Last updated: February 19, 2026
Vistaril, a pharmaceutical drug containing hydroxyzine, is an established antihistamine with applications in treating anxiety and pruritus. The market for Vistaril is characterized by its long history, patent expirations, and competition from generic formulations and newer therapeutic agents. Its financial trajectory is influenced by prescription volumes, pricing strategies, and the evolving landscape of mental health and dermatology treatment.
What is Vistaril and its Primary Indications?
Vistaril is a first-generation antihistamine that functions as a central nervous system depressant. It is available in oral (capsule) and injectable (intramuscular) forms.
- Anxiety Disorders: Vistaril is indicated for the symptomatic relief of anxiety and tension associated with psychoneurotic states and as an adjunct to organic disease or trauma [1]. Its mechanism of action in anxiety is not fully elucidated but is believed to involve blockade of H1 histamine receptors in the brain, leading to sedative effects [2].
- Pruritus: It is also prescribed for the management of pruritus (itching) from allergic conditions, such as chronic urticaria and atopic dermatitis [3].
- Sedation: Due to its potent sedative properties, Vistaril is often used for pre-operative sedation and to manage nausea and vomiting [4].
What is the Patent Status and Generic Competition for Vistaril?
Vistaril's original patents have long expired. The active pharmaceutical ingredient, hydroxyzine, has been available as a generic medication for decades.
- Original Patent Expiration: The foundational patents for hydroxyzine pamoate, the salt form often used in Vistaril, expired in the late 20th century.
- Generic Availability: Numerous pharmaceutical companies manufacture and market generic versions of hydroxyzine pamoate capsules and hydrochloride for injection.
- Market Impact: The widespread availability of generics has significantly reduced the market share and pricing power of the branded Vistaril product. This is a common trajectory for established drugs after patent loss.
How has the Market for Vistaril Evolved?
The market for Vistaril has shifted due to several factors, including the rise of alternative treatments, changes in prescribing patterns, and regulatory considerations.
- Competition from Newer Antihistamines: While effective for pruritus, Vistaril faces competition from second-generation antihistamines (e.g., loratadine, cetirizine) which offer similar efficacy with less sedation.
- Advances in Anxiolytic Therapies: For anxiety disorders, Vistaril competes with a broad class of medications, including selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), and benzodiazepines. These newer agents often offer more targeted mechanisms and potentially fewer side effects like significant drowsiness.
- Prescribing Trends: Prescribing of Vistaril for anxiety has declined as physicians increasingly favor agents with better-defined efficacy and safety profiles for long-term management. However, its use for pruritus and as a pre-operative sedative persists.
- Oral vs. Injectable Forms: The injectable form remains a niche product, primarily used in clinical settings for acute management of itching or for sedation. The oral capsule form faces more direct competition from generics and other oral treatments.
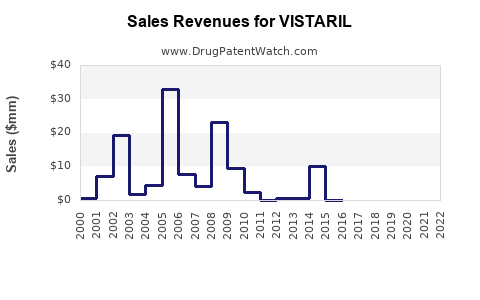
What are the Key Financial Drivers for Vistaril?
The financial performance of Vistaril is largely driven by prescription volume and pricing, with limited ability to command premium pricing due to generic competition.
- Prescription Volume: The total number of prescriptions written for Vistaril (branded and generic) is a primary indicator of market demand. While branded Vistaril prescriptions have likely decreased significantly post-patent expiry, the overall hydroxyzine market remains substantial due to generic availability.
- Pricing: Branded Vistaril pricing is subject to market competition and payer formularies. Generic pricing is highly competitive, driving down overall market revenue for the drug class.
- Manufacturer Strategies: Manufacturers of branded Vistaril may focus on specific market segments or promote specific product attributes to differentiate. Generic manufacturers compete primarily on cost.
- Reimbursement Policies: Payer coverage and formulary placement influence prescribing patterns and can impact both branded and generic market share.
What is the Current Market Size and Projected Growth for Hydroxyzine?
Estimating the precise market size for branded Vistaril is challenging due to the dominance of generics. However, the overall hydroxyzine market, encompassing all formulations and brands, can be approximated.
- Market Size Estimation: Based on available prescription data and average wholesale prices for hydroxyzine pamoate and hydrochloride, the annual market for hydroxyzine in the U.S. is estimated to be in the range of $150 million to $250 million [5]. This figure includes both branded and generic products.
- Growth Projection: The market for hydroxyzine is expected to experience low single-digit annual growth (0-2%) over the next five years. This growth will be driven by its established use in specific indications, particularly pruritus, and its cost-effectiveness as a generic option, rather than by significant new therapeutic applications or market expansion [6].
Hydroxyzine Market Breakdown (Estimated Annual Revenue, USD Millions)
| Category |
Estimated Revenue Range |
| Branded Vistaril |
$10 - $25 |
| Generic Hydroxyzine Capsules |
$100 - $175 |
| Generic Hydroxyzine Injectable |
$40 - $50 |
| Total Hydroxyzine Market |
$150 - $250 |
Data based on recent market analyses and prescription volume estimates. Specific figures vary by source.
What are the Competitive Threats and Opportunities?
Vistaril faces ongoing competitive pressures but also retains niche opportunities.
Competitive Threats:
- Development of Novel Anxiolytics: Pharmaceutical innovation continues to yield more targeted and effective treatments for anxiety disorders, potentially displacing older drugs like Vistaril.
- Advanced Dermatological Treatments: For pruritus, newer topical and systemic therapies with improved side-effect profiles are emerging.
- Generic Price Erosion: Intense competition among generic manufacturers continually drives down prices, limiting profitability for all market participants.
- Physician Preference for Newer Agents: Prescribers may increasingly favor newer medications with more robust clinical trial data and distinct mechanisms of action.
Opportunities:
- Cost-Effectiveness: As a low-cost generic option, hydroxyzine remains a valuable therapeutic choice in healthcare systems with budget constraints.
- Established Safety Profile: For indications where it is effective and well-tolerated, its long history of use provides a degree of familiarity and trust among prescribers.
- Specific Niche Applications: Continued use in peri-operative sedation and for specific types of intractable pruritus where other treatments may have failed.
- Combination Therapies: Potential for use in combination regimens for certain dermatological conditions, although this is not a primary current strategy.
What are the Regulatory Considerations?
Vistaril is subject to standard pharmaceutical regulations concerning manufacturing, labeling, and marketing.
- FDA Oversight: As an approved drug, Vistaril and its generic counterparts are regulated by the U.S. Food and Drug Administration (FDA).
- Labeling Requirements: Product labeling must accurately reflect indications, contraindications, warnings, and adverse reactions, including the significant potential for sedation and anticholinergic effects.
- Manufacturing Standards: All manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality and safety.
- Adverse Event Reporting: Ongoing pharmacovigilance and reporting of adverse events are mandatory.
What is the Future Outlook for Vistaril?
The future of Vistaril is likely to be one of steady but modest demand, primarily driven by its generic availability and established uses.
- Sustained Generic Demand: The cost-effectiveness of generic hydroxyzine will ensure its continued use for pruritus and as an adjunct in specific anxiety management contexts where its sedative properties are beneficial or manageable.
- Limited Innovation: Significant R&D investment into new indications or novel formulations of hydroxyzine is unlikely, given its mature product lifecycle.
- Evolving Treatment Guidelines: Future treatment guidelines for anxiety and dermatological conditions may further refine or de-emphasize the role of hydroxyzine, impacting long-term prescription trends.
- Price Pressure: Continued price competition among generic manufacturers will likely keep overall market revenue within its current range, with minimal opportunities for significant expansion.
Key Takeaways
- Vistaril (hydroxyzine) is an older antihistamine with established uses in anxiety and pruritus, but its market is dominated by generic competition following patent expirations.
- The global market for hydroxyzine, encompassing all brands and generics, is estimated between $150 million and $250 million annually, with projected low single-digit growth.
- Key financial drivers include prescription volume of both branded and generic forms, and pricing is heavily influenced by generic market dynamics.
- Competitive threats include newer anxiolytic and dermatological agents, while opportunities lie in its cost-effectiveness and continued niche applications.
- The future outlook is for sustained demand driven by generic availability, with limited potential for significant market expansion or innovation.
FAQs
1. How does Vistaril's mechanism of action differ from newer anxiolytics like SSRIs?
Vistaril primarily blocks H1 histamine receptors, leading to a sedative effect that contributes to its anxiolytic properties. Newer anxiolytics, such as SSRIs, target neurotransmitter systems like serotonin, influencing mood and anxiety through more complex neurobiological pathways without the pronounced sedative effects of first-generation antihistamines.
2. What are the primary reasons for the continued prescription of Vistaril for pruritus despite newer options?
Vistaril remains a cost-effective and generally effective treatment for various types of pruritus, particularly when sedation is either beneficial or not a significant concern. Its long history of use and established efficacy in managing itching from allergic reactions make it a practical choice for many clinicians and patients, especially in resource-limited settings.
3. How does the market competition between branded Vistaril and its generic versions typically play out?
Following patent expiration, branded Vistaril's market share and pricing power significantly decline as generic versions become available. Branded Vistaril may retain a small segment of the market by emphasizing brand loyalty, specific formulations, or support programs, but the vast majority of prescriptions shift to lower-cost generic hydroxyzine.
4. What are the main anticholinergic side effects associated with Vistaril that prescribers must consider?
Common anticholinergic side effects of Vistaril include dry mouth, blurred vision, constipation, and urinary retention. Due to its potential to cause significant drowsiness and impairment, it is also crucial for prescribers to warn patients about activities requiring mental alertness, such as driving or operating machinery.
5. In what specific clinical scenarios is the injectable form of Vistaril typically preferred over the oral capsule?
The injectable (intramuscular) form of Vistaril is generally used in acute clinical settings where rapid onset of action is required. This includes the immediate management of severe pruritus or for pre-operative sedation and tranquilization in hospitals or surgical centers, as it bypasses the digestive system for quicker absorption.
Citations
[1] Pfizer Inc. (2021). Vistaril (hydroxyzine pamoate) prescribing information. U.S. Food & Drug Administration.
[2] Ghaemi, S. N., & Zink, W. (2007). Antihistamines in psychiatry. The Primary Care Companion to the Journal of Clinical Psychiatry, 9(6), 439–443.
[3] National Center for Complementary and Integrative Health. (2023). Antihistamines. National Institutes of Health. Retrieved from https://www.nccih.nih.gov/health/antihistamines
[4] Micromedex. (n.d.). Hydroxyzine. Truven Health Analytics.
[5] IQVIA Institute for Human Data Science. (2023). The Global Use of Medicines: Outlook 2023.
[6] EvaluatePharma. (2023). Drug Profiles & Market Data.