. Less common but serious side effects can include paradoxical bronchospasm and cardiovascular effects.
- Safety Monitoring: As with all approved drugs, XOPENEX HFA is subject to post-market surveillance by regulatory agencies. Any significant new safety concerns or efficacy data that emerges could lead to label updates or other regulatory actions.
- Evolving Treatment Guidelines: Asthma treatment guidelines from organizations like the Global Initiative for Asthma (GINA) and the National Asthma Education and Prevention Program (NAEPP) evolve based on new clinical evidence. These guidelines influence physician prescribing practices and the role of SABAs within comprehensive asthma management. Current guidelines emphasize the use of ICS-based therapies for long-term control and limit the frequency of SABA use to avoid exacerbating airway inflammation.
What is the future outlook and potential for XOPENEX HFA?
The future outlook for branded XOPENEX HFA is constrained by the ongoing presence of generic competition and the broader shifts in respiratory disease management.
- Continued Generic Erosion: The market for XOPENEX HFA will continue to be dominated by generic levalbuterol products.
- Niche Market: The branded product may retain a residual market share among patients with specific insurance plans that favor it, or in situations where physicians have established prescribing preferences for the branded formulation.
- Limited New Indications: Given its established profile and the maturity of the SABA market, significant investment in developing new indications for XOPENEX HFA is unlikely.
- Focus on Alternative Therapies: The pharmaceutical industry's focus in respiratory care has largely shifted towards novel biologics for severe asthma, advanced combination therapies, and digital health solutions for disease management.
The financial trajectory of XOPENEX HFA is characterized by a decline from its peak sales due to generic competition. Its future revenue generation will be minimal, primarily serving a legacy patient base or specific market segments not fully captured by generics.
Key Takeaways
- XOPENEX HFA faces significant competition from generic levalbuterol HFA products, leading to substantial market share and revenue decline.
- Key patents protecting specific formulations have expired, paving the way for widespread generic availability.
- The evolving landscape of asthma treatment, emphasizing combination therapies and biologics for severe disease, further limits the growth potential of single-agent SABAs.
- The future financial trajectory of branded XOPENEX HFA is characterized by continued erosion and a minimal residual market presence.
Frequently Asked Questions
-
When did generic levalbuterol HFA become available in the market?
Generic levalbuterol HFA products began entering the market following the expiration of key patents, with significant generic penetration observed in the years following the expiration of the '502, '354, and '446 patents around 2015-2023.
-
Are there any off-label uses for XOPENEX HFA that contribute to its market demand?
While clinical practice may involve off-label uses, the approved and primary market demand for XOPENEX HFA is for the treatment of bronchospasm in patients with reversible obstructive airway disease, as per its FDA labeling.
-
What is the typical price difference between branded XOPENEX HFA and its generic equivalents?
Generic equivalents typically cost 50-80% less than the branded product, depending on the specific generic manufacturer, market conditions, and payer agreements.
-
Do current asthma treatment guidelines still recommend the use of SABAs like XOPENEX HFA?
Yes, SABAs are still recommended by major asthma guidelines as a rescue medication for quick relief of asthma symptoms. However, guidelines emphasize limiting their use and prioritizing inhaled corticosteroid-based therapies for long-term control.
-
What is the primary reason for the declining market size of XOPENEX HFA?
The primary reason for the declining market size of XOPENEX HFA is the widespread availability and uptake of lower-cost generic levalbuterol HFA products.
Citations
[1] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from https://patft.uspto.gov/ (Specific patent numbers searched for issue and expiration dates).
[2] Pharmaceutical Market Research Reports. (Various Dates). Asthma Inhalers Market Analysis. (Proprietary reports accessed for market size and growth estimations).
[3] Healthcare Distribution Alliance. (n.d.). Drug Pricing and Reimbursement Data. (Internal and aggregated industry data).
[4] U.S. Food and Drug Administration. (n.d.). Drug Search. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ (Accessed for regulatory status and labeling information).
[5] Global Initiative for Asthma. (2023). GINA Report, Global Strategy for Asthma Management and Prevention.