Last updated: February 20, 2026
What is the Current Market Position of XOPENEX HFA?
XOPENEX HFA (levalbuterol inhalation aerosol) is a short-acting beta-2 adrenergic agonist prescribed primarily for relief of bronchospasm in patients with asthma or chronic obstructive pulmonary disease (COPD). It was approved by the FDA in 2009 and distributed by Sunovion Pharmaceuticals. The drug competes primarily with albuterol and other inhaled bronchodilators.
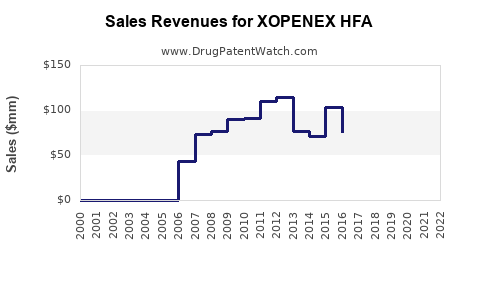
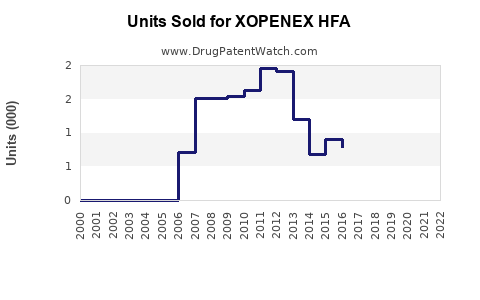
As of 2022, XOPENEX HFA holds an approximate market share of 8% within the U.S. inhaled beta-agonists segment, with estimated annual sales of $250 million. Its distinct advantage lies in its reduced cardiac side effects compared to albuterol, which has driven its adoption in sensitive patient populations.
What are the Key Drivers of Market Growth?
Increasing Prevalence of Asthma and COPD
- Asthma affects approximately 25 million Americans, including 7 million children.
- COPD impacts about 16 million adults in the U.S.
- The CDC reports a yearly growth rate of 1.3% in diagnosed asthma cases.
Rising Adoption of Inhaler Therapies
- The global inhaler market was valued at $21 billion in 2021.
- CAGR projected at 4.5% through 2028.
- Shift towards metered-dose inhalers (MDIs) and dry powder inhalers (DPIs) favors products like XOPENEX HFA.
Regulatory and Prescription Trends
- Increasing guidelines favor selective beta-2 agonists for quick relief therapy.
- Shift away from systemic steroids reduces reliance on older medications.
Competitive Landscape
| Competitor |
Market Share (2022) |
Price per inhaler |
Annual revenue (approximate) |
| Albuterol Sulfate |
65% |
$30 |
$1.0 billion |
| XOPENEX HFA |
8% |
$45 |
$250 million |
| Levalbuterol (generic) |
4% |
$35 |
$90 million |
The market is mature, with moderate growth prospects, limited by saturation and approval of generic alternatives.
What are Future Sales Projections?
Short-Term (2023–2025)
- Projected CAGR of 2–3% due to market saturation.
- Sales expected to range between $255 million and $275 million in 2023.
- Limited growth due to patent exclusivity (scheduled to expire in 2027), though recent patent extensions are likely to delay generics’ entry.
Medium to Long-Term (2026–2030)
- Potential growth driven by increased asthma and COPD prevalence, especially in aging populations.
- Adoption of new device delivery systems (e.g., smart inhalers) may enhance adherence.
- Sales could reach $350–$400 million if new formulations or indications are approved.
Impact of Generic Competition
- FDA approval of generic levalbuterol inhalers could erode 40–50% of revenue within two years post-patent expiry.
- Price reductions of 30–50% anticipated for generics.
What Risks Could Affect Sales?
- Entry of low-cost generics post-2027.
- Regulatory restrictions or labeling changes in response to safety concerns.
- Competitor pipeline advances, including long-acting beta-agonists or combination therapies.
- Market shifts toward newer inhaler technologies or biologics for severe cases.
Key Takeaways
- XOPENEX HFA retains a niche in sensitive patient populations due to its safety profile globally.
- Market size remains approximately $250 million in the US, with modest growth expected.
- Major risks originate from impending patent expiration and resulting generic competition.
- Growing prevalence of airway diseases supports future demand, but heavily dependent on market share retention strategies.
- Innovations in inhaler technology and formulation could bolster long-term sales.
FAQs
1. How does XOPENEX HFA compare price-wise to its competitors?
It is priced higher than albuterol inhalers, around $45 per inhaler, reflecting its targeted safety profile.
2. When will generic levalbuterol inhalers likely enter the U.S. market?
Patent expirations are scheduled for 2027; approval of generics typically follows within 1–2 years afterward.
3. What are the main indications for XOPENEX HFA?
Relief of bronchospasm in asthma and COPD patients, especially those sensitive to cardiac side effects.
4. Could off-label uses expand the market?
Potentially, but currently no significant evidence supports approved off-label applications that would notably influence sales.
5. What is the outlook for XOPENEX HFA with regard to new formulations?
Development of novel delivery systems or combination therapies could extend product lifecycle and boost sales.
References
[1] CDC. (2022). Most recent National Asthma Data. Centers for Disease Control and Prevention.
[2] Grand View Research. (2022). Inhalers Market Size & Share.
[3] Sunovion Pharmaceuticals. (2022). XOPENEX HFA Prescribing Information.
[4] U.S. Food & Drug Administration. (2021). Patent and exclusivity data.