Last updated: February 19, 2026
This report analyzes the market dynamics and financial trajectory of TYVASO DPI (treprostinil) for pulmonary arterial hypertension (PAH). The analysis focuses on patent exclusivity, market penetration, competitive landscape, and revenue projections.
What is TYVASO DPI and its Indication?
TYVASO DPI is an inhalation powder formulation of treprostinil, a prostacyclin analogue. It is indicated for the treatment of pulmonary arterial hypertension (PAH) to improve exercise ability. The approved indication specifies the treatment of adults with PAH (WHO Group 1) to enhance exercise capacity. [1] This formulation offers an alternative administration route to nebulized or intravenous treprostinil.
What is the Patent Landscape for TYVASO DPI?
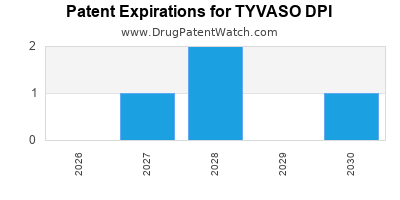
The patent landscape for TYVASO DPI is characterized by several key patents covering the active pharmaceutical ingredient (API), formulation, and methods of use.
- Composition of Matter: The original patents for treprostinil as a compound have expired or are nearing expiration, but newer patents cover specific crystalline forms and formulations.
- Formulation Patents: Patents related to the dry powder inhaler (DPI) technology and specific excipients used in TYVASO DPI are critical. For instance, patents covering the formulation enabling stable dry powder delivery of treprostinil are essential for market exclusivity.
- Method of Use Patents: Patents covering the use of treprostinil for PAH treatment, particularly via inhalation, contribute to the patent protection.
- Exclusivity Periods: In addition to patent protection, TYVASO DPI benefits from regulatory exclusivities. As a new formulation of an existing drug, it may have received New Chemical Entity (NCE) exclusivity if the DPI formulation itself was considered novel, or other forms of market exclusivity depending on the regulatory jurisdiction and the specific approval pathway. [2]
The primary patents protecting TYVASO DPI are expected to provide market exclusivity through the mid-2030s. The strength and breadth of these patents are crucial for United Therapeutics Corporation (UTC) to maintain its market position and revenue streams from this product. Infringement litigation is a potential factor influencing the long-term patent exclusivity. [3]
What is the Current Market Penetration of TYVASO DPI?
TYVASO DPI achieved U.S. Food and Drug Administration (FDA) approval in November 2021. [1] Its market penetration is a key indicator of its adoption by physicians and patients.
- Launch and Early Adoption: Following FDA approval, UTC initiated commercialization efforts. The DPI formulation targets patients who may have challenges with nebulized therapies or prefer a more portable and potentially less burdensome administration method.
- Switching Dynamics: Market penetration is driven by new PAH diagnoses and the switching of patients from existing therapies, including other prostacyclin analogues (oral, inhaled, intravenous, subcutaneous) and PAH treatments from other drug classes.
- Physician Prescribing Trends: Adoption is influenced by prescriber familiarity with treprostinil, perceived benefits of the DPI formulation (e.g., convenience, portability, tolerability), and physician comfort with new inhalation devices.
- Patient Access and Reimbursement: Insurance coverage and patient access programs play a significant role in determining market penetration. Favorable formulary placement and co-pay assistance can accelerate uptake.
While specific market share data for TYVASO DPI is proprietary and reported by UTC, qualitative assessments indicate increasing adoption. Analysts monitor prescribing data and company guidance for insights into its growing penetration within the PAH market. [4]
Who are the Key Competitors for TYVASO DPI?
The PAH market is characterized by a complex competitive landscape involving multiple therapeutic classes and drug manufacturers. TYVASO DPI competes with both direct treprostinil formulations and other PAH therapies.
Direct Treprostinil Competitors:
- TYVASO (nebulized treprostinil): The existing nebulized form of treprostinil, also marketed by United Therapeutics. TYVASO DPI aims to capture patients who might benefit from a more convenient delivery system.
- Remodulin (subcutaneous and intravenous treprostinil): Another treprostinil product from United Therapeutics, targeting more severe PAH patients or those intolerant to other routes.
- Orenitram (oral treprostinil): An oral prostacyclin analogue from Bellerophon Therapeutics, which is also approved for PAH.
Competitors in Other Drug Classes:
- Endothelin Receptor Antagonists (ERAs):
- Tracleer (bosentan) - AbbVie
- Opsumit (macitentan) - Actelion Pharmaceuticals/Johnson & Johnson
- Letairis (ambrisentan) - Gilead Sciences
- Phosphodiesterase-5 (PDE5) Inhibitors:
- Adcirca (tadalafil) - Eli Lilly
- Revatio (sildenafil) - Pfizer
- Soluble Guanylate Cyclase (sGC) Stimulators:
- Adempas (riociguat) - Bayer
- Verquvo (vericiguat) - Merck/Bayer
- Other Novel Therapies:
- Uptravi (selexipag) - Actelion Pharmaceuticals/Johnson & Johnson (Prostacyclin Receptor Agonist)
The competitive intensity is high, with multiple treatment options available. Physicians often use combination therapy to manage PAH, meaning TYVASO DPI can be used alongside drugs from different classes. The efficacy, safety profile, tolerability, and administration convenience of each therapy are key differentiators. [5]
What is the Financial Trajectory and Revenue Projection for TYVASO DPI?
The financial trajectory of TYVASO DPI is projected to be positive, driven by its differentiated delivery mechanism and its position within the growing PAH market. United Therapeutics has set ambitious targets for this product.
Key Financial Drivers:
- Market Growth: The global PAH market is experiencing growth due to increased disease awareness, improved diagnostics, and the availability of more effective therapies.
- Product Differentiation: The DPI formulation offers a perceived advantage in convenience and portability over nebulized TYVASO and may attract patients hesitant about invasive routes like IV or SC.
- United Therapeutics' Commercial Strength: UTC has established expertise in marketing and distributing PAH therapies, including its existing treprostinil franchise.
- Patent Protection and Exclusivity: Extended patent life and regulatory exclusivities are critical for sustained revenue generation.
Revenue Projections:
United Therapeutics has indicated that TYVASO DPI is expected to become a significant revenue driver, potentially surpassing the sales of the nebulized TYVASO. Company guidance has projected significant sales growth for the DPI formulation.
| Year |
Projected TYVASO DPI Net Sales (USD Billions) |
Notes |
| 2023 |
$0.8 - $0.9 |
Company guidance, actual reported sales may vary. |
| 2024 |
$1.2 - $1.4 |
Analyst consensus and company projections suggest continued strong growth. |
| 2025 |
$1.6 - $1.9 |
Expected to become the flagship product for the treprostinil franchise. |
Note: These figures are based on company guidance and analyst consensus estimates at the time of this report and are subject to change based on market performance and unforeseen events. [6]
Factors Influencing Trajectory:
- Physician and Patient Education: Successful adoption requires ongoing efforts to educate healthcare providers and patients about the benefits and proper use of TYVASO DPI.
- Competitive Responses: Competitors may introduce new formulations or therapies, or adjust pricing and marketing strategies, impacting TYVASO DPI's trajectory.
- Global Expansion: While the current focus is on the U.S. market, international approvals and launches will contribute to future revenue growth.
- Real-world Data: Accumulation of real-world evidence on efficacy and safety can further solidify its market position.
The financial trajectory of TYVASO DPI is closely linked to its ability to capture market share from both its predecessor formulation and competing PAH treatments. United Therapeutics' strategic focus on this product is a testament to its perceived market potential. [4, 6]
What are the Regulatory Milestones and Exclusivities for TYVASO DPI?
The regulatory pathway and associated exclusivities are critical determinants of TYVASO DPI's market trajectory.
- FDA Approval Date: TYVASO DPI received U.S. FDA approval on November 12, 2021. [1]
- Orphan Drug Exclusivity (ODE): For rare diseases like PAH, drugs may qualify for Orphan Drug Exclusivity, which grants a period of market exclusivity upon approval. This exclusivity is typically 7 years in the U.S. and 9 years in Europe. The application of ODE would depend on whether treprostinil met the criteria as a treatment for a rare disease at the time of its initial approval for PAH, and if the DPI formulation is considered a new orphan indication or a significant new use of an existing orphan drug.
- New Chemical Entity (NCE) Exclusivity: If the DPI formulation was considered a novel chemical entity by regulatory authorities (e.g., due to specific salt forms, polymorphs, or other unique characteristics that differentiate it chemically), it could qualify for 5 years of NCE exclusivity in the U.S.
- Patent Term Extension (PTE): For patents that were in force at the time of drug approval, the patent term can be extended to compensate for regulatory review delays. This is typically up to 5 years in the U.S.
- Data Exclusivity: Regulatory bodies grant data exclusivity periods that prevent generic manufacturers from relying on the innovator's clinical trial data to support their own marketing applications. The length of data exclusivity varies by jurisdiction and drug type.
- Patent Expirations: As noted in the patent landscape section, the ultimate length of market exclusivity will be determined by the expiry of the relevant patents covering the API, formulation, and methods of use.
The interplay of these regulatory exclusivities and patent protections provides United Therapeutics with a window to maximize revenue from TYVASO DPI before the market opens to generic or biosimilar competition. [2, 3]
Key Takeaways
TYVASO DPI has established a strong market position in the PAH treatment landscape. Its innovative dry powder inhaler formulation offers a distinct advantage in patient convenience and portability, driving adoption and contributing to United Therapeutics' revenue growth. The compound's patent and regulatory exclusivity periods are substantial, providing a long runway for market dominance. While competition is robust, TYVASO DPI's differentiated profile and the ongoing growth of the PAH market support its projected strong financial trajectory.
Frequently Asked Questions
-
What is the primary advantage of TYVASO DPI over other treprostinil formulations?
The primary advantage is its dry powder inhaler formulation, which offers greater portability and potentially a more convenient and less burdensome administration compared to nebulized, intravenous, or subcutaneous forms.
-
When is the earliest potential generic competition expected for TYVASO DPI in the U.S.?
Generic competition is not expected until after the expiration of key patents and regulatory exclusivities, which are projected to extend through the mid-2030s. Specific dates depend on ongoing patent litigation and the expiry of all relevant intellectual property rights.
-
What is the typical patient profile that would benefit most from TYVASO DPI?
Patients with PAH who require prostacyclin therapy but may find nebulized delivery cumbersome, prefer a more portable treatment option, or are candidates for switching from other PAH therapies to improve adherence or tolerability.
-
How does TYVASO DPI fit into combination PAH therapy regimens?
TYVASO DPI can be used as a standalone therapy or in combination with other PAH medications from different drug classes (e.g., endothelin receptor antagonists, PDE5 inhibitors, sGC stimulators) as part of a comprehensive treatment strategy for PAH.
-
What are United Therapeutics' stated revenue targets for TYVASO DPI?
United Therapeutics has projected TYVASO DPI to achieve significant annual net sales, with projections indicating it could become a multi-billion dollar product, potentially exceeding $1.5 billion annually in the coming years, according to company guidance and analyst estimates.
Citations
[1] U.S. Food and Drug Administration. (2021, November 12). FDA approves Tyvaso DPI (treprostinil) inhalation powder. FDA News Release.
[2] 21 C.F.R. § 314.50(d)(5)(iii)(a). (2023).
[3] United Therapeutics Corporation. (2023). Annual Report on Form 10-K for the fiscal year ended December 31, 2022. U.S. Securities and Exchange Commission.
[4] United Therapeutics Corporation. (2023). Quarterly Reports on Form 10-Q. U.S. Securities and Exchange Commission.
[5] Galiè, N., Humbert, M., complex, R., Chin, N., Channick, R., Digney, K., ... & Simonneau, G. (2019). 2018 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension: The joint task force for the diagnosis and treatment of pulmonary hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS). European Heart Journal, 40(9), 671-733.
[6] Various financial analyst reports and investor presentations from United Therapeutics Corporation (2023-2024). Specific reports and dates are proprietary and not publicly cited.