VIZAMYL Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Vizamyl, and what generic alternatives are available?
Vizamyl is a drug marketed by Ge Healthcare and is included in one NDA. There is one patent protecting this drug.
This drug has twenty-three patent family members in twenty countries.
The generic ingredient in VIZAMYL is flutemetamol f-18. One supplier is listed for this compound. Additional details are available on the flutemetamol f-18 profile page.
DrugPatentWatch® Generic Entry Outlook for Vizamyl
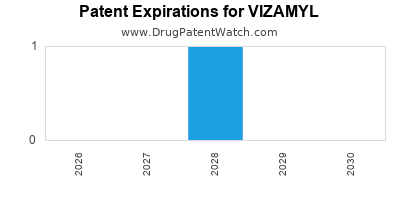
Vizamyl was eligible for patent challenges on October 25, 2017.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be September 16, 2028. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for VIZAMYL?
- What are the global sales for VIZAMYL?
- What is Average Wholesale Price for VIZAMYL?
Summary for VIZAMYL
| International Patents: | 23 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 5 |
| Clinical Trials: | 6 |
| Patent Applications: | 141 |
| Drug Prices: | Drug price information for VIZAMYL |
| What excipients (inactive ingredients) are in VIZAMYL? | VIZAMYL excipients list |
| DailyMed Link: | VIZAMYL at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for VIZAMYL
Generic Entry Date for VIZAMYL*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
INJECTABLE;INTRAVENOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for VIZAMYL
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Yale University | Phase 1 |
| University of Utah | Phase 2 |
| University Hospital, Toulouse | Early Phase 1 |
Pharmacology for VIZAMYL
| Drug Class | Radioactive Diagnostic Agent |
| Mechanism of Action | Radiopharmaceutical Activity |
US Patents and Regulatory Information for VIZAMYL
VIZAMYL is protected by one US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of VIZAMYL is ⤷ Start Trial.
This potential generic entry date is based on patent 8,916,131.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ge Healthcare | VIZAMYL | flutemetamol f-18 | INJECTABLE;INTRAVENOUS | 203137-001 | Oct 25, 2013 | DISCN | Yes | No | 8,916,131 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Ge Healthcare | VIZAMYL | flutemetamol f-18 | INJECTABLE;INTRAVENOUS | 203137-002 | Oct 25, 2013 | RX | Yes | Yes | 8,916,131 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for VIZAMYL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Ge Healthcare | VIZAMYL | flutemetamol f-18 | INJECTABLE;INTRAVENOUS | 203137-002 | Oct 25, 2013 | 8,236,282 | ⤷ Start Trial |
| Ge Healthcare | VIZAMYL | flutemetamol f-18 | INJECTABLE;INTRAVENOUS | 203137-001 | Oct 25, 2013 | 8,236,282 | ⤷ Start Trial |
| Ge Healthcare | VIZAMYL | flutemetamol f-18 | INJECTABLE;INTRAVENOUS | 203137-001 | Oct 25, 2013 | 7,270,800 | ⤷ Start Trial |
| Ge Healthcare | VIZAMYL | flutemetamol f-18 | INJECTABLE;INTRAVENOUS | 203137-002 | Oct 25, 2013 | 7,351,401 | ⤷ Start Trial |
| Ge Healthcare | VIZAMYL | flutemetamol f-18 | INJECTABLE;INTRAVENOUS | 203137-001 | Oct 25, 2013 | 8,691,185 | ⤷ Start Trial |
| Ge Healthcare | VIZAMYL | flutemetamol f-18 | INJECTABLE;INTRAVENOUS | 203137-002 | Oct 25, 2013 | 7,270,800 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for VIZAMYL
When does loss-of-exclusivity occur for VIZAMYL?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 08292201
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0815129
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 94084
Estimated Expiration: ⤷ Start Trial
China
Patent: 1790387
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 82988
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 82988
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 46710
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 500007
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 3316
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 67708
Estimated Expiration: ⤷ Start Trial
Patent: 10536931
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 10002196
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 3616
Patent: RADIOPHARMACEUTICAL COMPOSITION
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 15006
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 82988
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 82988
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 75267
Patent: РАДИОФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ (RADIOPHARMACEUTICAL COMPOSITION)
Estimated Expiration: ⤷ Start Trial
Patent: 10101935
Patent: РАДИОФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1000718
Patent: Radiopharmaceutical composition
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1571572
Estimated Expiration: ⤷ Start Trial
Patent: 100055440
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 64715
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering VIZAMYL around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Poland | 1611115 | ⤷ Start Trial | |
| Brazil | 0113470 | Composto de ligação amilóide, composição farmacêutica e métodos de sìntese de composto, de detecção in vivo de depósitos amilóides num indivìduo e em tecido humano ou animal, de quantificação de depósito amilóide em tecido de biópsia ou post-mortem e de distinção de um cérebro de doença de alzheimer de um cérebro normal | ⤷ Start Trial |
| China | 1285582 | ⤷ Start Trial | |
| New Zealand | 583616 | RADIOPHARMACEUTICAL COMPOSITION | ⤷ Start Trial |
| China | 1535268 | 用于早老性痴呆的死前诊断和淀粉样蛋白沉积的体内成象和预防的硫黄素衍生物 (Thioflavin derivatives and their use in diagnosis and theraphy of alzheimer's disease) | ⤷ Start Trial |
| Poland | 215711 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VIZAMYL
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2182988 | 1590009-5 | Sweden | ⤷ Start Trial | PRODUCT NAME: FLUTEMETAMOL (18F); REG. NO/DATE: EU/1/14/941 20140826 |
| 1611115 | C 2015 004 | Romania | ⤷ Start Trial | PRODUCT NAME: FLUTEMETAMOL (18 F) SAU O SARE, HIDRAT SAU SOLVAT ALACESTUIA ACCEPTABILEFARMACEUTIC; NATIONAL AUTHORISATION NUMBER: EU/1/14/941; DATE OF NATIONAL AUTHORISATION: 20140822; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/14/941; DATE OF FIRST AUTHORISATION IN EEA: 20140822 |
| 1611115 | 574 | Finland | ⤷ Start Trial | |
| 2182988 | SPC/GB15/012 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: FLUTEMETAMOL (18F) OR A PHARMACEUTICALLY ACCEPTABLE SALT, HYDRATE OR SOLVATE THEREOF; REGISTERED: UK EU/1/14/941 20140826 |
| 2182988 | 573 | Finland | ⤷ Start Trial | |
| 1611115 | CR 2015 00009 | Denmark | ⤷ Start Trial | PRODUCT NAME: FLUTEMETAMOL (18F); REG. NO/DATE: EU/1/14/941 20140826 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for VIZAMYL: A Comprehensive Analysis
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.