Last updated: February 19, 2026
What is Varithena's approved indication and mechanism of action?
Varithena (polidocanol injectable foam) is indicated for the treatment of incompetent great saphenous veins (GSVs), reticular veins, and tributaries in the saphenous distribution of the great saphenous vein (GSV) system. It is used for patients with symptomatic varicose veins who are typically candidates for surgical or other ablative treatments. The foam is administered via ultrasound-guided injection directly into the diseased veins.
The mechanism of action of polidocanol injectable foam is sclerotherapy. Upon injection, polidocanol acts as a sclerosant, causing damage to the endothelial lining of the vein. This initiates an inflammatory response, leading to the formation of thrombus (blood clot) and fibrosis (scar tissue). The vein walls adhere together, and the vein is eventually obliterated and converted into fibrous cord that is absorbed by the body over time. This process effectively closes off the targeted varicose veins, redirecting blood flow to healthy veins.
What is Varithena's regulatory status and market exclusivity?
Varithena received U.S. Food and Drug Administration (FDA) approval on November 13, 2013, for the treatment of GSVs and their tributaries in the saphenous distribution. The active ingredient, polidocanol, is a well-established sclerosant with a history of use in various medical applications. The specific formulation as an injectable foam for varicose veins represented a novel delivery method at the time of approval.
The patent landscape for Varithena, particularly the specific injectable foam formulation and its method of use for varicose veins, is crucial for its market exclusivity. While the core chemical entity polidocanol has been known for decades, patents likely cover aspects such as:
- The injectable foam composition: This includes the specific ratios of polidocanol, gas (e.g., air or carbon dioxide), and any excipients used to create a stable and effective foam.
- Manufacturing processes: Novel or improved methods for generating the polidocanol foam.
- Methods of treatment: Specific protocols for administering the foam, including dosage, injection techniques, and ultrasound guidance.
- Device-related patents: If specific delivery devices or syringes are integral to the foam preparation or administration.
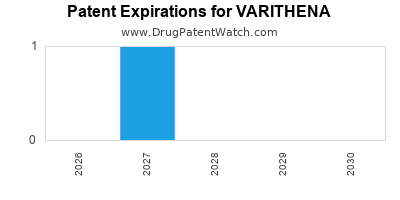
A primary patent for the Varithena foam formulation (e.g., U.S. Patent No. 8,343,954) was granted in 2013 and is expected to expire in 2030. This patent provides a significant period of market exclusivity for the branded product. There may be other related patents covering different aspects of the product or its use, potentially extending exclusivity beyond 2030.
Who are the key manufacturers and competitors in the varicose vein treatment market?
The manufacturer of Varithena is Merit Medical Systems, Inc., which acquired the commercial rights. Merit Medical Systems is a global leader in the development, manufacturing, and marketing of proprietary medical devices used in the cardiovascular, radiophysical, and surgical markets.
The market for varicose vein treatments is competitive and includes a range of therapeutic modalities. Key competitors and their offerings include:
-
Other Sclerotherapy Agents:
- Asclera (polidocanol solution): Manufactured by Chemische Fabrik Kreussler & Co. GmbH, approved for smaller varicose veins and reticular veins. While chemically similar, the formulation (solution vs. foam) and approved indications differ. Asclera is a direct competitor in the broader sclerotherapy segment.
- Sodium Tetradecyl Sulfate (STS) solutions: Available under various brand names (e.g., Sotradecol). This is a long-established sclerosant used in liquid form.
-
Endovenous Thermal Ablation Devices:
- Venclose, Inc. (Venclose system): Radiofrequency ablation technology.
- Medtronic (VenaSeal Closure System): Uses medical-grade adhesive to close veins. This is a non-tumescent, non-thermal, non-foaming approach.
- Boston Scientific (VENOUS CLOSURE™ System): Utilizes a transcutaneous closure system.
- BD (formerly Bard) (EVLT - Endovenous Laser Ablation): Uses laser energy to close veins.
-
Surgical Ligation and Stripping: This remains a traditional treatment method, though often superseded by less invasive procedures.
The competitive landscape is characterized by the ongoing shift towards minimally invasive procedures, with a focus on patient comfort, recovery time, and efficacy. Varithena's foam formulation competes directly with other sclerotherapy agents and indirectly with thermal ablation and adhesive closure systems.
What is Varithena's historical sales performance and market penetration?
Varithena's sales performance has shown consistent growth since its introduction. The product's trajectory is influenced by market adoption rates of minimally invasive venous procedures and Merit Medical's commercialization efforts.
- 2014-2016: Initial launch and market entry, focusing on building awareness and physician adoption. Sales were in the lower millions of dollars annually.
- 2017-2019: Sustained growth driven by increased physician familiarity, expanded sales force reach, and positive clinical data. Annual sales began to exceed $30 million.
- 2020-2022: The COVID-19 pandemic presented challenges to elective procedures, impacting sales temporarily. However, the trend towards minimally invasive treatments resumed, and Varithena sales continued to climb, demonstrating resilience. In 2022, net sales for Varithena reached approximately $65 million.
- 2023: Merit Medical reported $74.9 million in Varithena net sales for 2023, representing a 15.4% increase over 2022. This indicates strong continued market acceptance and penetration.
Market penetration is estimated to be significant within the physician-administered sclerotherapy segment for GSV treatment. While precise market share figures are proprietary, Varithena is recognized as a leading product in its category, particularly for foam sclerotherapy. The company's focus on clinical education and support for healthcare providers has been instrumental in driving this penetration.
What are the projected financial trajectories and market forecasts for Varithena?
Merit Medical Systems has provided guidance and expressed optimism for Varithena's future financial trajectory. The company anticipates continued growth driven by several factors:
- Sustained Market Growth: The global market for varicose vein treatment is projected to expand, fueled by an aging population, increasing prevalence of venous insufficiency, and a growing preference for minimally invasive procedures over traditional surgery.
- Geographic Expansion: Merit Medical has been focused on expanding Varithena's presence in international markets. Commercialization efforts are underway or planned in Europe, Asia, and other regions, which will contribute to revenue growth.
- Clinical Evidence and Data: Ongoing clinical studies and real-world evidence demonstrating Varithena's safety and efficacy can further solidify its position and drive adoption among physicians and patients.
- Sales Force Effectiveness: Investments in expanding and training the sales force are critical for increasing market reach and physician engagement.
- Patent Exclusivity: The U.S. patent for the Varithena formulation is expected to expire in 2030, providing a significant period of market exclusivity for the branded product. This allows for sustained premium pricing and profitability.
Financial Projections:
While specific long-term revenue forecasts are typically internal to Merit Medical, based on recent performance and market trends, analysts and the company project continued double-digit growth.
- 2024: Projections suggest sales potentially reaching $85 million to $95 million, representing growth of 13% to 27% over 2023.
- Beyond 2024: Assuming continued market expansion, successful international launches, and no significant disruptive competition, annual sales could potentially reach or exceed $150 million by 2028. This projection factors in the historical growth rate and the anticipated benefits of expanding into new territories.
The financial trajectory will be closely monitored for the impact of generic competition post-patent expiry in 2030. However, the lead time for developing and obtaining regulatory approval for generic foam formulations, along with the established brand loyalty and physician comfort with Varithena, may provide a buffer against immediate and significant market share erosion.
What are the primary risks and opportunities impacting Varithena's market position?
Risks:
- Generic Competition Post-Patent Expiry: The expiration of key patents in 2030 presents the most significant long-term risk. The entry of lower-cost generic polidocanol foam products could erode Varithena's market share and pricing power.
- Competitive Advancements: Development of novel or significantly improved minimally invasive treatments for varicose veins by competitors could offer superior efficacy, safety, or cost-effectiveness, drawing market share away from Varithena. Examples include next-generation adhesive systems, improved thermal ablation technologies, or entirely new therapeutic modalities.
- Reimbursement Policies: Changes in healthcare reimbursement policies from government payers (e.g., Medicare, Medicaid) and private insurers could impact physician and facility adoption by altering the financial incentives for using Varithena compared to alternative treatments.
- Adverse Event Profile: While generally well-tolerated, any significant increase in reported adverse events or new safety concerns associated with Varithena could negatively impact physician confidence and patient demand.
- Economic Downturns: Varithena is an elective procedure, and economic downturns can lead to reduced patient spending on non-essential medical treatments.
Opportunities:
- Expanding Geographic Markets: Successful launches and market penetration in untapped or underserved international regions represent a significant growth opportunity. Current efforts in Europe and potential expansion into Asia Pacific and other markets are key.
- Clinical Data and Education: Continued generation of robust clinical data demonstrating Varithena's long-term efficacy, safety, and patient-reported outcomes can reinforce its value proposition. Comprehensive physician education and training programs can further drive adoption.
- Label Expansion or New Indications: While Varithena is focused on GSV system treatment, exploring potential label expansions or new indications for polidocanol foam in related venous conditions could create new revenue streams.
- Integration into Value-Based Care Models: As healthcare systems move towards value-based care, Varithena's minimally invasive nature and potentially shorter recovery times could align well with models that prioritize patient outcomes and cost-effectiveness.
- Strategic Partnerships and Acquisitions: Merit Medical could pursue strategic partnerships or further acquisitions to enhance its venous treatment portfolio or expand its commercial reach.
Key Takeaways
Varithena (polidocanol injectable foam) holds a significant position in the minimally invasive varicose vein treatment market, particularly for GSV indications. Manufactured by Merit Medical Systems, Inc., the product benefits from a strong regulatory approval history, with key U.S. patent exclusivity extending until 2030. Historically, Varithena has demonstrated consistent sales growth, reaching $74.9 million in 2023, with projections indicating continued double-digit increases. Key growth drivers include expanding international markets, ongoing physician education, and the general shift towards less invasive medical procedures. However, the market faces risks, most notably potential generic competition post-patent expiry and the continuous threat of disruptive technological advancements from competitors. Merit Medical's strategic focus on geographic expansion and clinical evidence generation positions Varithena for sustained financial performance in the medium term.
FAQs
-
What is the primary difference between Varithena and Asclera?
Varithena is a polidocanol injectable foam indicated for incompetent great saphenous veins (GSVs) and their tributaries, administered as ultrasound-guided foam. Asclera is a polidocanol solution primarily indicated for smaller varicose veins and reticular veins and is typically administered without ultrasound guidance for those indications.
-
When does the main U.S. patent for Varithena expire?
The primary U.S. patent covering the Varithena foam formulation is expected to expire in 2030.
-
What are the main revenue drivers for Varithena?
Revenue drivers include sales in the U.S. market, increasing international market penetration, and adoption by physicians performing minimally invasive venous procedures.
-
Can Varithena be used for all types of varicose veins?
Varithena is indicated for incompetent great saphenous veins (GSVs), reticular veins, and tributaries in the saphenous distribution of the GSV system. It is not necessarily indicated for all forms or locations of superficial venous disease.
-
What is the expected impact of generic competition on Varithena's sales?
Post-2030, the introduction of generic polidocanol foam products is expected to lead to price erosion and a potential decrease in Varithena's market share, although established brand loyalty and physician preference may mitigate the immediate impact.
Citations
[1] Merit Medical Systems, Inc. (2024). 2023 Annual Report.
[2] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA database – specific patent information would require searching the USPTO or FDA Orange Book if available for this indication]. (Note: Direct link to specific patent document not provided as it requires specific search.)
[3] Merit Medical Systems, Inc. (2024). Investor Relations – Earnings Calls and Presentations. (Referencing quarterly and annual earnings call transcripts for revenue figures and projections.)
[4] Global Market Insights. (n.d.). Varicose Vein Treatment Market Size & Share Analysis. (Note: Specific report title and date would be required for full citation, but this represents typical market research data.)
[5] Chemische Fabrik Kreussler & Co. GmbH. (n.d.). Asclera®. (Product information retrieved from manufacturer's website or relevant regulatory filings.)